Abstract
The traditional treatment for small-cell lung cancer (SCLC) has been traditional systemic platinum-containing chemotherapy because the response rate is 50–90%. Durvalumab is an immune checkpoint inhibitor that blocks the binding of programmed cell death protein 1 and programmed cell death 1 ligand 1. Durvalumab combined with traditional chemotherapy agents has been recommended as the first-line treatment for extensive-stage SCLC, but its use may cause immune-related adverse events. Autoimmune encephalitis is a rare and potentially fatal neurological adverse event. This current case report describes a male patient in his late 50s with ES-SCLC who developed autoimmune encephalitis associated with durvalumab treatment after three cycles of combination chemotherapy. This current case furthers the understanding of autoimmune encephalitis caused by durvalumab treatment.
Introduction
Globally, almost 20 million new cancer cases were diagnosed and almost 10 million cancer-related deaths occurred in 2020, with lung cancer being the leading cause of cancer-related deaths. 1 Small-cell lung cancer (SCLC), which is characterized by a severe clinical course, accounts for 14% of lung cancer cases. 2 Many patients with SCLC have extensive-stage disease at diagnosis and their prognosis remains poor despite cytotoxic chemotherapy. 3 SCLC is sensitive to platinum-based chemotherapy, with a response rate of 50–90%. 4 However, lung cancer is prone to recurrence and rapid progression. Immunotherapies targeting immune checkpoint inhibitors (ICIs) have led to a major paradigm shift in cancer treatment, improving overall survival rates for a wide range of cancers.5,6
In recent years, with the expansion of immunotherapy research, ICIs combined with chemotherapy have gradually become a first-line treatment for SCLC. 7 The US Food and Drug Administration approved atezolizumab and durvalumab in combination with chemotherapy in March 2019 and March 2020, respectively, for the first-line treatment of patients with extensive SCLC. 4 ICIs are a class of monoclonal antibodies that inhibit the activation of T cells by inhibiting immune checkpoints and the class is mainly composed of monoclonal antibodies against programmed cell death protein 1 (PD-1), programmed cell death 1 ligand 1 (PD-L1) and cytotoxic T lymphocyte antigen-4. 8 The CASPIAN trial assessed durvalumab in combination with etoposide with either cisplatin or carboplatin as a first-line therapy for patients with extensive-stage (ES)-SCLC. 9 Survival benefits from the use of PD-1 and PD-L1 inhibitors have been demonstrated by a 5-year survival rate for some patients with nonsmall-cell lung cancer that exceeds 30%. 5 The prognosis of ES-SCLC was also significantly improved. 9 However, despite the improved outcomes for cancer treatment, ICI treatment has also been associated with a large number of toxicities, described as immune-related adverse events (irAEs).6,10 Among them, neurotoxicity involving the nervous system belongs to a rare category of irAEs called neurological immune-related advert events. Autoimmune encephalitis due to treatment with ICIs is a type of neurological adverse events, 11 which also includes the occurrence of anti-γ-GABAB receptor (GABABR) antibody-associated encephalitis. The probability that patients will develop autoimmune encephalitis when treated with ICIs is low. 12 This current case report describes a patient with ES-SCLC who developed autoimmune encephalitis associated with durvalumab treatment.
Case report
In July 2021, a Chinese man in his late 50s was admitted to the Department of Endocrinology, The First affiliated Hospital of Soochow University, Suzhou, Jiangsu Province, China due to ‘intractable hyponatraemia’. He reported being previously healthy. After admission, computed tomography (CT) examination of the chest, abdomen and pelvis showed that the lymph nodes were enlarged at the root of the neck, pulmonary hilum and mediastinum and there was emphysema of both lungs with pulmonary bullosa; and there were no obvious abnormalities found in the abdominal pelvis (Figures 1(a)–1(f)).

Computed tomography images of a previously healthy male patient in his late 50s who was admitted due to ‘intractable hyponatraemia’: (a) lesion on the right side of the pulmonary window; (b) lesions on the right side of mediastinal window; (c & d) intrapulmonary metastasis; (e) lymph node enlargement in the contralateral mediastinum and (f) puncture to obtain the pathological sample (arrow).
A biopsy was taken and the pathology showed metastatic SCLC (Figure 2). Immunohistochemistry was undertaken and it showed the following for the tumour cells: cytokeratin (+), thyroid transcription factor 1 (+), cluster of differentiation (CD) 56 (+), chromogranin A (+), synapsin (+), antigen identified by monoclonal antibody Ki-67 (+, 90%), terminal deoxynucleotide transferase (–), epithelial membrane antigen (+), CD20 (–), CD79a (–), CD2 (–) and CD3 (–). Head magnetic resonance imaging (MRI) showed multiple ischaemic lesions in the bilateral subfrontal cortex and bilateral ethmoid sinus inflammation. No lesions were found in the pituitary. Based on the biopsy results and other auxiliary tests, the diagnosis was made as follows: ES-SCLC (stage IIIc [T3N3M0]), hyponatraemia and abnormal antidiuretic hormone secretion syndrome. PD-L1 expression was not evaluated in the study.

Posterior right neck lymph node biopsy taken from a previously healthy male patient in his late 50s who was admitted due to ‘intractable hyponatraemia. The tissue section was stained with haematoxylin and eosin. Scale bar 50 µm. The colour version of this figure is available at: http://imr.sagepub.com.
After active sodium supplementation and tolvaptan tablets (antidiuretic hormone receptor antagonist) treatment, the patient was treated with 100 mg etoposide intravenous (i.v.) once a day for 5 days and 65 mg cis-platinum i.v. once a day for 2 days (EC) plus 1000 mg durvalumab i.v. once a day for 1 day as the first-line therapy for ES-SCLC once every 21 days. There were no obvious adverse reactions during the two rounds of chemotherapy combined with immunotherapy. Chest CT imaging showed a partial response of lung cancer and immunotherapy-related adverse reactions were monitored (Figure 3). Pituitary function, thyroid function, electrocardiograph and brain natriuretic peptide levels were all normal. The patient was discharged from hospital successfully with no obvious adverse reactions after the third round of chemotherapy combined with immunotherapy, but 7 days later he was hospitalized as a result of a convulsion. The patient presented with left upper limb convulsions, difficulty in extending the tongue and an inability to speak. He experienced seizures and electroencephalography (EEG) showed slow wave emissions. No positive neurological symptoms were found.

Computed tomography images of a previously healthy male patient in his late 50s who was admitted due to ‘intractable hyponatraemia’: (a) before and (b) after two cycles of etoposide + cis-platinum + durvalumab showing that the lymph node enlargement at the root of the neck, hilar and mediastinum was significantly reduced; and there was a partial response in the lung cancer.
An urgent CT scan of the head showed no obvious abnormalities and chest CT was similar to that examined previously. After admission, there were no abnormalities in his routine blood, biochemistry and tumour markers. He was treated with 400 mg sodium valproate i.v. with normal saline (50 ml at 8 ml/h) for the first day. Then, he received 500 mg sodium valproate oral twice a day for 16 days combined with levetiracetam tablets orally to control seizures as follows: 250 mg levetiracetam oral twice a day for 7 days followed by 750 mg levetiracetam oral twice a day for 10 days. On the day of admission, an EEG (Figure 4(a)) showed no abnormal wave or slow wave emissions. MRI scans showed bilateral subfrontal cortex, hemioval centre and lateral ventricular ischaemia (Figures 4(c) and 4(d)). MRI showed no brain metastases. Cerebrospinal fluid (CSF) was collected via a lumbar puncture 2 days later and showed the following: colourless and transparent; pressure, 190 mmHg; white blood cell count, 54 × 106/l; monocytes, 94.5%; chlorine, 119.6 mmol/l; and protein, 0.6 g/l. No bacterial and cryptococcus growth was detected. An examination of the CSF showed no malignant cells. Tuberculosculoid-related antibodies were negative. The following antibodies related to paraneoplastic tumour syndrome were tested and found to be negative: anti-HU antibody, Ri antibody, anti-YO antibody, anti-CV2 antibody, anti-amphiphysin antibody and anti-paraneoplastic Ma antigen antibody. Another lumbar puncture was performed 4 days later and the results were as follows: CSF pressure was normal; white blood cells, 63 × 106/l; monocyte percentage, 100%; glucose, 4.6 mmol/l; protein, 0.6 g/l; and chlorine, 119.1 mmol/l. Immunoglobulin G antibody against GABABR was moderately positive by autoimmune encephalitis spectroscopy. Serum analysis results were as follows: anti-NMDA receptor (NMDA) antibody (–), anti-α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid l (AMPA1) receptor antibody (–), anti-α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid 2 (AMPA2) receptor antibody (–), anti-leucine-rich glioma-inactivated (LGI 1) antibody (–), anti-GABABR antibody (++) 1:32, anti-dipeptidyl-peptidase-like protein-6 (DPPX) receptor antibody (–), anti-immunoglobulin-like cell adhesion molecule 5 (IgLON5) receptor antibody (–), anti-glutamate decarboxylase 65 (GAD65) receptor antibody (–), anti-metabotropic glutamate receptor 5 (mGluR5) receptor antibody (–) and anti-myelin oligodendrocyte glycoprotein (MOG) receptor antibody (–). CSF analysis results were as follows: NMDA (–), AMPA1 (–), AMPA2 (–), LGI 1 (–), GABABR (++) 1:10, DPPX (–), IgLON5 (–), GAD65 (–), mGluR5 (–) and MOG (–). Considering the above test results, the patient was diagnosed with anti-GABABR antibody-associated autoimmune encephalitis. 13 After stopping durvalumab and treating the patient with 60 mg methylprednisolone i.v. twice a day for 2 days (followed by methylprednisolone 40 mg i.v. twice a day for 5 days) plus 5 g gamma globulin i.v. once a day for 5 days, his symptoms improved (Figures 5 and 6). Seizures occurred on the 2nd and 5th days of i.v. injections of methylprednisolone and gamma globulin, so 100 mg phenobarbital intramural injection twice a day was added on the 5th day. After 16 days of admission, the patient was delirious, did not answer questions and could not correctly cooperate with instructions.
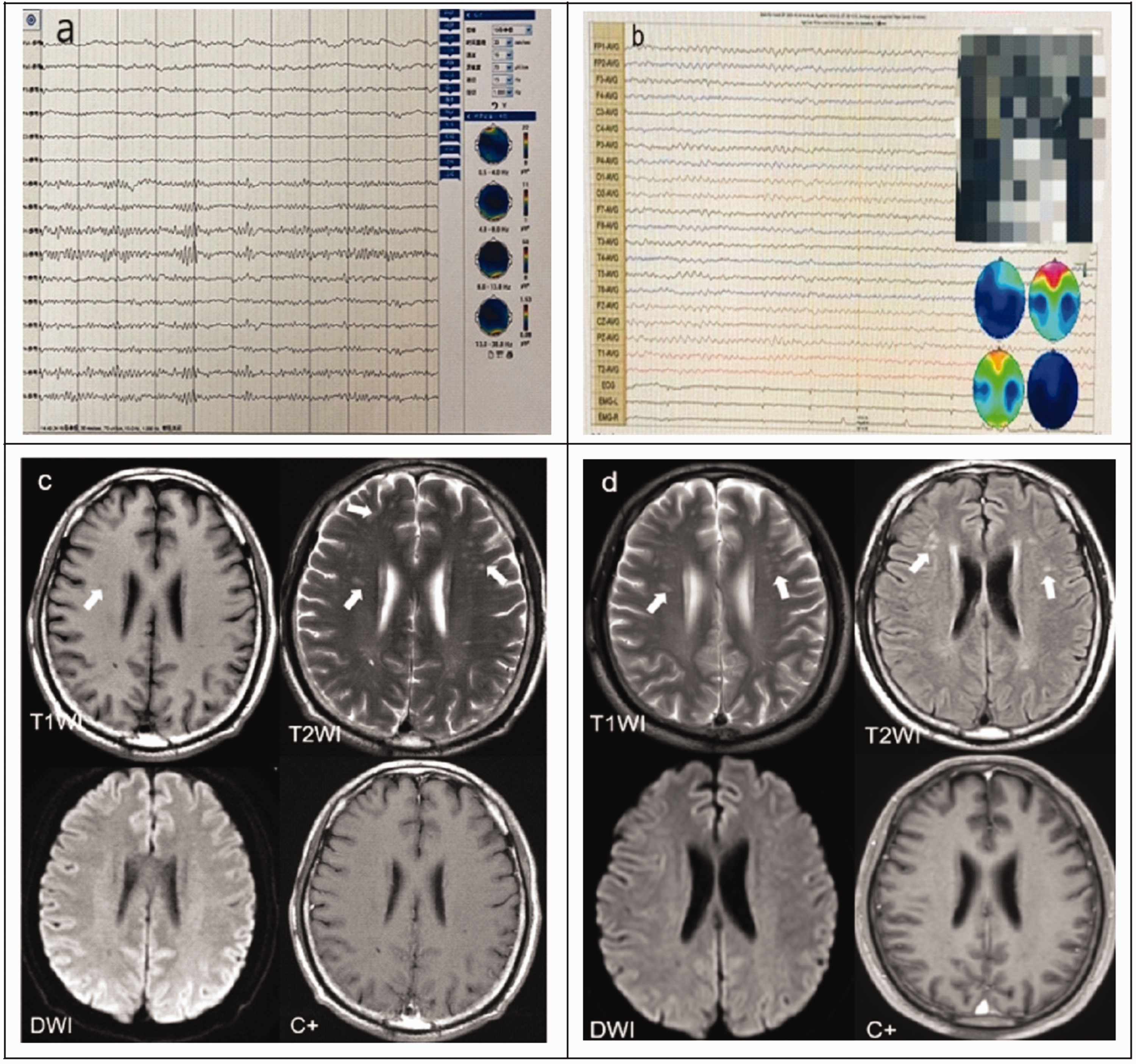
Electroencephalograms (EEG) and computed tomography (CT) images of a previously healthy male patient in his late 50s who was admitted due to ‘intractable hyponatraemia after he had experienced seizures: (a) 23 September 2021, no obvious abnormality on EEG; (b) 14 October 2021, abnormal EEG findings, moderate background abnormalities and small epileptoid discharges on the right anterior head; (c) 24 September 2021, bilateral frontal subcortical white matter and centrum semiovale area show multiple abnormal signals on CT images, which appear isointense or slightly hypointense in T1 weighted images, slightly hyperintense in T2 weighted images and isointense in diffusion-weighted images. No significant enhancement was observed in the enhanced images and (d) 17 January 2022, multiple abnormal CT signal shadows can be seen in the bilateral frontal subcortical white matter and the centrum semiovale area, which appear as slightly high signals in T2 weighted images and T2 FLAIR images and isointense in diffusion-weighted images. No significant enhancement was observed in the enhanced images. Compared with the magnetic resonance imaging undertaken on 24 September 2021, the lesions were roughly similar. The colour version of this figure is available at: http://imr.sagepub.com.

Schematic diagram of the medication regimens used during the entire course of treatment of a previously healthy male patient in his late 50 s who was admitted due to ‘intractable hyponatraemia’. The colour version of this figure is available at: http://imr.sagepub.com. NEURO, Department of Neurology; IVIG, intravenous immunoglobulin; iv, intravenous; po, per oral; bid, twice a day; st, statim/immediately; qd, once a day; qn, quaque nocte.

Schematic diagram of the events that occurred between diagnosis of small-cell lung cancer (SCLC) and the development of encephalitis in a previously healthy male patient in his late 50 s who was admitted due to ‘intractable hyponatraemia. The colour version of this figure is available at: http://imr.sagepub.com. EC, etoposide plus cisplatin; EEG, electroencephalography; CSF, cerebrospinal fluid; MRI, magnetic resonance imaging; GABAR, anti-γ-GABAB receptor; NEURO, Department of Neurology.
After discussion, the patient was administered 500 mg methylprednisolone i.v. four times a day for 3 days, plus antiviral therapy as follows: 3 g foscarnet sodium injection i.v. twice a day for 5 days. Three days later, the dose of methylprednisolone was decreased to 80 mg four times a day. The patient was transferred to the Department of Neurology, The First affiliated Hospital of Soochow University, Suzhou, Jiangsu Province, China for continued treatment after another seizure 4 days later. The patient was given 250 mg methylprednisolone i.v. once a day for 3 days, 120 mg methylprednisolone i.v. once a day for 3 days, 80 mg methylprednisolone i.v. once a day for 3 days, 40 mg methylprednisolone i.v. once a day for 3 days, followed by 30 mg prednisone oral once a day for 3 days; and enhanced anti-seizure drugs as follows (Figure 5): before day 83, 400 mg valproate i.v. once a day for 1 day followed by 500 mg sodium valproate oral twice a day for 37 days; 250 mg levetiracetam oral twice a day for 7 days followed by 750 mg levetiracetam oral twice a day for 10 days; and 100 mg phenobarbitone intramuscular (i.m.) twice a day for 1 day on day 75. After day 83, the patient received 500 mg sodium valproate oral twice a day, 1000 mg levetiracetam oral twice a day and 100 mg phenobarbitone i.m. once a day; and he also received 10 mg diazepam i.v. once a day for 3 days (Figure 5).
Two days after the patient was transferred to the Department of Neurology, a 24-h EEG showed abnormal findings, moderate background abnormalities and small epileptoid discharges on the right anterior head (Figure 4(b)). During this period, there were no seizures and his mental health status was normal. Eight days after he was transferred to the Department of Neurology, the patient developed emotional irritability at night, repeated abnormal and aggressive behaviours. He was administered symptomatic treatment of olanzapine and quetiapine as follows: 5 mg olanzapine oral twice a day for 19 days; and 50 mg quetiapine once a day for 4 days followed by 100 mg quetiapine once a day for 15 days. Sixteen days later, the patient was transferred to a psychiatric hospital for treatment of psychiatric symptoms, during which monoclonal antibodies and chemotherapy drugs were discontinued. Antipsychotic drugs and antiepileptic drugs were maintained 1 month. Two months later, a follow-up CT showed several slightly enlarged lymph nodes in the mediastinum and no obvious enlarged lymph nodes in the two pulmonary hilus. Calcification was observed in the aortic arch and coronary artery, but no pleural effusion was observed. Two months later, the EC chemotherapy regimen was repeated and supplemented with radiotherapy. During this period, chest CT was undertaken repeatedly and reviewed for the progression of lung cancer (Figure 7). After 120 mg docetaxel i.v. once a day for 1 day was used for treatment, lung cancer still progressed and showed distant metastasis. The patient’s personal information has been de-identified. The patient subsequently died so consent could not be obtained.

Computed tomography imaging at various stages of treatment of a previously healthy male patient in his late 50s who was admitted due to ‘intractable hyponatraemia’: (a) 24 March 2022, after treatment with the etoposide + cis-platinum chemotherapy regimen; (b) 15 August 2022, after multiple chemoradiotherapy and (c) February 2023, after the chemotherapy regimen was changed to docetaxel, the images showed that the mediastinum lymph nodes were significantly enlarged compared with those before treatment with immunotherapy.
Discussion
The current patient was diagnosed with ES-SCLC (stage IIIc [T3N3M0]). After three cycles of treatment with the EC chemotherapy regimen combined with durvalumab, he developed convulsions in the left upper limb, difficulty in extending the tongue and speech failure. The incidence of irAEs in the nervous system after ICI treatment is 2–6%,14,15 of which the incidence of immune-related encephalitis is 0.05–0.2%.16–18 Immune-associated encephalitis caused by durvalumab is rarely reported. The time of onset varies widely and patients should be monitored for more than 1 year after the last administration of ICIs. 19
Immune-related encephalitis associated with ICI is a diagnosis of exclusion and can be challenging. MRI is not very sensitive for markers of ICI-induced encephalitis (ICI-iE) because MRI findings are normal in nearly half the patients at presentation while CSF samples from patients with ICI-iE are almost universally abnormal. 20 In this current case, the patient’s encephalitis was not evident on MRI. At the same time, the CSF was positive for anti-GABABR antibodies, so the diagnosis was ICI-iE. A previous study reported one of the largest case series of patients with immune-related encephalitis, of which it suggested that lung cancer patients had the highest rate of encephalitis at 32.6%, followed by melanoma at 31.8%. 21 Most patients with anti‑GABABR antibodies develop limbic encephalitis with prominent epilepsy, which is usually refractory to multiple antiepileptic agents but responds to immunotherapy. 22 Most of the guidelines recommend corticosteroids combined with intravenous propyl/plasma exchange; followed by second-line immunotherapy (e.g. rituximab, cyclophosphamide.) if nonresponsive.13,23
Although the detection rate of antibodies is not very high, it is still an important diagnostic index for the diagnosis of immune-associated encephalitis. It has been speculated that the ICI-iE response might cross-react with an auto-antigen within the central nervous system, 24 leading to the occurrence of an autoimmune reaction in the central nervous system. Therefore, it is very important to identify the type of encephalitis quickly and accurately after the occurrence of nervous system-related symptoms.
In conclusion, this current case furthers the understanding of autoimmune encephalitis caused by durvalumab treatment. Lessons that were learned during the treatment of this current case included the following: (i) the corticosteroid reduction was too rapid, which resulted in multiple seizures; (ii) after treatment with 500 mg methylprednisolone four times a day, the patient had no further seizures again, but after continued corticosteroid reduction, the patient showed mental health abnormalities. These were similar to those seen during corticosteroid reduction in a retrospective study. 21
Footnotes
Author contributions
Yu-Lan Qian and Yin-Hua Gong: drafted the manuscript; Jian-Kang Yu: undertook a grammar check; Chao Liu and Wen-Ting Wu: data analysis; Ye Jiang: graphics and literature analysis; Dan Shen: case management during hospitalization and revision of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interest
All authors declare no conflicts of interest.
Funding
This research received grants from the Jiangsu Provincial Medical Key Discipline (no. ZDXK202201), Jiangsu Provincial Medical Key Discipline (no. ZDXK202247) and Jiangsu Pharmaceutical Association-Hospital Pharmacy Research Project (no. H202312).
