Abstract
Multiple primary lung cancer is a very rare type of tumour that occurs when two or more primary malignant tumours develop simultaneously or sequentially in the same patient’s lungs. When two or more primary sites are present at the same time, it is called synchronous multiple primary lung cancer. Currently, the pathogenesis, clinical features, and prognostic factors of synchronous multiple primary lung cancer are unclear, and there are no guidelines for diagnosis and treatment. We present a 74-year-old male patient with a combination of three histological types of synchronous primary lung cancer. He underwent partial lobectomy and was diagnosed with synchronous multiple primary lung cancer. The pathological diagnosis revealed adenosquamous carcinoma in the right lower lobe and small-cell lung cancer in the left lower lobe. After comprehensive treatment, the patient is currently stable and is under follow-up. Refining the diagnosis and treatment of multiple primary lung cancers remains challenging. Our case and case studies in the literature may provide some ideas for more standardized diagnosis and treatment.
Introduction
Multiple primary lung cancer (MPLC) is defined as the simultaneous or sequential occurrence of two or more primary malignant tumours in the lung of the same patient. Adenocarcinoma (80%–99%) is the most common type, followed by squamous cell carcinoma (SCC; 13%). 1 The incidence of MPLC ranges from ~0.2% to 8% in different clinical series. 2 MPLC can be classified into synchronous MPLC (sMPLC) and metachronous MPLC (mMPLC), depending on whether the primary lesions occur at intervals or not. 3 sMPLC is much rarer than mMPLC, with an incidence rate of only 0.26%–1.33%. 4 Martini and Melamed 5 and Antakli et al 6 first proposed the relevant diagnostic criteria in 1975. sMPLC must meet one of the following three conditions: (1) lung cancers in different locations, isolated from each other; (2) different histological types; and (3) when histological types are the same, located in different lung segments, lobes, or sides of the lung, originating from different carcinomas in situ, and there is no tumour in the common lymphatic drainage sites of the lung cancers, with no extrapulmonary metastases detected at the time of diagnosis. As the understanding of the disease has improved, the American College of Chest Physicians (ACCP) has added to this diagnosis on several occasions, 7 emphasizing that sMPLC requires a multidisciplinary and comprehensive diagnosis and that the histological type, genetic features, imaging features, and clinical presentation must be taken into account in the diagnosis.
Case presentation
A 74-year-old man with cough and sputum for more than 1 month presented to our hospital with a history of Behcet disease for more than 30 years and smoking for more than 40 years. The patient reported that in January 2024, he had coughing sputum and white mucous sputum without any obvious trigger, accompanied by generalized fatigue but without chest tightness and shortness of breath. He came to our hospital on February 16. The chest computed tomography (CT) images show a nodule with a cavity in the right lower lung, a solid nodule in the left lower lung, with enlarged mediastinal lymph nodes (Figure 1(a1)–(a3)). Further positron emission tomography (PET)–CT imaging revealed increased glucose metabolism in the aforementioned nodules and mediastinal lymph nodes (Figure 1(b1)–(b3)). After the multidisciplinary consultation, we all agreed that we need to pay attention to both diagnosis and treatment. We performed surgical intervention, excising the aforementioned lesions and clearing mediastinal lymph nodes. Based on postoperative pathology, the diagnosis was adenosquamous carcinoma (ASC) (60% SCC component + 40% adenocarcinoma component) in the right lower lobe (Figure 2), accompanied by right mediastinal lymph node metastasis, and small-cell lung cancer (SCLC) in the left lower lobe (Figure 3), accompanied by left mediastinal lymph node metastasis. The next-generation sequencing (NGS) shows the following: ASC cells: TP53, five exon, p.A 159V, 2.82%; programmed death-ligand 1 (PD-L1) = 1%; SCLC cells: TP53, eight exon, p.E287, 58.58%; and PD-L1 < 1%. The patient was diagnosed with sMPLC adenosquamous carcinoma (ASC; T1bN2M0) combined with SCLC (T1bN2M0).

Images of the patient’s chest CT scan: a nodule with cavitation in the right lower lobe (a1); a solid nodule in the left lower lobe (a2); enlarged lymph nodes and widened mediastinum (a3). Images of the patient’s PET–CT scan: nodules in the right lower lobe (b1); nodules in the left lobe (b2); enlarged lymph nodes and widened mediastinum (b3), all show increased glucose metabolism.

Right lower lung adenosquamous carcinoma (a) HE staining (200×). (b) CK5/6 staining (100×). P40 (+) (c) CK6/7 staining (100×). TTF-1 (+). (d) P40 staining (100×). NapsinA (+).

Small-cell lung cancer of the left lower lung (a) HE staining (200×). (b) Syn staining (200×). (c) CgA staining (200×). Tissue immunohistochemistry: Syn (+), CgA (+).
Given the patient’s concurrent mediastinal lymph node metastasis, we decided to add adjuvant therapy post-surgery. However, due to inconsistent NGS mutation sites and PD-L1 expression, we did not consider targeted therapy or immune checkpoint inhibitors. For the chemotherapy regimen, docetaxel was chosen as a broad-spectrum antitumour agent effective in both non-SCLC and SCLC. We ultimately selected carboplatin (0.45 g) plus docetaxel (120 mg). Both the lung ART and the PORT-C study8,9 indicate that although postoperative radiotherapy does not improve disease-free survival or overall survival, it can reduce the mediastinal recurrence rate. Therefore, after completing four cycles of chemotherapy, we implemented mediastinal radiotherapy for the patient based on the consideration of preventing recurrence, following joint decision-making between the doctors and the patient. Prophylactic irradiation of the mediastinum is 50 Gy/25 times. At present, the patient is under follow-up.
Discussion
This is a rare case of sMPLC with three different histological subtypes, with differential expression of p53 and PD-L1 within the tumour. To gain a deeper understanding of the characteristics of this disease at the same time, we counted previously reported sMPLC combining three different histological subtypes by searching PubMed and Web of Science, and a total of eight references10 –17 were retrieved, involving eight patients. Relevant information (including the present case) is summarized as follows (see Table 1). In comparison, males, smokers, and those with chronic respiratory diseases were more likely to develop the disease, and the combination of the three subtypes of adenocarcinoma, SCC, and SCLC was more common. Most cases were diagnosed by postoperative pathology; there were no lymph node metastases at the time of diagnosis, and all underwent surgical treatment, with half of the patients having stable disease after treatment. Additionally, the survival curves were constructed based on follow-up data from eight of these patients (Figure 4).
Ten cases of concurrent sMPLC of three different histological types.
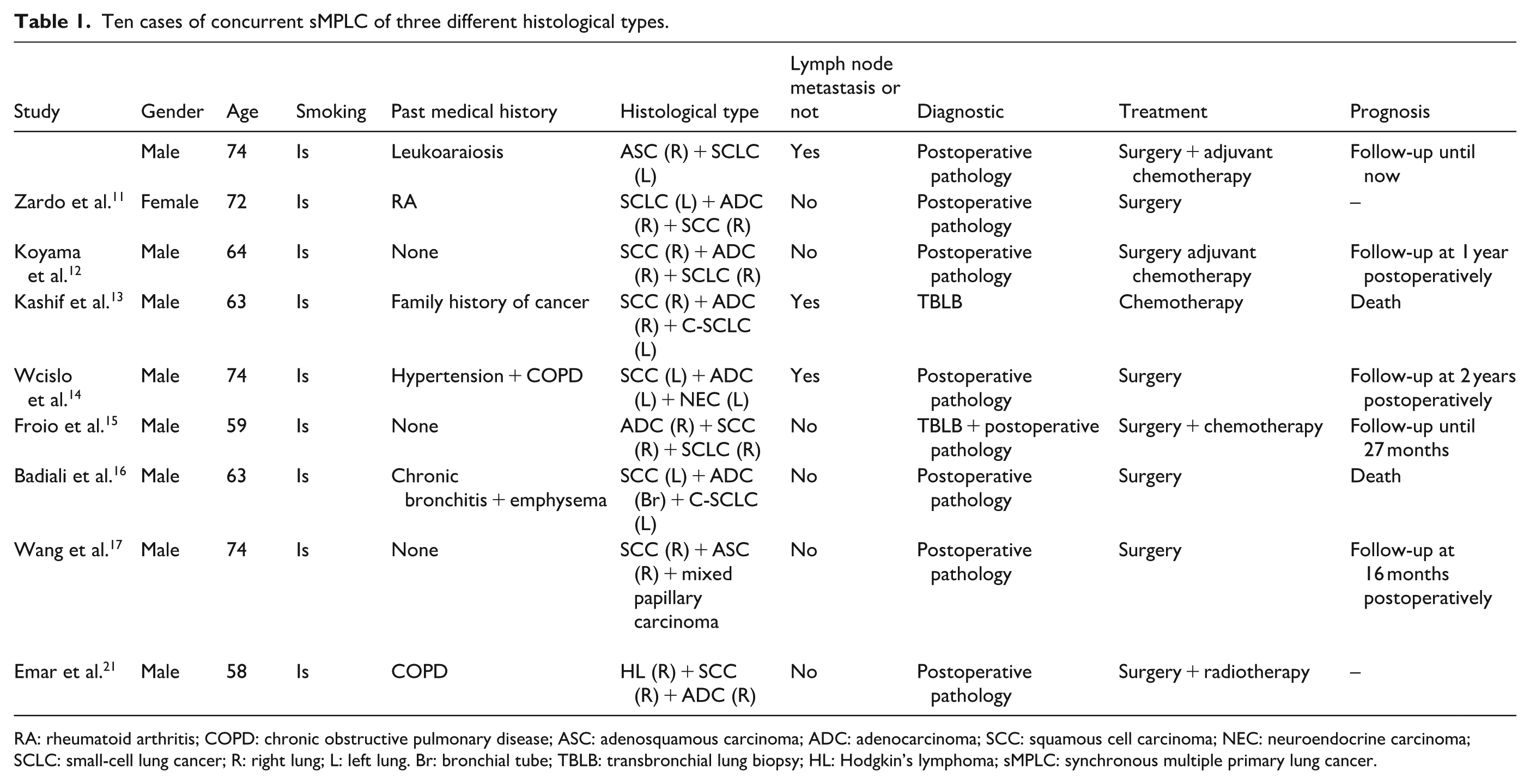
RA: rheumatoid arthritis; COPD: chronic obstructive pulmonary disease; ASC: adenosquamous carcinoma; ADC: adenocarcinoma; SCC: squamous cell carcinoma; NEC: neuroendocrine carcinoma; SCLC: small-cell lung cancer; R: right lung; L: left lung. Br: bronchial tube; TBLB: transbronchial lung biopsy; HL: Hodgkin’s lymphoma; sMPLC: synchronous multiple primary lung cancer.

Survival prognosis curves for eight patients.
The specific molecular mechanisms underlying sMPLC remain unclear. Most current studies suggest that the molecular characteristics of multiprime lung cancer are similar to those of single-prime lung cancer. For example, they have similar driver genes that are enriched in somatic mutations, including EGFR, KRAS, and TP53. Some studies also suggest that multiple abnormally expressed immune response-related factors may be present in MPLC, such as the inflammation and immune response regulator TNFRSF18.18,19 However, due to the limited research conducted on MPLC to date, further exploration and validation of the specific underlying molecular mechanisms are required. 20
For sMPLC, pathological diagnosis is the gold standard, and a comprehensive evaluation in conjunction with chest CT or PET is required. In addition, as multiple cancer nodules in the lung, sMPLC must be differentiated from intrapulmonary metastases, and sMPLC usually presents earlier and has a better prognosis. 21 sMPLC of the same histological subtype is more difficult to differentiate and often requires molecular biological identification, whereas sMPLC of three different histological subtypes is easier to diagnose and has a higher 5-year survival rate. 22 sMPLC, including the SCLC subtype, must be distinguished from mixed SCLC, which usually occurs in a different lobe or part of the lung, whereas the latter is more likely to occur in the same lobe and is often associated. For disease staging, the TNM staging system does not accurately reflect the survival and prognosis of multifocal lesions. It has been suggested that multifocal lesions should be staged separately, with the higher stage representing the patient’s terminal stage, 23 which may help guide treatment. However, there are no authoritative clinical treatment guidelines. The expert Consensus of the Chinese Anti-Cancer Association 18 emphasizes that multidisciplinary team treatment should be prioritized for most primary lung cancers. Specifically, this involves developing personalized, comprehensive treatment strategies tailored to the specific clinical characteristics of each patient. These strategies must take individual circumstances into account when selecting surgical or non-surgical interventions, and be supplemented by corresponding clinical follow-up protocols. Surgical resection remains the mainstay of treatment for sMPLC. ACCP 24 recommends surgery as the first-line treatment for patients with MPLC. Survival rates for patients with sMPLC who undergo surgery are significantly better than those who do not. Early detection and surgical resection significantly improve the prognosis of patients. 25 The decision to operate and the surgical approach should be based on a combination of factors such as the number and location of lesions, the presence of distant metastases, and patient tolerance. For patients who are unsuitable for surgery or who refuse it, as well as for those who have residual lesions after surgery, non-surgical treatments, including radiotherapy, chemotherapy, targeted therapy, and immune checkpoint inhibitors, may be selected. However, MPLC often exhibits low tumour mutational burden and PD-L1 positivity rates, along with significant inter-lesion heterogeneity in the immune microenvironment, and the efficacy of targeted therapy and immune checkpoint inhibitors could not be assessed due to the different tissue sources and often driver gene heterogeneity. Nevertheless, combining targeted therapy with surgery could be a promising approach for managing MPLC with sensitive driver mutations such as EGFR in MPLC.
Postoperative follow-up relies primarily on imaging examinations. Differentiated follow-up strategies should be developed based on the type of residual nodules present after surgery. 18 sMPLC patients have a slower progression compared to solitary tumours, and the prevalence of multiple lesions in the lung suggests a stable “coexistence” with the host organism. Relevant clinical analyses have shown that only a history of smoking, a maximum lesion size >2 cm, the presence of lymph node metastases, and pleural involvement are independent risk factors for prognostic survival in sMPLC.1,19,26 The presence of the same histological subtype was not an independent risk factor, and there was a trend towards a relatively better but not statistically different prognosis for sMPLC of different histological subtypes. 14 Our patient had all three of these independent risk factors, completed postoperative adjuvant chemotherapy and radiotherapy, and is still being followed up.
Conclusions
In conclusion, this paper reports a rare type of lung cancer, sMPLC with three different histologic types (ADC, SCC, and ASC), which deepens the understanding of sMPLC; however, sMPLC still requires the accumulation of a large amount of clinical data and corroborating evidence to determine the optimal diagnostic criteria and effective treatment strategies.
Footnotes
Acknowledgements
The authors are grateful to Dan Shen for his guidance.
Ethical considerations
We have de-identified personal information.
Consent for publication
We obtained written informed consent from the patient.
Author contributions
Wenting Wu: drafting the manuscript. Yinhua Gong: revise manuscript. Chengcheng Xu: perform the thoracic surgery. Dan Shen: case management during hospitalization and give manuscript guidance. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangsu Provincial Medical Key Discipline (no. ZDXK 202201). Jiangsu Pharmaceutical Association-Hospital Pharmacy Research Project (grant no. H202312).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Available.
