Abstract
The Prevotella genus consists of obligate anaerobic Gram-negative bacteria that are symbiotic with the oral, intestinal, and vaginal mucosa. While several species of Prevotella have been implicated in pulmonary infections, identification of Prevotella as the causative agent of lung abscess is uncommon because of the requirement for stringent anaerobic culture conditions. In this report, we highlight a case of lung abscess caused by Prevotella salivae and Prevotella veroralis, underscoring the importance of third-generation metagenomic sequencing using devices from Oxford Nanopore Technologies for the precise diagnosis of specific pathogens.
Background
The Prevotella genus consists of obligate, anaerobic, Gram-negative, rod-shaped bacteria, with Prevotella melaninogenica serving as the representative species. These microorganisms are frequently present in the oral cavity, female reproductive tract, and feces of both humans and animals. 1 While no specific pathogenic species within the Prevotella genus has been identified, this genus is linked to a range of ailments such as inflammatory autoimmune diseases, opportunistic infections, endogenous infections, bacterial vaginosis, and the development and occurrence of oral biofilm-related diseases. Although some Prevotella species have been demonstrated as pathogens in pulmonary infections or pleural effusion,2,3 it is rare to isolate Prevotella as the cause of a lung abscess. 4
In recent years, with advances in diagnostic techniques, third-generation metagenomic sequencing technology has been applied to assist in clinical diagnoses, enabling timely diagnosis and identification of anaerobic infections. As a result, reports of anaerobic infections have been increasing. However, there are currently no reports of Prevotella salivae and Prevotella veroralis, especially in relation to lung infections. Using third-generation metagenomic pathogen detection technology, we identified a rare case of right upper lobe lung abscess caused by Prevotella. This case report aims to highlight a rare presentation of Prevotella lung abscess and to provide insights that could assist physicians in diagnosing and managing similar cases, thereby filling an important gap in current clinical knowledge.
Case presentation
A 67-year-old male patient recently diagnosed with superinfected lung cancer, with characteristic symptoms, immunosuppression, and resistance to the initial treatment, was sent to a reference center, where complex investigations identified Prevotella infection. Antibiotic therapy was indicated as the appropriate treatment.
Investigations
The patient exhibited elevated C-reactive protein, procalcitonin, serum amyloid A, and interleukin-6 levels. His C-reactive protein level was 157.3 mg/L, and the complete blood count revealed an elevated white blood cell count, primarily consisting of neutrophils. Computed tomography imaging of the chest before treatment showed right upper lung lesions with local necrosis and concomitant infection as shown in Figure 1(a). After treatment, the compression and dilation of the middle and lower lobe of the right lung were reduced compared with the pre-treatment condition (Figure 1(b)).

Chest imaging. (a) Pre-treatment and (b) post-treatment.
Conventional culture techniques proved ineffective in isolating pathogens from blood and sputum cultures. To address this diagnostic challenge, the patient’s bronchoalveolar lavage fluid sample underwent third-generation metagenomic sequencing using Oxford Nanopore Technologies’ MinION device (Oxford, UK). In addition to pathogenic microorganisms, the original microbial comparison results also indicated a large number of background microorganisms. Therefore, the following specific criteria were used for pathogenic bacteria identification: (1) The species coverage ratio of bacteria, viruses, and parasites was 10 times higher and the coverage ratio of fungi was more than five times higher than that of other microorganisms. For Mycobacterium tuberculosis, because the extracted DNA content was low, only one sequence was detected, which was considered positive. (2) Microorganisms including bacteria (except for Mycobacterium tuberculosis), fungi, and viruses with less than three sequences were excluded. 5 Meanwhile, the sample was subjected to laboratory culture and microscopic examination for verification. Microscopic analysis revealed a substantial presence of inflammatory white blood cells and bacteria with diverse morphology, exhibiting phagocytic phenomena. The third-generation metagenomic analysis further identified the presence of Prevotella salivae (16,894 reads) and Prevotella veroralis (14,136 reads).
Treatment and outcome
The patient received initial empirical antimicrobial therapy consisting of imipenem/cilastatin sodium combined with vancomycin, but the response was deemed unsatisfactory. On the eighth day of hospitalization, percutaneous lung puncture drainage was conducted under ultrasound guidance. The drainage yielded grayish-green pus-like fluid, which was subsequently subjected to metagenomic sequencing and culture. The next day, metagenomic sequencing of the lung puncture drainage indicated the presence of Prevotella salivae and Prevotella veroralis. The drainage fluid culture exhibited the presence of Gram-negative bacteria after 30.8 hours within the laboratory setting. Subsequently, 6 days later (on the 14th day), the culture indicated the presence of Prevotella salivae. Considering the comprehensive diagnostic findings, the antibiotic regimen was modified to include clindamycin (0.6 g) administered every 6 hours, in addition to piperacillin/tazobactam (5 g) administered every 8 hours. In addition, the drainage process was intensified. The patient’s body temperature gradually normalized, and his symptoms improved (Figure 2). The last chest computed tomography scan before discharge indicated improvement.
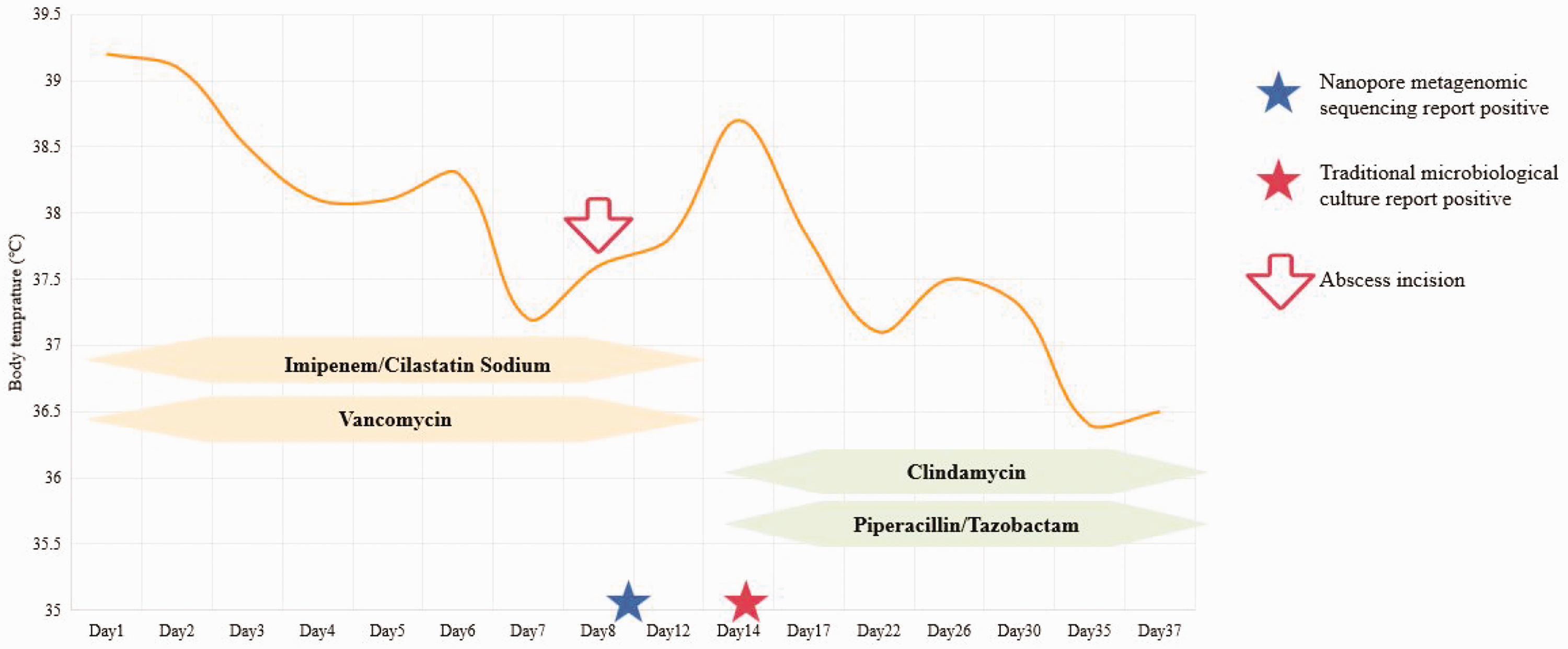
Body temperature curve and treatment during hospitalization.
Discussion
In this study, we describe the first report of a patient with Prevotella salivae and Prevotella veroralis lung abscess diagnosed using Nanopore sequencing. Lung abscess, a potential complication of pneumonia, is a rare occurrence in adults. A PubMed literature search revealed multiple case reports showing the involvement of Prevotella in infections, such as Prevotella pleuritidis, Prevotella intermedia, and Prevotella dentalis, as the sole or common causative agent.4,6,7 Most Prevotella infection reports are presented as cases caused by the genus Prevotella because of poor oral hygiene or oral infections or after dental surgery. In our case, the patient had no signs or symptoms of dental infection and denied recent dental procedures.
If the quantity of a pathogen is below the detection limit of traditional culture methods or if the viability and recovery rate of the pathogen are compromised because of unfavorable specimen processing, diagnosis is often delayed. 4 The low equipment cost, short turnaround time, and portable size of the MinION device from Oxford Nanopore Technologies allow its use in clinical microbiology laboratories. 8 Nanopore sequencing based on third-generation sequencing platforms helps overcome cost and time limitations and provides increased diagnostic accuracy. This was well demonstrated in our patient’s case, where Nanopore sequencing was able to identify potential pathogens associated with lung abscess, guiding targeted therapy. The Nanopore sequencing results include the sequence number and relative abundance. Pathogens reported at high abundance may be associated with true infection, while low-abundance pathogens may be associated with true infection or contamination. Because sequence readings from colonizers and/or contaminants are commonly encountered in metagenomic next-generation sequencing analysis, these results must be interpreted with caution in the clinical setting of the patient. It is necessary to interpret this Nanopore sequence result in conjunction with other clinical data, such as the physical examination, imaging findings, and other laboratory results.
Bacterial lung abscess is a disease process that poses a significant threat and necessitates timely identification and treatment by healthcare professionals to mitigate potentially fatal complications.9,10 The growing accessibility of molecular diagnostic methods, such as third-generation metagenomics, for elucidating the microbial composition of abscesses aligns with the diagnostic findings of the microbiology laboratory in this instance and offers more conclusive information regarding microbial abundance. This advancement has the potential to shape future development of the most effective treatment approaches for lung abscess.
In the case of our patient, the administration of moxifloxacin, a commonly prescribed medication for community-acquired pneumonia, did not yield any clinical improvement. By utilizing Nanopore sequencing and the microbiology laboratory diagnosis, we were able to confirm the presence of a Prevotella infection. Prevotella sp. typically exhibit sensitivity to metronidazole, imipenem, meropenem, piperacillin/tazobactam, ceftriaxone, and tigecycline while demonstrating resistance to ampicillin, clindamycin, tetracycline, and moxifloxacin.11,12 Hence, an unequivocal diagnosis can contribute to the clinical rationale and precise administration of antimicrobial therapy, resulting in a favorable prognosis for the patient.
This case highlights the diagnostic challenges associated with Prevotella lung abscesses and the potential of third-generation metagenomic sequencing to overcome these limitations. By providing a comprehensive analysis of the microbial composition and abundance, this technique can assist in the accurate identification of pathogens and guide targeted antimicrobial therapy.
Footnotes
Acknowledgements
The authors would like to thank Dian Diagnostics Group Co., Ltd. for metagenomic DNA detection. We thank Dr Guannan Ma and Lei Zhang for helpful discussions.
Author contributions
Xiaorui Wu, Tianqi Qi, and Mingding Zhang were responsible for the collection, collation, and writing of the manuscript. Junfang Bi was responsible for the concept development, revision, and review of the manuscript. All authors contributed to the article and approved the submitted version.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics statement
The publication of this case was approved by the ethical committees of Aerospace Center Hospital. Written informed consent was obtained from the patient for publication of the case report and the accompanying images.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
