Abstract
Atopic dermatitis (AD) is a common, chronic, and recurrent inflammatory skin condition that affects a considerable portion of the population, and is particularly prevalent among children. The development of AD is influenced by environmental and genetic factors, which cause epidermal barrier dysfunction, immune dysregulation, and dysbiosis. In immune dysregulation, there is excessive production of cytokines. Among the cytokines, interleukin (IL)-13 plays a major role in the pathogenesis of AD. Searching for new and more selective treatments for moderate-to-severe cases is important because of the considerable effect of AD on the quality of life. Tralokinumab and lebrikizumab are selective IL-13 inhibitors that have demonstrated safety and efficacy as treatment options for AD in phase III trials. Tralokinumab is approved for use in Europe and the USA, while lebrikizumab is approved only in Europe. Cendakimab, which is another IL-13 selective inhibitor, has shown promising results in phase II trials, providing safe and effective outcomes. Eblasakimab, which disrupts IL-13 and IL-4 signaling pathways, is currently in phase II trials following well-tolerated administration in phase I studies. This narrative review aims to outline the current state of knowledge regarding the effectiveness and safety of these four biologic agents targeting IL-13 signaling.
Keywords
Introduction
Atopic dermatitis (AD), also known as atopic eczema, is a prevalent, chronic, and recurrent inflammatory skin disease. While AD frequently manifests in childhood, it can persist into adulthood, affecting up to 25% of children and 2% to 10% of adults.1,2 A family history of atopy is the main risk factor for AD, and it is estimated to increase a child’s risk of developing AD by 1.5 fold. This risk increases to approximately 3 fold if one parent has AD and up to 5 fold if both parents are affected by AD. 3
Itchy, dry, and erythematous plaques are the main manifestations of AD. These symptoms often lead to sleep disturbances, decreased productivity, lower self-esteem, social withdrawal, and in severe cases, depression and suicidal thoughts.4,5 AD is associated with various other atopic conditions, such as asthma, allergic rhinitis, food allergies, and eosinophilic esophagitis. Additionally, individuals with AD appear to have a higher susceptibility to cardiovascular issues and infections. This increased vulnerability is attributed to factors, such as compromised skin barrier function, immune dysregulation, reduced levels of antimicrobial peptides, enhanced bacterial colonization and skin infection, and the use of immunosuppressive medications. 6
AD is an immune-mediated disorder, which is driven by the aberrant activation of type 2 T helper cells (Th2) and type 2 innate lymphoid cells, which are accompanied by elevated concentrations of inflammatory cytokines, notably interleukin-4 (IL-4) and interleukin-13 (IL-13). 7 In recent years, the introduction of targeted biologic and small molecule therapies has greatly expanded the treatment options for individuals with moderate-to-severe AD. Dupilumab was the first biologic agent to be approved for the treatment of moderate-to-severe AD in 2017. Dupilumab acts as an inhibitor of IL-4 receptor α (IL-4Rα), effectively hindering IL-4 and IL-13 signaling pathways and inhibiting downstream inflammatory processes. 8
Although IL-4 and IL-13 are implicated in the pathogenesis of AD, IL-13 has been suggested as the primary cytokine involved. This possibility has led to an increased research focus on IL-13 as a potential therapeutic target for treating AD.
This narrative review aims to outline the current state of knowledge regarding the effectiveness and safety of four biologic agents, tralokinumab, lebrikizumab, cendakimab, and eblasakimab, which target IL-13 signaling. Additionally, this review examines the role of IL-13 in the pathogenesis of AD.
Methods
A comprehensive literature search was conducted from 2011 to April 2024 to investigate the pathogenic role of IL-13 and the efficacy of IL-13 inhibitors in treating AD. This search was conducted in the PubMed, Google Scholar, Science Direct, and clinicaltrials.gov databases using the following search terms, alone or in combination: “atopic dermatitis,” “monoclonal antibodies,” “IL-13,” “tralokinumab,” “lebrikizumab,” “cendakimab,” and “eblasakimab,” with a focus on titles, abstracts, and article bodies.
The inclusion criteria encompassed original and review articles published in English, with a specific emphasis on the pathogenic mechanisms of IL-13 and the therapeutic applications of monoclonal antibodies against IL-13 in the context of AD. The bibliographic references of included articles were scrutinized to identify additional pertinent studies. This search yielded 47 articles and 21 clinical trials in clinicaltrials.gov.
Role of IL-13 in the pathogenesis of AD
The development of AD involves environmental and genetic factors, which interact to induce inflammation, dysbiosis, and immune dysregulation within the epidermis. This interaction enhances transepidermal water loss and promotes the penetration of irritants, microbes, and allergens into the skin.7,9 These antigens trigger an abnormal type 2 immune response, prompting naïve T cells to commit to the Th2 lineage. This process results in an excessive production of cytokines crucial to the atopic manifestations of AD and itching, specifically IL-4, IL-5, IL-13, and IL-31. IL-4 and IL-13 contribute to dysfunction in the epidermal barrier by inducing the production of immunoglobulin E, attracting eosinophils, enhancing Th2 cell differentiation, and reducing filaggrin expression. 7
IL-13 plays a major role in the pathogenesis of AD, with gene polymorphisms associated with a higher risk of AD.10,11 IL-13 is a cytokine produced by Th2 cells, natural killer cells, mast cells, basophils, eosinophils, and type 2 innate lymphoid cells. IL-13 shares structural similarities with IL-4, although their sequence homology is only 25%. The receptor for IL-13 is a heterodimeric complex comprising IL-4Rα and IL-13 receptor α1 (IL-13Rα1). Upon heterodimerization, this complex activates STAT6 signaling, which is crucial for initiating type 2 allergic responses. Additionally, IL-13 binds to IL-13Rα2, which has a limited cytoplasmic domain and functions as a decoy receptor, without mediating signaling. The expression of IL-13Rα2 can be induced by various pro-inflammatory cytokines, including IL-13, IL-4, and tumor necrosis factor, suggesting a negative feedback mechanism.11,12
Overexpression of IL-13 compromises the integrity of the epithelial barrier by downregulating crucial components, including lipids and proteins (e.g., filaggrin, loricrin, and involucrin). Skin biopsy samples from patients with AD show notable overexpression of IL-13 in lesion and non-lesion skin, whereas only mild overexpression of IL-4 is detectable in 40% of AD lesions.13–15
IL-13 inhibition in AD
IL-13 is a promising therapeutic target for effectively managing AD because of its substantial efficacy potential and minimal toxicity concerns.5,15 The first biologic agent approved for AD, dupilumab, targets IL‐4Rα, thereby blocking the IL-13 pathway through the type 1 receptor and the IL-4 pathway through the type 1 and type 2 receptors. 7 Randomized, controlled trials and real-world studies have demonstrated the efficacy and safety of subcutaneous (SC) dupilumab for treating AD. The safety profile of dupilumab is well documented, and the most relevant adverse events (AEs) are conjunctivitis, facial redness, arthralgia, and an immunological shift towards Th1/Th17.16–19
Four other distinct monoclonal antibodies targeting IL-13 signaling are being developed or have already been approved. Tralokinumab has obtained approval from Health Canada, the Food and Drug Administration (FDA), and the European Medicines Agency (EMA), and is currently available for individuals with moderate-to-severe AD. Lebrikizumab has received approval from the EMA for treating AD, but it has yet to receive FDA approval. Cendakimab and eblasakimab are currently undergoing investigation in phase II trials.20–22
Tralokinumab
Tralokinumab, a fully human immunoglobulin 4 monoclonal antibody originating from a human phage display library, was the first biologic approved in the European Union (EU) and the USA with a mechanism of targeting and selectively neutralizing IL-13. Tralokinumab effectively prevents IL-13 from binding to IL-13Rα1 (interleukin-13 receptor α1) and IL-13Rα2 receptors (Figure 1). IL-13Rα2 receptor, which is characterized by a short cytoplasmic tail lacking signaling motifs, serves as a decoy with an anti-inflammatory function by internalizing excess IL-13. By blocking IL-13-mediated signaling downstream of IL-4Rα/IL-13Rα1 heterodimerization, tralokinumab not only inhibits IL-13 activity but also potentially disrupts the endogenous regulation of IL-13 mediated by IL-13Rα2.7,22–24

(a) Tralokinumab binds to the cytokine IL‐13 at an epitope that overlaps with the binding site of IL‐13Rα, preventing IL‐13 from binding to both IL‐13Rα1 and IL‐13Rα2 and (b) Lebrikizumab binds to IL‐13 at an epitope that overlaps with the binding site of IL‐4Rα, preventing heterodimerization of the IL‐4Rα/IL‐13Rα1 subunits. IL‐13 can still bind to IL‐13Rα2. IL-4, interleukin-4; IL-13, interleukin-13; IL-4Rα, interleukin-4 receptor α; IL-13Rα1, interleukin-13 receptor α1. The image was retrieved from an article by Gonçalves et al. 24 This image is licensed under the Creative Commons Attribution-Non-commercial 4.0 International License. This license permits any non-commercial use, sharing, adaptation, distribution, and reproduction in any medium or format, as long as appropriate credit is given to the original author(s) and the source, and a link to the Creative Commons license is provided. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc/4.0/ (accessed 17 August 2024).
A phase I trial, which was a single-blind, randomized controlled study, examined the effects of SC tralokinumab at doses of 150, 300, or 600 mg in 30 healthy Japanese adults (Table 1). 25 This study showed dose-dependent increases in the observed maximum concentration, area under the serum concentration-time curve from time 0 to the last measurable time point (AUC(0-t)), and area under the serum concentration-time curve from time 0 to infinity (AUC(0-inf)) with tralokinumab administration. The incidence and nature of AEs were consistent across all dosage levels, and injection site pain was the most predominant treatment-related AE, reported by 83% to 100% of individuals across all treatment groups. The authors of this trial concluded that a single SC dose of tralokinumab ranging from 150 to 600 mg showed a favorable safety and pharmacokinetic profile in adults. 22
Completed clinical trials that investigated tralokinumab for the treatment of AD.
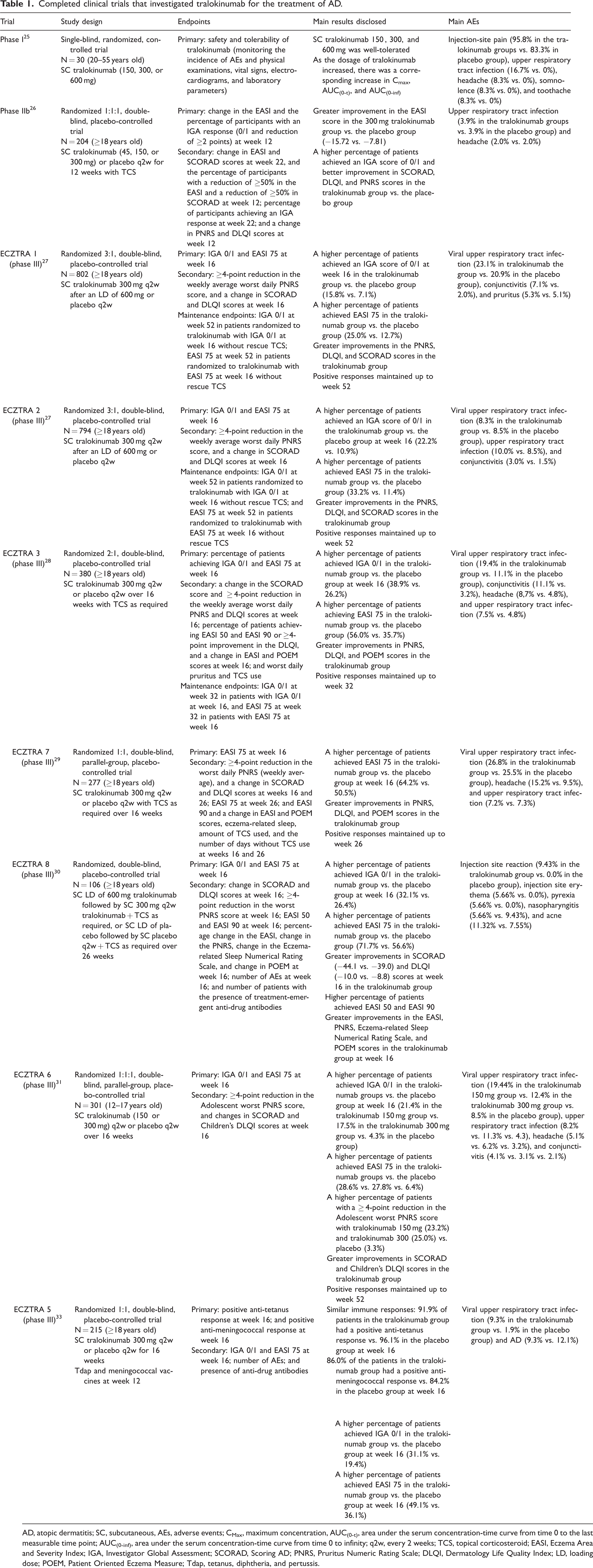
AD, atopic dermatitis; SC, subcutaneous, AEs, adverse events; CMax, maximum concentration, AUC(0-t), area under the serum concentration-time curve from time 0 to the last measurable time point; AUC(0-inf), area under the serum concentration-time curve from time 0 to infinity; q2w, every 2 weeks; TCS, topical corticosteroid; EASI, Eczema Area and Severity Index; IGA, Investigator Global Assessment; SCORAD, Scoring AD; PNRS, Pruritus Numeric Rating Scale; DLQI, Dermatology Life Quality Index; LD, loading dose; POEM, Patient Oriented Eczema Measure; Tdap, tetanus, diphtheria, and pertussis.
In a phase IIb trial, a cohort of 204 healthy adults with moderate-to-severe AD received treatment comprising 45, 150, or 300 mg of SC tralokinumab or placebo, administered every 2 weeks (q2w) over a span of 12 weeks with topical corticosteroid (TCS) (Table 1). 26 Individuals in the 300 mg tralokinumab group showed notable enhancement in the Eczema Area and Severity Index (EASI) score. Additionally, in this group, a higher proportion of patients achieved an Investigator Global Assessment (IGA) score of 0 or 1 and marked improvements in the Scoring Atopic Dermatitis (SCORAD), Dermatology Life Quality Index (DLQI), and Pruritus Numeric Rating Scale (PNRS) scores relative to those in the placebo group. The most significant clinical improvement was observed in the 300 mg cohort, with the most frequently reported AE of upper respiratory infections. Tralokinumab showed favorable safety and tolerability profiles, as well as early and sustained improvement in disease symptoms in patients with moderate-to-severe AD. 22
In the ECZTRA 1 (802 patients enrolled) and ECZTRA 2 (794 patients enrolled) trials, two extensive 52-week-long, randomized, double-blind, placebo-controlled, phase III studies were conducted to assess the safety and efficacy of tralokinumab (Table 1). 27 In these trials, the patients received SC tralokinumab 300 mg q2w or a placebo. Only the patients who achieved an IGA score of 0 or 1 and/or EASI 75 (proportion of patients achieving ≥75% improvement from baseline) with tralokinumab by week 16 underwent rerandomization to receive tralokinumab q2w, every 4 weeks (q4w), or placebo for an additional 36 weeks. In both clinical trials, patients who received tralokinumab showed higher rates of achieving an IGA score of 0/1 and EASI 75 at week 16 than those who received placebo. Additionally, patients treated with tralokinumab experienced improvements in pruritus, sleep, the DLQI, and SCORAD, with positive responses maintained up to week 52. 22
In the ECZTRA 3 study, a phase III trial was designed to evaluate the effectiveness and safety of tralokinumab in combination with TCS for patients who were diagnosed with moderate-to-severe AD and required systemic therapy (Table 1). 28 A double-blind methodology was used, and patients were randomly allocated in a 2:1 ratio to receive SC tralokinumab at a dosage of 300 mg or a placebo q2w, along with a TCS as required, over 16 weeks. This trial showed a notable increase in the proportion of patients who achieved an IGA score of 0 or 1 and EASI 75 at the end of the 16-week treatment period in those administered tralokinumab compared with those who had placebo. Following the initial treatment phase, a subset of patients from both arms of the study underwent rerandomization to receive SC tralokinumab q2w or q4w for an additional 16 weeks. A high percentage of patients maintained their positive response to treatment. A total of 89.6% of patients who received tralokinumab q2w sustained their IGA responses at 32 weeks. Additionally, 92.5% of patients who received tralokinumab q2w sustained EASI 75 responses at 32 weeks. A total of 77.6% and 90.8% of patients who received tralokinumab q4w sustained their IGA and EASI 75 responses at 32 weeks, respectively. 22
ECZTRA 7 was a phase III trial that evaluated the effectiveness and safety of tralokinumab combined with TCS in adults with severe AD who had contraindications for cyclosporin A (CyA) treatment or previously failed CyA treatment (Table 1). 29 In this multicenter, randomized, double-blind study, 277 adults received tralokinumab or placebo injections q2w for 26 weeks, along with TCS as required. A higher number of patients treated with tralokinumab plus TCS attained the primary efficacy target of EASI 75 by week 16 than those who received placebo plus TCS. This study suggests that combining tralokinumab with TCS on an as-needed basis notably enhances the signs and symptoms of AD in adults with severe disease refractory to CyA treatment. 22
ECZTRA 8 was a randomized, double-blind, placebo-controlled trial involving 106 participants (Table 1). 30 The primary endpoints of this trial were the percentage of patients who achieved an IGA score of 0 or 1 and EASI 75 at week 16. The participants received an SC 500-mg loading dose (LD) of tralokinumab followed by SC 300 mg q2w with TCS as required or a placebo. This trial showed the efficacy of tralokinumab, with a higher percentage of patients who achieved an IGA score of 0 or 1 (32.1% vs. 26.4%) and EASI 75 (71.7% vs. 56.6%) than those who had placebo.
ECZTRA 6 was a recent phase III trial, which evaluated tralokinumab monotherapy versus placebo in adolescents with moderate-to-severe AD who required systemic treatment (Table 1). 31 This trial included 301 adolescents aged 12 to 17 years who received SC tralokinumab 150 or 300 mg or placebo q2w for 16 weeks. Tralokinumab resulted in an improvement in various patient-reported outcomes, such as eczema-related sleep, hospital anxiety, depression, and PNRS scores, compared with placebo. Overall, tralokinumab was well-tolerated and its efficacy and safety profiles in adolescents were similar to those observed in phase III trials involving adults.
Another phase III clinical trial evaluated the effectiveness of tralokinumab in adolescents (Table 2). 32 This trial involved a single-arm design and focused on individuals aged 12 years and older. The participants received an initial SC dose of tralokinumab 600 mg, followed by self-administered doses of 300 mg q2w for a total duration of 14 weeks. This trial aimed to assess the safety and efficacy of tralokinumab in this age group. Although tralokinumab has already been approved by the FDA and EMA for treating AD in adults, it is not yet approved for use in adolescents. 22
The ECZTRA 5 trial was a randomized, double-blind, placebo-controlled phase II study, which aimed to investigate the potential effect of tralokinumab on the immune response to vaccines (Table 1). 33 Over 16 weeks, 107 adult patients were administered tralokinumab 300 mg, while 108 received a placebo q2w. At week 12, all participants received the tetanus, diphtheria, and pertussis vaccine (Tdap) and the meningococcal vaccine, and the primary endpoints focused on positive immune responses to either vaccine during weeks 12 and 16. This trial showed that the tralokinumab and placebo groups exhibited similar immune responses to those with the Tdap (91.9% vs. 96.1%) and meningococcal (86.0% vs. 84.2%) vaccines at week 16. This finding indicates that tralokinumab-treated adults with moderate-to-severe AD can continue their treatment without interruption because standard non-live vaccines have proven effective and safe in this population. 22
Some retrospective analyses have indicated the efficacy of tralokinumab in treating AD in patients who previously failed other systemic treatments. One study on 12 adults who did not achieve EASI 50 with 16-week dupilumab treatment showed that tralokinumab led to a rapid improvement, with patients reaching EASI 75 within 8 weeks and experiencing notable reductions in itch and sleep disturbances. 34 Another analysis of 10 patients who received tralokinumab for at least 26 weeks, after unsuccessful trials with CyA and dupilumab, demonstrated superior outcomes compared with tralokinumab phase III trials. 35 These superior outcomes included greater itch reduction and a higher proportion of patients who achieved EASI 75 and IGA 0/1. Additionally, a multicenter, retrospective study of 85 adult patients with severe AD showed that tralokinumab treatment for 16 weeks resulted in improvements in the EASI, SCORAD, and peak PNRS scores. 36 Additionally, a higher proportion of systemic treatment-naive patients showed achievement of EASI 75 than those with prior systemic therapy experience. These findings highlight tralokinumab’s real-world effectiveness in providing short-term relief for AD symptoms in patients who are unresponsive to other systemic treatments. 22
The ECZTRA 4 trial examined whether tralokinumab alters the metabolism of specific cytochrome P450 substrates in adults diagnosed with moderate-to-severe AD (Table 2). 37 This trial involved 14-week treatment with tralokinumab administration of a single dose (SD). This was an open-label, multicenter, drug–drug interaction study aimed at investigating the effects of tralokinumab on the pharmacokinetics of selected cytochrome P450 substrates in adult subjects with moderate-to-severe AD. The TraSki phase II trial included 16 patients who received an SC 600-mg LD of tralokinumab followed by 300 mg q2w (Table 2). 38 The primary outcome of this trial is to study skin barrier function, specifically the change in transepidermal water loss at one non-lesional and one lesional marker skin site at week 16. As of the date of the bibliographic review search, no results from these two trials have been posted.
Ongoing clinical trials investigating tralokinumab, lebrikizumab, and eblasakimab for the treatment of AD.

AD, atopic dermatitis.
Overall, tralokinumab demonstrated a favorable safety profile across all studies, with no important life-threatening AEs reported. The most frequent systemic AEs observed included upper respiratory tract infection, headache, somnolence, toothache, conjunctivitis, pruritus, pyrexia, nasopharyngitis, and acne. Locally, cases of an injection site reaction with pain and erythema were reported.25–31,33
Lebrikizumab
Lebrikizumab is a humanized monoclonal antibody and shows high affinity for soluble IL-13 by binding to its non-receptor binding domain. When IL-13 is bound by lebrikizumab, it retains the capacity to form a complex with IL-13Rα1, but impedes its heterodimerization with IL-4Rα, thereby blocking signal transduction. Consequently, lebrikizumab effectively inhibits the formation of the IL-4Rα/IL-13Rα1 signaling complex, while concurrently modulating endogenous IL-13 through the stimulation of IL-13Rα2 (Figure 1).7,39
Two phase II trials studied the safety and efficacy of lebrikizumab for the treatment of moderate-to-severe AD.40–42 In the TREBLE trial, the efficacy and safety of lebrikizumab as an adjunct therapy to TCS were evaluated in 209 adults with AD (Table 3). 40 This trial demonstrated that lebrikizumab 125 mg administered q4w for 12 weeks improved the primary endpoint of achieving a 50% reduction in the EASI score compared with placebo (82.4% vs. 62.3%). Additionally, the lebrikizumab group showed significant improvements in secondary endpoints, including EASI 75, SCORAD 50, and an IGA score of 0/1. While SD groups did not show significant responses at week 12, the results on the 250-mg SD group suggested a potential dose–response relationship. A phase IIb trial investigated the efficacy, dose–response relationship, and safety of lebrikizumab monotherapy, with TCS used if necessary, in 280 adults with AD who were unresponsive to standard topical treatment (Table 3). 42 The patients were randomized into four arms: lebrikizumab with an LD of 250 mg followed by 125 mg q4w, lebrikizumab with an LD of 500 mg followed by 250 mg q4w, lebrikizumab with an LD of 500 mg at baseline and at week 2 followed by 250 mg q2w and placebo q2w. In the primary endpoint, a dose-dependent reduction in EASI scores across all lebrikizumab groups compared with placebo was observed at week 16, with significant differences observed as early as week 4. Specifically, the lebrikizumab 250 mg groups showed superior response rates in secondary endpoints, including IGA 0/1 EASI 50, EASI 75, and EASI 90 responses. Furthermore, placebo-treated patients required approximately three times more rescue TCS use, which was initiated earlier and sustained for a longer duration, than in lebrikizumab-treated patients.
Completed clinical trials that investigated lebrikizumab for the treatment of AD.

AD, atopic dermatitis; SD, single dose; q4w, every 4 weeks; TCS, topical corticosteroid; EASI, Eczema Area and Severity Index; IGA, Investigator Global Assessment; SCORAD, Scoring AD; LD, loading dose; q2w, every 2 weeks; PNRS, Pruritus Numeric Rating Scale; DLQI, Dermatology Life Quality Index; SC, subcutaneous; AEs, adverse events; BSA, body surface area; COVID-19, coronavirus disease 2019.
Four major phase III trials have been completed to thoroughly assess the efficacy and safety profile of lebrikizumab (Table 3). These trials were ADhere, ADvocate 1, ADvocate 2, and ADore.41,43–46 The ADhere trial was a multicenter, double-blind, placebo-controlled study of 211 adolescents aged 12 to 16 years and adults (Table 3). 43 The participants were randomized to receive SC lebrikizumab with an LD of 500 mg at baseline and week 2, followed by 250 mg q2w or a placebo. Both groups received treatment in combination with a low- to mid-potency TCS. The primary endpoints included achieving an IGA score of 0 or 1 with a reduction of at least 2 points from baseline and EASI 75 at 16 weeks. The secondary endpoints included improvements in the EASI score, PNRS score, sleep loss due to pruritus, and the DLQI score. At week 16, improvement was observed in the lebrikizumab plus TCS group compared with the placebo group. Specifically, 41.2% of patients in the lebrikizumab group achieved an IGA score of 0 or 1 with a reduction of at least 2 points from baseline compared with 22.1% in the placebo group. Additionally, 69.5% of patients in the lebrikizumab group achieved EASI 75 compared with 42.2% in the placebo group. Clinically meaningful improvements across all key secondary endpoints were also observed in the lebrikizumab group as early as 4 weeks.
In the ADvocate 1 and ADvocate 2 trials, lebrikizumab administered q2w showed improvements in primary and secondary endpoints, including skin clearance, pruritus, sleep interference, and quality of life, compared with placebo (Table 3).44,45 At week 16, a higher proportion of lebrikizumab-treated patients achieved an IGA score of 0 or 1 with a reduction of at least 2 points from baseline compared with placebo. Similarly, more lebrikizumab-treated patients achieved EASI 75 and EASI 90 responses than those with placebo at week 16. During the maintenance period, patients who maintained their response to lebrikizumab q2w or q4w showed sustained improvements in IGA scores, EASI 75 and EASI 90 responses, and PNRS scores compared with those in the withdrawal arm.
The ADore study was a single-arm trial that evaluated the safety and efficacy of lebrikizumab in adolescents with moderate-to-severe AD (Table 3). 46 The patients received two doses of lebrikizumab of 250 mg initially and at week 2, followed by a single injection q4w from weeks 4 to 52. Throughout the 52-week treatment period, lebrikizumab showed clinically meaningful improvements in skin clearance, which were noticeable as early as the first assessment at week 4, and an increasing number of patients experienced improvement over time. The positive results of the study indicate that treating moderate-to-severe AD in adolescents by targeting IL-13 with lebrikizumab is a promising approach. Clinically significant improvements were observed across all primary and secondary endpoints in the four above-mentioned studies.
The ADopt trial was a phase III randomized, double-blind, placebo-controlled study of a cohort of 254 participants that aimed to investigate the effect of lebrikizumab on vaccine immune responses in adults diagnosed with moderate-to-severe AD (Table 3). 47 The primary endpoints were a positive anti-tetanus response and a positive anti-meningococcal response at week 16. This study showed that the immune response was similar in both groups. A total of 73.6% of participants who took lebrikizumab achieved a positive anti-tetanus response compared with 73.4% of participants who received placebo. A positive anti-meningococcal immune response was achieved by 86.9% of the participants who took lebrikizumab and 75.0% of participants who had placebo.
The ADhere-J trial was a randomized, double-blind, placebo-controlled study, which was designed to assess the efficacy and safety of lebrikizumab combined with TCS in Japanese individuals with moderate-to-severe AD (Table 3). 48 In the lebrikizumab treatment groups, a higher percentage of patients achieved considerable improvement than those in the placebo group across all of the measures. Specifically, a greater proportion of patients achieved EASI 75, with 33.4% in the 250 mg q2w group and 29.1% in the 250 mg q4w group, compared with only 6.1% in the placebo group. Additionally, a higher percentage of patients in the lebrikizumab groups achieved IGA scores of 0 or 1 with at least a 2-point reduction from baseline. A total of 51.2% of patients in the 250 mg q2w group and 47.2% in the 250 mg q4w group achieved IGA 0/1 compared with 13.4% in the placebo group.
ADjoin is an ongoing phase III study focusing on evaluating the long-term efficacy and safety of lebrikizumab (Table 2). With a participant pool of 1000 individuals, the study spans 110 weeks. The primary aim of this study is to assess the long-term effectiveness of lebrikizumab and to monitor any safety concerns that may arise over the extended period. This long-term extension study encompasses patients who have completed their involvement in a previous lebrikizumab trial. 49
Overall, lebrikizumab showed a favorable safety profile across all studies, with no major life-threatening AEs. The most frequent systemic AEs observed included upper respiratory tract infections, nasopharyngitis, headache, coronavirus disease 2019 (COVID-19), oral herpes, and conjunctivitis. Locally, injection site reactions were reported, with frequencies ranging from 1.3% to 5.7% in the lebrikizumab groups.40,42–48
Lebrikizumab was approved by the EMA for use in AD in November 2023 but it is still waiting for FDA approval.20,21
Cendakimab
Cendakimab (RPC-4046) is a recombinant humanized anti-IL-13 monoclonal antibody. Cendakimab binds to IL-13 at an epitope overlapping with the binding sites of the IL-13Rα1 and IL-13Rα2 subunits, thereby preventing their interaction and blocking subsequent signaling (Figure 2). 50
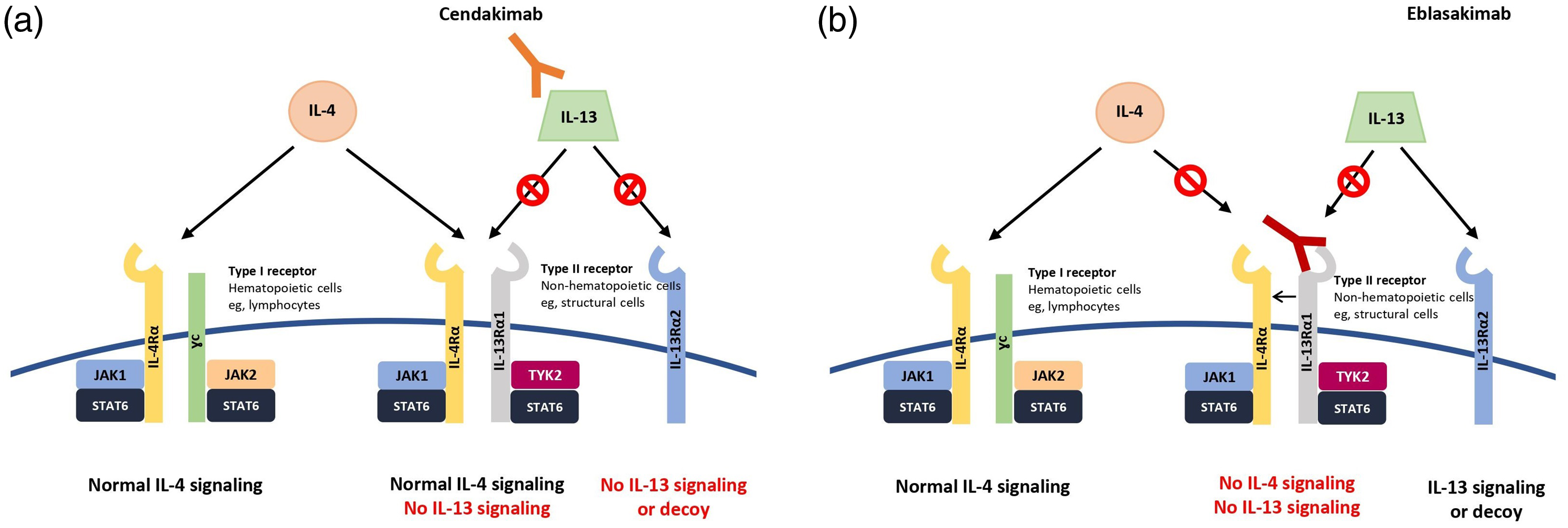
a) Cendakimab binds to the cytokine IL‐13, preventing IL‐13 from binding to IL‐13Rα1 and IL‐13Rα2. b) Eblasakimab binds to IL-13Rα1, blocking IL-4 and IL-13 signaling through the type 2 receptor. This prevents IL‐13 from binding to both IL‐13Rα1 and IL‐13Rα2, and IL-4 to IL‐13Rα1. IL‐4 can still bind and signal through the type 1 receptor. IL-4, interleukin-4; IL-13, interleukin-13; IL-4Rα, interleukin-4 receptor α; IL-13Rα1, interleukin-13 receptor α1. The image was retrieved from Gonçalves et al. 2021. 24 This image is licensed under the Creative Commons Attribution-Non-commercial 4.0 International License. This license permits any non-commercial use, sharing, adaptation, distribution, and reproduction in any medium or format, as long as appropriate credit is given to the original author(s) and the source, and a link to the Creative Commons license is provided. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc/4.0/ (accessed on 17 August 2024).
A phase II multicenter, randomized, double-blind, parallel-group, placebo-controlled trial (NCT04800315) evaluated the effectiveness and safety of three dosing regimens of cendakimab in adult participants with moderate-to-severe AD (Table 4).51,52 This study consisted of four experimental groups. In group 1, the participants received SC cendakimab 720 mg once weekly (qw) for 16 weeks. In group 2, the participants were administered SC cendakimab 720 mg q2w, with placebo administered alternately q2w. The participants in group 3 received SC cendakimab 360 mg q2w, with placebo administered SC weekly. The participants in the placebo group received SC placebo qw. The primary outcome of the study was the mean percentage change in the EASI score from baseline to week 16. This trial showed a greater decrease in the EASI score in the cendakimab 720 mg qw group (−84.41%) than in the cendakimab 720 mg q2w group (−76.03%), cendakimab 360 mg q2w group (−78.93%), and the placebo group (−62.7%). The highest proportion of patients who achieved an IGA score of 0 or 1 was observed in the cendakimab 360 mg q2w group (38.2%) compared with 33.3% in the 720 mg qw group and 24.4% in the 720 mg q2w group. The most reported AEs in this study were upper respiratory tract infection, allergic conjunctivitis, COVID-19, and headache.
Completed clinical trials that investigated cendakimab for the treatment of AD
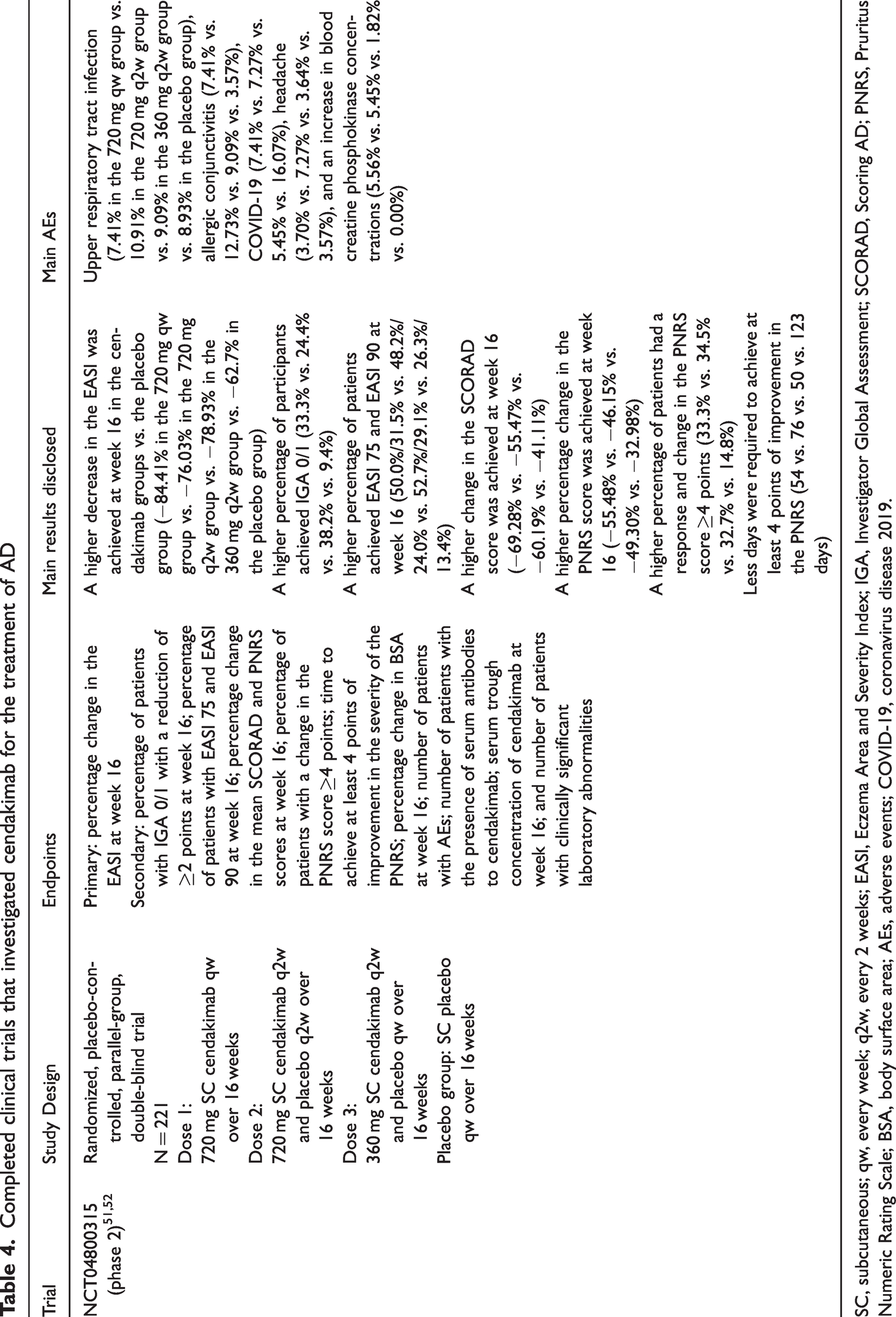
SC, subcutaneous; qw, every week; q2w, every 2 weeks; EASI, Eczema Area and Severity Index; IGA, Investigator Global Assessment; SCORAD, Scoring AD; PNRS, Pruritus Numeric Rating Scale; BSA, body surface area; AEs, adverse events; COVID-19, coronavirus disease 2019.
The development of cendakimab has been discontinued, even after successfully completing its phase II trial. Despite achieving the primary endpoint, the company that produced cendakimab noted that the treatment landscape for AD is highly competitive, and cendakimab did not provide a considerable advantage over existing therapies. 53
Eblasakimab
Eblasakimab is a monoclonal antibody designed to specifically bind with high affinity to IL-13Rα1. Unlike other monoclonal antibodies, the mechanism of eblasakimab involves disrupting the signaling pathways of IL-13 and IL-4 (Figure 2).54,55
A phase Ia study, which featured an open-label, single ascending dose design, examined the mechanistic actions of eblasakimab and its effect on IL-13Rα1 signaling (Table 5). 55 In this study, 44 healthy male volunteers of Asian descent received single ascending doses of eblasakimab via intravenous or SC injection. The data obtained showed that eblasakimab was safe and well tolerated, regardless of the route of administration. Notably, eblasakimab effectively blocked IL-13Rα1 and impeded IL-13-induced activation of STAT6 (at an SD of 3 mg/kg intravenously and 300 mg SC), which is a pivotal transcription factor responsible for the expression of Th2 effector cytokines. In the intravenous cohorts, the most frequent AEs were upper respiratory tract infection, decreased appetite, headache, oropharyngeal pain, and pyrexia. In the SC cohorts, headache, elevated C-reactive protein concentrations, and pruritus at the injection site were the most commonly related AEs.
Completed clinical trials that investigated eblasakimab for the treatment of AD.

In the IV route (6 cohorts), the doses were 0.1, 0.3, 1, 3, 10, and 20 mg/kg (optional). In the SC route (4 cohorts), the doses were 75, 150, 300, and 600 mg (optional).
SD, single dose; IV, intravenous; SC, subcutaneous; AEs, adverse events; AUC(0-t), area under the serum concentration-time curve from time 0 to the last measurable time point; CMax, maximum concentration, AUC(0-inf), area under the serum concentration-time curve from time 0 to infinity; BSA, body surface area, EASI, Eczema Area and Severity Index; IGA, Investigator Global Assessment; PNRS, Pruritus Numeric Rating Scale; POEM, Patient Oriented Eczema Measure; q2w, every 2 weeks; q4w, every 4 weeks; AD, atopic dermatitis; SCORAD, Scoring AD; DLQI, Dermatology Life Quality Index; SD-NRS, Sleep Disturbance Numerical Rating Scale.
A phase Ib randomized, double-blinded study evaluated the efficacy of eblasakimab in patients with AD (Table 5). The preliminary data included 52 patients who were randomized to receive eblasakimab at doses of 200, 400, or 600 mg compared with a placebo group. After 8 weeks of treatment, considerable improvement was observed in the eblasakimab groups compared with the placebo group. A reduction in the EASI score was notable across all dose groups as follows: 50% in the 200 mg group, 63% in the 400 mg group, and 61% in the 600 mg group compared with 32% in the placebo group. Similarly, EASI 50 and EASI 75 responses were higher in the eblasakimab groups than in the placebo group. The peak PNRS score was lower in the 600 mg eblasakimab group than in the placebo group. A further analysis indicated that the 400 mg and 600 mg doses of eblasakimab produced greater clinical responses than the 200 mg dose. At week 8, eblasakimab 600 mg resulted in considerable improvement in the mean percentage change in the EASI regarding the proportion of patients achieving EASI 50 and EASI 75 compared with placebo. This study also showed improvements in the PNRS and Patient Oriented Eczema Measure (POEM) scores and sleep disturbance with eblasakimab treatment compared with placebo. The most reported AEs were pruritus, injection site erythema, and injection site swelling. 56
Overall, the phase I studies suggested that the administration of eblasakimab to adults with moderate-to-severe AD was well tolerated. The data from these trials demonstrate considerable clinical enhancement compared with placebo, as shown by physician- and patient-reported outcomes. These findings highlight the promising therapeutic potential of eblasakimab in managing AD. Consequently, further investigation through phase II trials is warranted to better understand the efficacy and safety profile of eblasakimab in larger patient populations and to determine its potential as a treatment option for this inflammatory skin condition.
TREX-AD (NCT05158023) was a phase IIb, randomized, double-blind, placebo-controlled study that highlighted eblasakimab’s efficacy and potential for monthly dosing from initiation (Table 5).57,58 This study included a post-hoc analysis of patients with severe AD, representing 63% of the intent-to-treat population. A 600-mg monthly dose of eblasakimab for 16 weeks led to a 74.5% reduction in EASI scores compared with 38.0% with placebo. Additionally, 53.6% of patients achieved EASI 75 compared with 12.9% on placebo. Eblasakimab demonstrated rapid efficacy, with a considerable improvement in EASI scores by week 4. The study's primary endpoint showed statistical significance across three dosing arms. Eblasakimab was generally well-tolerated, with low rates of conjunctivitis and injection site reactions.
TREX-DX (NCT05694884) is an ongoing phase II multicenter, randomized, double-blind, placebo-controlled, parallel-arm clinical trial. This trial aimed to assess the effectiveness and safety of eblasakimab in individuals with moderate-to-severe AD who stopped dupilumab treatment owing to non-response, intolerance, or AEs (Table 2). The trial spans a 16-week treatment period followed by an 8-week follow-up period for 24 weeks. Approximately 75 participants are estimated to be enrolled in the study. The study officially began in December 2022 and is expected to reach its primary completion by December 2024. 59
New positive data from the TREK-DX study showed that 60% of patients treated with 400 mg eblasakimab weekly achieved EASI 90 and 66.7% reached a validated Investigator’s Global Assessment score of 0 or 1 (clear or almost clear skin) compared with only 14.3% of patients who had placebo after 16 weeks. 60 These results indicate the efficacy of eblasakimab even in patients who do not respond adequately to dupilumab. New data also showed that eblasakimab led to a rapid improvement, with over half of the patients achieving EASI 75 by week 6 and 73% by week 16. Pruritus scores showed a rapid reduction in itch, which was evident as early as week 2. Discontinuation rates were notably lower in eblasakimab-treated patients (13%) than in those who had placebo (43%).
Discussion
AD is a chronic disease with considerable implications for patients’ quality of life. Therefore, there is a continuous need for the development of new, more effective, and targeted therapies for AD. Opting for selective IL-13 inhibitors in AD treatment appears to offer advantages over broadly targeting IL-4 and IL-13. By specifically inhibiting IL-13, these medications can potentially avoid interfering with the functions of IL-4, which plays a role in a range of immune and non-immune cells. This specificity allows for a more refined approach to modulating the immune response, potentially reducing the risk of AEs associated with broader cytokine inhibition.
Among the selective IL-13 inhibitors, tralokinumab, lebrikizumab, and cendakimab show distinct mechanisms of action. Tralokinumab, which is a fully human immunoglobulin 4 monoclonal antibody, selectively binds to IL-13, neutralizing its signaling activity by preventing its binding to IL-13Rα1 and IL-13Rα2. In contrast, lebrikizumab, which is a high-affinity monoclonal antibody, targets IL-13 by binding to a different epitope. This binding prevents IL-4Rα/IL-13Rα1 heterodimerization, thus blocking downstream signaling pathways. However, unlike tralokinumab, lebrikizumab does not inhibit the binding of IL-13 to IL-13Rα2, allowing for endogenous regulation of IL-13 to continue. Similarly, cendakimab is a selective, high-affinity monoclonal antibody, which blocks IL-13 binding to both IL-13 receptors (IL-13Rα1 and IL-13Rα2) with potent efficacy, thereby inhibiting downstream signaling pathways involved in the pathogenesis of AD.7,39,50
A study compared the
Eblasakimab and dupilumab block IL-13 and IL-4 pathways, but they have distinct mechanisms of action. Eblasakimab selectively binds to the IL-13Rα1 subunit of the type 2 receptor, preventing the formation of the IL-13Rα1/IL-4Rα heterodimer receptor signaling complex. This approach exclusively blocks IL-4 and IL-13 signaling via the type 2 receptor, while sparing the type 1 receptor, potentially reducing unwanted effects associated with broad cytokine inhibition. In contrast, dupilumab, which is a fully human immunoglobulin 4κ monoclonal antibody, targets the α subunit in the IL‐4 receptor, blocking IL‐4 and IL-13 signaling through the type 2 receptor (IL-4Rα/IL-13Rα1), and also inhibiting IL-4 signaling via the type 1 receptor (IL-4Rα/common γ chain). This broader inhibition may lead to more widespread effects on the immune response. Eblasakimab’s selective targeting of the type 2 receptor may offer advantages in modulating Th2 cytokines without inducing an increase in Th1 cytokines compared with the broader inhibition achieved by blocking the IL-4 receptor entirely with dupilumab.7,16,54,55
Conjunctivitis is an important AE associated with the use of dupilumab, reported in up to 25% of patients in clinical trials and real-world studies. The exact mechanism behind this occurrence is not fully understood. However, dupilumab, by targeting IL-4, may induce a Th1 response, leading to interferon γ-mediated goblet cell apoptosis and reduced mucin production, resulting in eye dryness and conjunctivitis. 61
The prevalence of conjunctivitis with IL-13 selective inhibitors is lower than that with Dupilumab, which affects the IL-4 and IL-13 signaling pathways. In clinical trials of tralokinumab, the prevalence of conjunctivitis was 11.1% in ECZTRA 3 and less than 10% in all other studies.25–33 Lebrikizumab showed a prevalence of conjunctivitis as an AE of only up to 9.76%.40–48 Additionally, up to 12.73% of patients treated with cendakimab experienced conjunctivitis. 51 In all three selective IL-13 inhibitors, the prevalence of conjunctivitis as an AE was slightly higher in the treatment groups than in the placebo groups. 41
Direct comparisons between different IL-13 inhibitors or between IL-13 inhibitors and other therapies, such as dupilumab and Janus kinase inhibitors, are challenging because no head-to-head studies have been conducted, and individual studies have different designs. This gap in knowledge needs to be addressed in future trials and network meta-analyses to establish the place-in-therapy of selective IL-13 inhibitors in managing AD.
Conclusion
AD represents a major health issue with profound implications for patients’ quality of life. The pivotal role of IL-13 in the pathogenesis of AD has been indicated by the clinical success of therapies targeting this cytokine.
Tralokinumab and lebrikizumab have emerged as effective and safe treatment options for individuals with moderate-to-severe AD, and show notable efficacy and safety in clinical settings. Two other IL-13 inhibitors, eblasakimab and cendakimab, each with slightly different mechanisms of action, are currently under study. Further investigations are warranted to determine their therapeutic efficacy. Because of the scarcity of sustainable therapeutic options for severe and refractory AD, the outcomes achieved through selective IL-13 inhibition shed light on the path forward in managing this disease.
Footnotes
Author contributions
All of the authors contributed equally to the preparation of this review. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Data availability
Data availability is not applicable to this article because no new data were created or analyzed in this study. This is a narrative review, and all sources of information are cited appropriately in the manuscript.
Declaration of conflicting interest
Orhan Yilmaz, Carlos Teixeira, and Diana Bernardo declare that there is no conflict of interest. Tiago Torres has received consultancy and/or speaker’s honoraria from and/or participated in clinical trials sponsored by AbbVie, Amgen, Almirall, Amgen, Arena Pharmaceuticals, Biocad, Biogen, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Fresenius-Kabi, Janssen, LEO Pharma, Eli Lilly, MSD, Mylan, Novartis, Pfizer, Samsung-Bioepis, Sanofi-Genzyme, Sandoz, and UCB.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
