Abstract
This paper presents the case of a 45-year-old woman who developed aquaporin-4 (AQP4) antibody-positive neuromyelitis optica spectrum disorder (NMOSD) approximately 6 weeks after a herpes zoster infection. Her initial symptoms included area postrema syndrome, which was marked by persistent nausea, vomiting, and belching. This report also provides a summary of the demographic and clinical features, disease progression, magnetic resonance imaging findings, cerebrospinal fluid analysis, treatment plans, and recovery outcomes of 11 patients—including this case and 10 others reported in the literature since 2008—who developed AQP4 antibody-positive NMOSD following herpes zoster infection. This review aims to improve clinicians’ understanding of the characteristics, treatment, and prognosis of this disease. It also highlights the importance for pain management specialists to consider central pain and use the MIDNIGHTS or VINDψCATE mnemonic devices to systematically consider the differential diagnoses of a patient’s new pain symptoms.
Keywords
Introduction
Neuromyelitis optica (NMO) is an immune-mediated, primary inflammatory demyelinating disease of the central nervous system, primarily characterized by optic neuritis and longitudinally extensive transverse myelitis. 1 First described by Devic in 1894, it was originally known as Devic’s disease. 2 Recent studies have identified aquaporin-4 (AQP4) antibody as a specific immunological marker for NMO, 3 significantly advancing our understanding of the disease. In 2015, the international NMO diagnostic panel expanded the definition to include NMO spectrum disorder (NMOSD) and established new diagnostic criteria. The revised NMOSD criteria classify the condition into AQP4 antibody-positive (AQP4+) and AQP4 antibody-negative (AQP4−) subtypes. 4
The exact etiology of NMOSD remains unclear, but infectious diseases, particularly viral infections, are suspected triggers. Literature suggests that viruses such as herpes simplex virus, varicella-zoster virus (VZV), 5 mumps virus, 6 cytomegalovirus, 7 human immunodeficiency virus, 8 and severe acute respiratory syndrome coronavirus 2 9 can provoke AQP4+ NMOSD. Wang et al. 5 explored the correlation between prodromal infection and NMOSD through Mendelian randomization studies and found that herpes simplex virus and VZV may have a causal relationship with AQP4+ NMOSD, further indicating that infectious pathogenesis might play a central role in NMOSD. Herpes zoster, caused by the reactivation of latent VZV in the dorsal root ganglion (DRG) or cranial nerve ganglion, has a global incidence of 3 to 5 cases per 1000 person-years, 10 making VZV-related NMOSD a plausible concern.
Post-VZV infection can lead to VZV myelitis and may coexist with AQP4+ NMOSD, although these conditions are relatively rare, 11 and there is some debate regarding VZV myelopathy or myelitis. 12 Currently, literature on AQP4+ NMOSD following herpes zoster mainly consists of case reports, which do not provide a comprehensive understanding of its clinical characteristics and prognosis. Early diagnosis is crucial because NMOSD requires aggressive treatment. Most patients with herpes zoster initially present to a dermatology or pain clinic with neuropathic pain as their primary symptom, where clinicians may overlook other symptoms such as pain not corresponding to the DRG sites affected by VZV, nausea, vomiting, sensory disorders, and ataxia.
We herein report a case in which a patient developed left upper limb pain that was misdiagnosed as new-onset shingles in the left arm following herpes zoster in the right upper limb, ultimately leading to an AQP4+ NMOSD flare-up. This case, along with a summary of similar cases, aims to improve clinicians’ understanding of the characteristics, treatment, and prognosis of NMOSD.
Case report
On 28 August 2023, a 45-year-old woman presented with herpes zoster on her right upper limb, experiencing stabbing and burning pain. She reported having taken antiviral medication and traditional Chinese medicine at a local clinic, but the specifics of the treatment were unknown. One week after starting the medication, she developed nausea and began vomiting saliva-like substances. Gastroscopy at a local hospital indicated chronic atrophic gastritis, but her condition did not significantly improve. On 10 September 2023, she was admitted to our hospital’s gastroenterology department because of a 1-week history of nausea and vomiting. Physical examination revealed no significant abnormalities, but she was found to have notable hypokalemia (serum potassium: 2.94 mmol/L). She was treated with omeprazole enteric-coated capsules, hydrotalcite chewable tablets for gastroprotection, mosapride and domperidone to enhance gastric motility, pregabalin for neuropathic pain, and potassium supplements for symptomatic support. Brain magnetic resonance imaging (MRI) conducted on 13 September 2023 showed no significant abnormalities (Figure 1).

Magnetic resonance imaging of the brain.
On 14 September 2023, the patient was transferred to the pain management department because of significant right upper limb pain. Upon transfer, her visual analogue scale (VAS) score was 6, with breakthrough pain upon touching the right upper limb reaching VAS scores of 8 to 10. The pain was primarily located along the right C5–C6 nerve distribution, affecting the lateral upper arm, forearm, and thumb, and was accompanied by nausea, vomiting, loss of appetite, and skin itchiness. Physical examination showed herpes scars on the right upper limb, normal muscle strength and tendon reflexes in all limbs, and a negative Babinski reflex. Post-transfer treatment included 20 mg oral duloxetine twice daily, topical compound lidocaine cream, and patient-controlled intravenous analgesia using a mix of 100 mg morphine, 200 mg flurbiprofen axetil, and 5 mg tropisetron hydrochloride in 65 mL 0.9% NaCl (background dose: 1 mL/hour, bolus: 5 mg). The next day, her pain improved (VAS score of 3), and on 16 September, she underwent computed tomography-guided selective right C5–C6 DRG pulsed radiofrequency modulation (temperature: 42°C, frequency: 2 Hz, pulse width: 20 ms, duration: 600 s). This treatment reduced the pain to a VAS score of 2. However, she still experienced mild nausea and vomiting.
On 18 September, the patient developed stabbing pain in the left neck, shoulder, and upper limb along with small red rashes. Because of significant anxiety and the nature of the symptoms, a dermatological consultation was requested. A high probability of herpes zoster led to the initiation of 3 g intravenous foscarnet sodium for antiviral treatment. On 19 September 2023, her nausea and vomiting worsened, and she developed urinary retention; these were considered adverse reactions to the opioids. Treatment adjustments included catheterization, reduction of the patient-controlled intravenous analgesia background dose, and the addition of 5 mg oral metoclopramide tablets three times daily. By 21 September 2023, the patient reported significant left upper limb pain (VAS score of 5) and strongly requested nerve block treatments. She received a left C5–C6 DRG block and a left stellate ganglion block, which alleviated her pain (VAS score of 3).
On September 25, she continued to experience significant pain and worsening nausea, vomiting, and hiccups. Consultations with gastroenterology and dermatology considered the small red rashes on the left neck and shoulder area to be folliculitis but could not identify a clear cause of the vomiting. It was recommended to temporarily administer 40 mg intravenous methylprednisolone once, then discontinue. By 27 September, the patient’s pain and nausea had improved, and she requested discharge.
On 10 October 2023, the patient developed dysfunction in urinary and bowel control, leading to urinary retention, which was symptomatically managed by the urology outpatient service. On 18 October, she exhibited frequently occurring spasmodic episodes in the right upper limb; these were characterized by excessive flexion of the right fingers and wrist, resembling a clenched hand. She was admitted to our hospital’s neurology department. Physical examination revealed non-cooperation during muscle strength tests of the right upper limb, while all other muscle strengths were at grade 5. She also exhibited unstable right-sided finger-to-nose testing, bilateral knee reflexes (+++), bilateral ankle reflexes (++++), positive bilateral Babinski reflexes, and negative meningeal signs.
On 22 October 2023, cervical spine MRI with contrast showed longitudinal high signal intensity on T2-weighted imaging from the medulla to C1–C7, with mild enhancement visible on gadolinium contrast MRI (Figure 2). Cerebrospinal fluid (CSF) analysis after lumbar puncture revealed a white blood cell count of 19/μL (reference range, 0–8/μL), protein concentration of 0.64 g/L (reference range, 0.2–0.4 g/L), and chloride concentration of 134.8 mmol/L (reference range, 120–132 mmol/L). A cell-based assay detected positive AQP4 antibodies (1:100), whereas tests for anti-myelin oligodendrocyte glycoprotein, anti-glial fibrillary acidic protein, and anti-myelin basic protein antibodies were negative. The CSF also showed IgG oligoclonal bands with ≥2 bands; serum tests mirrored these findings with positive AQP4 antibodies (1:100) and negative anti-myelin oligodendrocyte glycoprotein, anti-glial fibrillary acidic protein, and anti-myelin basic protein antibodies (Figure 3). Serum anti-nuclear antibodies were positive with a titer of 1:1000, and anti-centromere B antibodies were also positive, suggesting a possible coexisting connective tissue disorder. The patient was treated with methylprednisolone pulse therapy (1 g/day for 5 days), followed by gradual tapering; intravenous immunoglobulin (17.5 mg/day for 5 days); and oral mycophenolate mofetil (0.5 g twice daily). Upon discharge, she still experienced right upper limb spasms, although they were less frequent. Follow-up cervical spine MRI with contrast on 7 March 2024 showed no significant abnormalities (Figure 4). The spasms in her right upper limb had significantly decreased, and she was maintaining treatment with oral mycophenolate mofetil (0.75 g twice daily) and prednisone (10 mg daily). The timeline of the patient’s disease is shown in Figure 5.
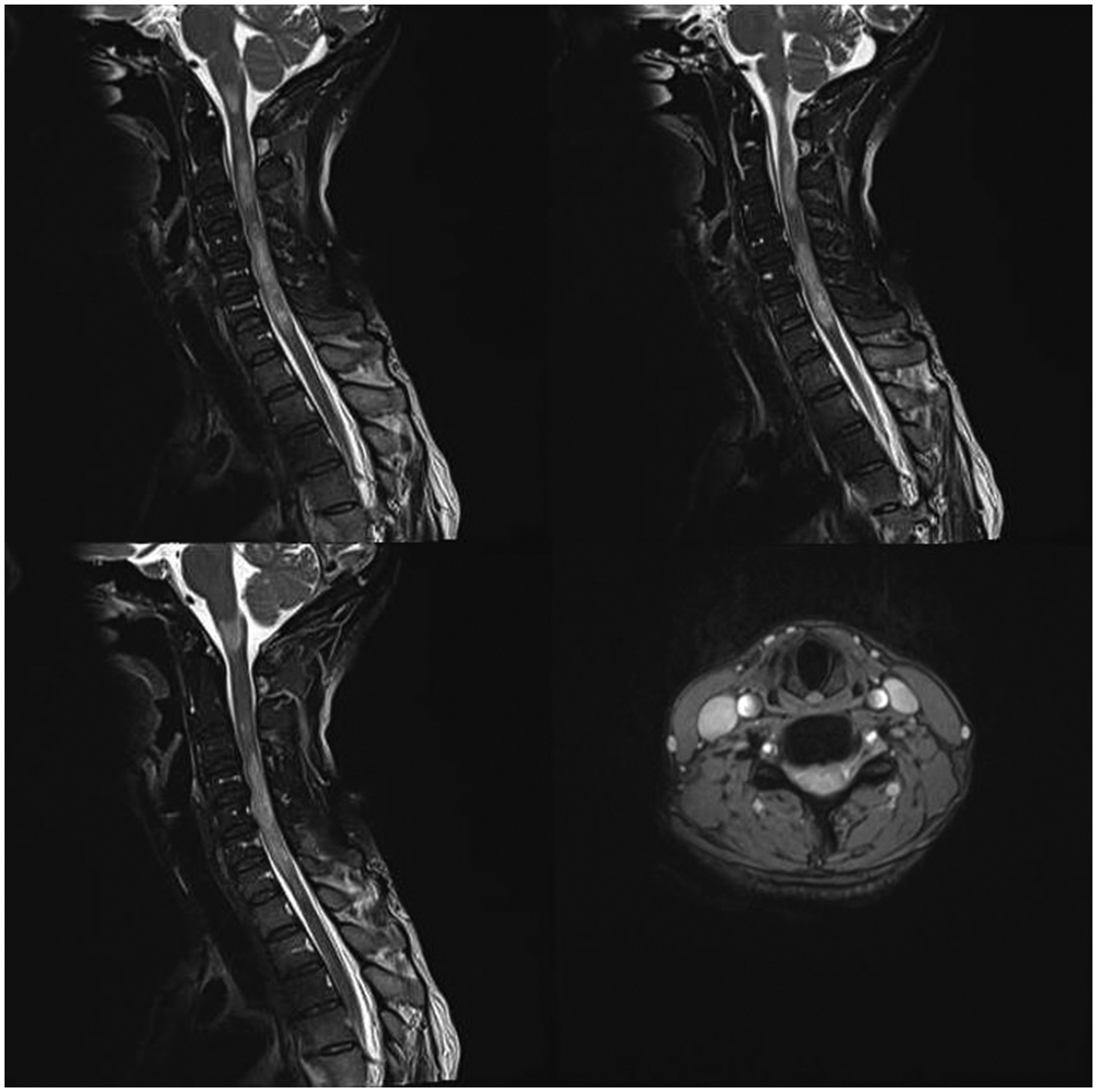
Initial magnetic resonance imaging of the cervical to thoracic spinal cord.

Detection of serum antibodies related to central nervous system demyelinating diseases using a cell-based assay. AQP4, aquaporin-4; MOG, myelin oligodendrocyte glycoprotein; GFAP, glial fibrillary acidic protein; MBP, myelin basic protein.

Follow-up magnetic resonance imaging of the cervical to thoracic spinal cord.
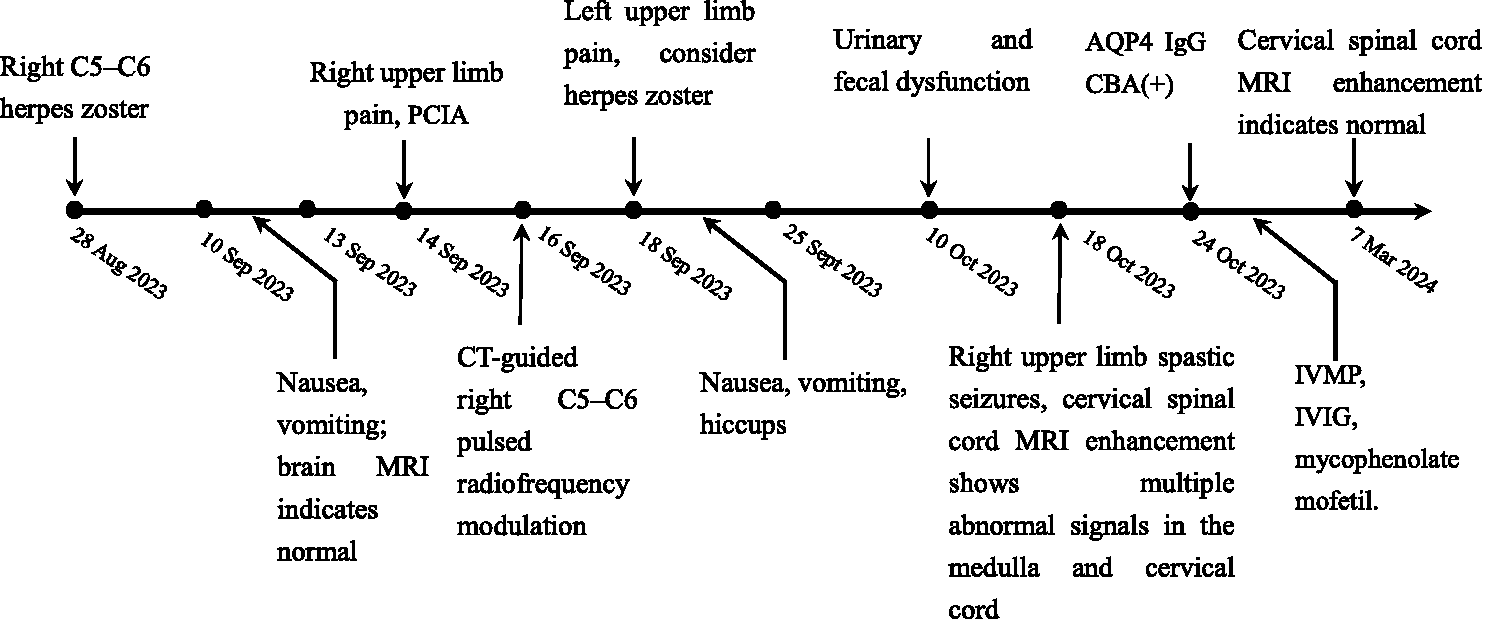
Timeline of patient’s disease. PCIA, patient-controlled intravenous analgesia; AQP4, aquaporin-4; CBA, cell-based assay; MRI, magnetic resonance imaging; CT, computed tomography; IVIG, intravenous immunoglobulin; IVMP, intravenous methylprednisolone.
To ensure the patient’s privacy, detailed information has been removed. Written informed consent was obtained prior to treatment, and the patient provided written informed consent for the publication of her data. The reporting of this study conforms to the CARE guidelines. 13
Discussion
We analyzed the demographic, clinical, imaging, laboratory, treatment, and recovery outcomes of 11 patients with AQP4+ NMOSD triggered by herpes zoster, including 10 cases published after 2008 and our own reported case. We found that all 11 patients were female, including 1 pregnant woman. The average age at onset was 47 years (range, 17–77 years), with a median age of 48 years; this is older than the 18-year median age reported by Sellner et al. 14 for virus-associated NMO syndromes. The patient group comprised seven Asians and four Europeans. Of these, five patients had VZV infections involving the cervical nerve roots. Two patients had involvement of both the cervical and thoracic nerve roots, another two had only thoracic nerve root involvement, and the remaining two had lumbar nerve root involvement. The median time to onset of AQP4+ NMOSD following VZV infection was approximately 3 weeks, similar to the 5 weeks reported in previous literature. 14 All patients exhibited motor, sensory, or sphincter dysfunctions, and MRI scans at the time of onset showed longitudinally extensive transverse myelitis affecting more than three vertebral segments. Seven patients did not have optic nerve damage, two patients had optic neuritis, and two patients’ records did not report on this aspect. CSF analysis revealed that four patients had elevated VZV antibodies at onset, three patients had normal levels, and four patients’ records did not report these details. When serum AQP4 antibodies were positive, CSF polymerase chain reaction for VZV DNA was negative in seven patients, with four not reported.
Regarding treatment, all patients were treated with methylprednisolone; eight used acyclovir, seven of whom (within 1–4 weeks of onset) used it in combination with methylprednisolone. Two patients received intravenous immunoglobulin, one underwent plasma exchange, and four received immunosuppressive therapy. Only one patient relapsed 3 years 6 months after being diagnosed and treated for AQP4+ NMOSD; the others did not relapse during the follow-up period. In terms of treatment outcomes, two patients fully recovered, seven patients had residual motor or sensory impairments, and two patients’ records did not report on this aspect (see Table 1 for details).
AQP4-Ab Positive NMOSD Cases Associated with Herpes Zoster: Review of Literature

F, female; AQP4, aquaporin-4; ND, not determined; MP, methylprednisolone; IVMP, methylprednisolone pulse therapy; PLEX, plasma exchange; IVIG, intravenous immunoglobin; VZV, varicella zoster virus; LETM, longitudinally extensive transverse myelitis; CSF, cerebrospinal fluid; MMF, mycophenolate mofetil; MRI, magnetic resonance imaging; PCR, polymerase chain reaction.
This case further reinforces the notion that infections can trigger attacks of AQP4+ NMOSD. According to the literature, 31.0% of patients with NMOSD reported a preceding influenza-like illness, 15 and 25.4% experienced a viral illness before the onset of NMO. 16 However, reports linking these illnesses to anti-AQP4 antibodies have been rare. Increasingly, case reports suggest a causal relationship between viral infections and AQP4 antibodies.5–9,11,12,17–24 This relationship can be explained by theories such as bystander activation, wherein inflammatory damage to epithelial cells expressing AQP4 activates AQP4-specific B cells, or molecular mimicry, where structural epitopes or peptide sequences of the infectious agent mimic those of the host’s own proteins. 14 Recent studies also indicate that infections can increase interleukin-6 in the CSF, activating an inflammatory cascade that promotes the production of AQP4 antibodies. 25
Research by Sellner et al. 14 shows that parainfectious NMOSD typically follows a monophasic course: most patients experience visual problems (63.6%) and neurological symptoms that progressively worsen, with only 25% fully recovering, and the highest rate of relapse occurs within the first year of symptom onset. However, our study showed that among 11 patients, only 1 relapsed 3.5 years after being diagnosed and treated for AQP4+ NMOSD. Interestingly, unlike traditional NMOSD, where more than half of patients develop severe vision loss in at least one eye within 5 years of an attack, 16 our findings suggest that AQP4+ NMOSD following VZV infection tends to spare the optic nerves. This difference may be related to the shorter follow-up duration or the involvement of different viral infections, but the specific mechanisms require further investigation. This case is also the first reported instance of a patient with VZV-induced AQP4+ NMOSD presenting initially with persistent nausea and vomiting and experiencing fluctuating relapses throughout the disease course.
Patients with herpes zoster neuralgia often initially seek treatment from pain management or dermatology specialists because of rashes and pain, after which they receive antiviral therapy, anti-neuropathic pain medications, and nerve blocks. However, during the course of the disease, some patients with herpes zoster may develop or have concurrent conditions such as tumors
26
or NMOSD, and clinical symptoms not directly associated with VZV infection may be overlooked by physicians. Although NMOSD following herpes zoster is rare, it remains a critical consideration for clinicians. For example:
Our patient experienced severe, persistent nausea and vomiting. Although an initial head MRI in the gastroenterology department showed normal results and some improvement, the symptoms recurred after transferring departments. At that time, only adverse reactions to opioids were considered, not the symptoms caused by the disease itself (e.g., area postrema syndrome, one of the core symptoms of NMOSD). The area postrema, located on either side of the fourth ventricle,
27
serves as the human vomiting center and, together with the solitary nucleus, controls the vomiting and belching chemoreceptor. The blood capillaries in this area have fenestrated endothelium and lack tight junctions, resulting in a relatively permeable blood–brain barrier. Astrocytes in this region highly express AQP4, making them susceptible to AQP4 antibody attacks.
28
As a core symptom of NMOSD, area postrema syndrome can appear early in the disease as an isolated clinical symptom, primarily manifesting as intractable belching, nausea, and vomiting. This may be related to the high AQP4 expression and disruption of the blood–brain barrier at this site.
29
Therefore, when encountering unexplained belching, nausea, and vomiting in clinical settings, APS should be considered, and further investigations such as serum or CSF AQP4 antibody testing and cranial and spinal MRI should be carried out. During the course of the disease, our patient developed new-onset stabbing pain in the left neck, shoulder, and upper arm; this was initially misdiagnosed as new-onset left herpes zoster. This ultimately suggested the possibility of acute myelitis presenting with radicular neuralgia, painful spasms, and Lhermitte’s sign. Pain specialists often overlook symptoms of central pain. When new symptoms appear that are inconsistent with the DRG locations affected by VZV, such as persistent nausea, vomiting, sensory disturbances, and ataxia, and particularly when they are challenging to diagnose, it may be useful to apply diagnostic principles such as the MIDNIGHTS
30
or VINDψCATE frameworks.
31
These principles remind us of the importance of conducting multiple physical examinations when new symptoms arise during the course of a disease. In China, the MIDNIGHTS framework is commonly used; it stands for Metabolism/Malnutrition, Inflammation/Immune, Degeneration, Neoplasm, Infection, Gland, Hereditary, Toxication/Trauma, and Stroke. The clinical manifestations of NMOSD are varied and easily misdiagnosed. If one of the six core symptoms of NMOSD appears after a VZV infection
4
(i.e., optic neuritis, acute myelitis, area postrema syndrome, acute brainstem syndrome, acute diencephalic syndrome, or cerebral syndrome), clinicians should test for serum AQP4 antibodies.
Footnotes
Acknowledgment
We thank the patient described in this report.
Author contributions
All authors contributed to the study conception and design. Material preparation and case collection and analysis were performed by YH, AX, HH, SX, JC, SL, XH, ZZ, and DW. The first draft of the manuscript was written by YH and AX, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Data availability statement
Data are available upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Ethics statement
For this case report, the authors obtained approval for an exemption from ethics approval from the Ethics Committee of The Fifth Affiliated Hospital of Wenzhou Medical University. All identifiable patient information has been removed to protect the patient’s privacy. Written informed consent for treatment was obtained from the patient, and a separate consent form was signed by the patient for the publication of the data and images.
Funding
This study was supported by the Lishui Science and Technology Plan Project [Grant Number: 2022SJZC020] and the Medical Health Science and Technology Project of Zhejiang Provincial Health Commission [Grant Number: 2024KY564].
