Abstract
Objective
To observe the effects of 4 weeks of voluntary wheel running on depressive-like behavior in a rat chronic stress-induced depression model to explore the anti-depressive mechanism of exercise.
Methods
In this observational study, 36 Sprague-Dawley rats were randomly divided into control, stress model, and stress exercise groups (12 rats/group). The control group received no intervention, and the stress model and stress exercise group rats underwent chronic mild unpredictable stress and isolation. The stress exercise group rats also underwent 4 weeks of voluntary wheel running. Behavioral changes and hippocampal protein and mRNA expression levels were detected.
Results
Voluntary wheel running significantly increased horizontal and vertical movements, sucrose intake, and the sucrose preference percentage and reduced immobility time in the forced swimming test in depression model rats. The hippocampal tumor necrosis factor-α, interleukin (IL)-6, IL-1β, and quinolinic acid levels were significantly decreased, while the IL-4, IL-10, and kynurenic acid levels were significantly increased. Kynurenine-3-monooxygenase and 3-hydroxyanthranilate-3, 4-dioxygenase mRNA levels were downregulated, and kynurenine aminotransferase mRNA was upregulated.
Conclusion
Voluntary wheel running improved depressive-like behavior in depression model rats. The mechanism may be related to a kynurenine pathway metabolite level imbalance, which has neurotoxic and neuroprotective effects, caused by long-term voluntary wheel running.
Keywords
Introduction
Depression is often manifested by mood changes, decreased interest, feelings of inferiority, unwillingness to communicate with others, and suicidal tendencies. Studies have shown that decreases in 5-hydroxytryptamine levels in various brain regions are correlated with the occurrence of depression. 1 The mechanisms of action of various antidepressants used in clinical practice are mostly based on the monoamine hypothesis of depression, but one-third of patients do not respond to conventional drug therapy. 2 The kynurenine pathway (KP) is the main decomposition and metabolic pathway of the essential amino acid tryptophan (TRP) in the body. 3 In recent years, the imbalance between the neurotoxic and neuroprotective metabolites of the KP has been proposed as the main driving factor of depression. For example, when KP activation is genetically or pharmacologically blocked, systemic immune stimulation (i.e., lipopolysaccharide administration) cannot induce depressive-like behavior in rodents. 4 Similarly, studies of patients with inflammatory diseases or interferon-alpha treatment have shown a strong correlation between depressive symptoms and production of the neurotoxic metabolite kynurenine (KYN), indicating the KP as a potential mechanism. 5 In addition, several cross-sectional studies have shown that patients with depressive spectrum disorders, including major depression, bipolar depression, and suicidal tendencies, have decreased and/or increased kynurenic acid (KYNA) and 3-hydroxy-kynurenine or quinolinic acid (QUIN) levels in the blood and cerebrospinal fluid.6–9
Sports exercise, as a low-cost, non-invasive, and non-pharmacological therapy without complications, has been recognized as a psychological treatment method. 10 A survey in the United States showed that 80% of 1750 psychologists believed that sports exercise was an effective means of treating depression. 11 Many college students suffer from depression because of academic and other setbacks, and sports exercise can help alleviate or eliminate these psychological disorders. 12 Studies have also shown that exercise can improve symptoms of many existing mental illnesses or help prevent them before mental health problems arise. 13 Research has also confirmed that both acute and long-term exercise can affect the production of KP components in the body, as evidenced by changes in KP-related metabolites and enzyme systems. 14 In addition, acute or long-term exercise of low to moderate intensity can significantly reduce the plasma neurotoxic metabolite QUIN and increase the neuroprotective metabolite KYNA in clinical patients (type 2 diabetes, depression, dementia, and multiple sclerosis). 15 Therefore, further research is needed to confirm whether the possible mechanism by which exercise improves depression involves balancing the levels of KP metabolites with neurotoxic and neuroprotective effects. This study used a widely accepted method, chronic mild unpredictable stress stimulation combined with social isolation, to replicate the rat depression model. The effects of exercise on behavior and KP components in the hippocampus of depressed rats were explored to understand the mechanism by which exercise protects against depression and to help prevent and treat depression.
Materials and methods
Experimental animals and grouping
Thirty-six clean-grade male Sprague-Dawley rats were used, with a weight of 224.53 ± 15.27 g, purchased from the Shanghai Slaik Experimental Animal Co., Ltd. (Shanghai, China) [Experimental Animal Production License Number: SCXK(Hu)2018-0006]. The laboratory temperature and humidity were 20 ± 2°C and 55% ± 10%, respectively, and the rats were maintained on a 12-hour light/dark cycle. During the experiment, the rats were allowed free access to food and water (except for the fasting and water deprivation periods for the stress model and stress exercise groups). The rats were acclimated to the laboratory environment for 1 week. They were then randomly divided into a blank control group, stress model group, and stress exercise group, with 12 rats in each group. The stress model and stress exercise groups were individually housed and fed. The animal study was reviewed and approved by the experimental animal ethics committee (No: 2022/1205-05/2L). The reporting of this study conforms to the STROBE guidelines. 16
Instruments and reagents
The following equipment was used: 7500 Real-Time PCR instrument, constant voltage and current electrophoresis apparatus (Beijing Liuyi Instrument, Beijing, China), digital gel imaging system (Shanghai Tianneng Technology Co., Ltd., Shanghai, China), Image-Pro Plus Analysis Software computer image analysis instrument (Bio-Rad, Hercules, CA, USA), low-temperature, high-speed centrifuge (Shanghai Chuanhong Experimental Instrument Co., Ltd., Shanghai, China), electronic balance BS224S (Shanghai Wanning Precision Scientific Instrument Co., Ltd., Shanghai, China), nucleic acid ultraviolet spectrophotometer (Shandong Luyao Scientific Instrument Co., Ltd., Qingdao, China), and rat voluntary running wheel (Shanghai Rongqi Biotechnology Co., Ltd., Shanghai, China). The QUIN enzyme-linked immunosorbent assay (ELISA) kit (product number: HB-PS2797G) and the KYNA ELISA kit (product number: ARD40379) were purchased from Guangzhou Auride Biotechnology Co., Ltd. (Guangzhou, China).
Replication of the chronic stress depression model
The depression model was replicated using chronic mild unpredictable stress combined with isolation rearing. 17 The stress stimuli included 24-hour water deprivation, 5-minute tail suspension, 2-hour restraint stress, 5-minute forced swimming in cold water (4°C), 24-hour food deprivation, reversed light–dark cycle, and 20-minute foot shock (0.8 A, 4-s duration, 5-minute interval), totaling seven stimuli, with one stimulus administered daily in a random order. The same stimulus was not administered consecutively, and the order of appearance of each stimulus was different in each round, for a total of 4 weeks.
Interventions for each rat group
1) The normal control group underwent normal feeding without any intervention measures; 2) the stress model group was subjected to stress for 4 weeks starting from the second week; 3) the stress exercise group underwent voluntary wheel training throughout the experiment with a freely rotating running wheel (33-cm diameter, 10-cm width) in the cage, and the animals were allowed to exercise freely. Infrared detection equipment and a data analysis system (Shenzhen Rayward Life Technology Co., Ltd., Shenzhen, China) automatically collected data. Rats were subjected to stress once a day for 4 weeks.
Sucrose preference test
Rats were housed individually in cages. To train the rats to adapt to a 1% sucrose solution, two identical plastic bottles were placed at opposite ends of the cage, one containing drinking water and the other containing a 1% sucrose solution, for 5 days before the experiment. The position of the two bottles was randomly changed every day to complete habituation to sucrose. The sucrose preference index was calculated by removing the water bottle for 16 hours, allowing the rats to drink for 4 hours, and then measuring the amount of water and sucrose solution consumed during this period. 18
Open field test
On the day after the forced swimming test, rats in each group were subjected to an open field test. 19 The box was 80 × 80 ×40 cm, with the inside painted black and the bottom divided into 19 equal squares with white lines. The rats were gently placed in the center square when they were calm, and the numbers of times they crossed the squares (horizontal movement) and stood on their hind legs (vertical movement) were counted for 5 minutes.
Forced swimming test
On the day after the sucrose preference test, rats were placed in a circular plastic container with their heads facing the container wall, and the container was slowly lowered into the water. The height of the circular plastic container was 60 cm, and the diameter was 40 cm. The water temperature was controlled to approximately 22°C. The rats swam freely in the container for 5 minutes, and their activity was recorded on video. The immobility time of the rats during the 5-minute test was calculated.20–22 The water in the container was changed for the next rat.
Sample preparation and detection methods
Detection of KYNA and QUIN content in the rat hippocampus
After the final week of behavioral testing, the rats were subjected to a 24-hour fasting period. The rats were anesthetized with isoflurane (3%–4% induction, 2%–2.5% maintenance), and the hippocampal tissue was quickly removed on a super-clean bench, placed in a sterilized centrifuge tube, and stored at −80°C. The levels of KYNA and QUIN and the cytokines tumor necrosis factor (TNF)-α, interleukin (IL)-6, IL-1β, IL-4, and IL-10 in the rat hippocampus were detected strictly according to the instructions of the ELISA kit. An appropriately-sized tissue block was washed in pre-cooled phosphate-buffered saline (0.01 M, pH 7.0–7.2) to remove blood (lysis of red blood cells in the homogenate affects the measurement). After weighing, the tissue was cut into small pieces and added to a glass homogenizer with the corresponding volume of phosphate-buffered saline (generally in a mass-to-volume ratio of 1:9). The mixture was ground thoroughly on ice. To further lyse the tissue cells, ultrasonic disruption of the homogenate was performed (with specific attention to cooling in an ice bath during this process). Finally, the prepared homogenate was centrifuged at 5000 × g for 5 to 10 minutes, and the supernatant was collected for detection. 23
Quantitative real-time PCR detection of the relative expression levels of kynurenine-3-monooxygenase (KMO), 3-hydroxyanthranilate-3, 4- dioxygenase (3-HAO), and kynurenine aminotransferase (KAT) mRNA in the rat hippocampus
The amplification reaction conditions were as follows: pre-denaturation at 94°C for 1 minute, denaturation at 94°C for 5 s, annealing at 60°C for 20 s, and extension at 72°C for 10 minutes for 40 cycles. A melting curve was plotted using the 2−ΔΔCt method for relative quantification analysis. The primers used were as follows: KMO forward 5′-TCCCTTCCACCTGAAGTCAC-3′, reverse 5′-AGTCTCAAAGCCCGCATTC-3′; 3-HAO forward 5′-AGGTGACAATGGGGAGGACAG-3′, reverse 5′-TTACAGGCAGGGTCTTGTGT-3′; KAT forward 5′-GCCTTGTTCACAGCCTTTCA-3′, reverse 5′-ACCTCCAGCCATCATTGTCA-3′; GAPDH forward 5′-CAACTCCCTCAAGTTGTCAGCAA-3′, reverse 5′-GGCATGGACTGTGGTCATGA-3′. The 2−ΔΔCt method was used to calculate the relative expression levels of KMO, 3-HAO, and KAT mRNA.
Statistical methods
Data analysis was performed using SPSS 21.0 statistical software (IBM Corp., Armonk, NY, USA). Measurement data are expressed as the mean ± standard deviation (x ± s). One-way analysis of variance was used for analysis of intergroup differences, and further pairwise comparisons were performed using the least significant difference test. A value of P < 0.05 was considered statistically significant.
Results
This is an observational study, and the experimental design and implementation are shown in Figure 1.

Schematic diagram of the experiment.
Comparison of body weights of rats in each group after chronic stress
Compared with the control group, the stress group rats showed a significant decrease in body weight after 4 weeks of chronic stress (P < 0.01). Compared with the stress group, the stress exercise group rats showed a significant increase in body weight (P < 0.05) (Table 1).
Comparison of body weight of rats in each group after chronic stress (n = 36).

Note: Compared with the control group, **: P < 0.01. Compared with the stress group, &: P < 0.05.
Changes in voluntary wheel running distance in the stress exercise group
The wheel running distance gradually increased from weeks 1 to 4 in the stress exercise group, from 9.95 ± 1.12 km in the first week to a peak of 27.58 ± 2.09 km in the fourth week. Compared with the wheel running distance in the first week, the distance in the second to fourth weeks was significantly increased (P < 0.01) (Table 2).
Changes in voluntary wheel exercise in rats in the stress exercise group (n = 12).

Note: SE: stress exercise group. Compared with the distance in the first week of wheel exercise, **: P < 0.01.
Behavioral changes in each group of rats
The sucrose preference test results showed that compared with the control group, the consumption and preference index of sucrose in the stress group were significantly decreased (P < 0.01). Compared with the stress group, the consumption and preference index of sucrose in the stress exercise group were significantly increased (P < 0.01) (Table 3).
Sugar water consumption and preference index of rats in each group (n = 36).
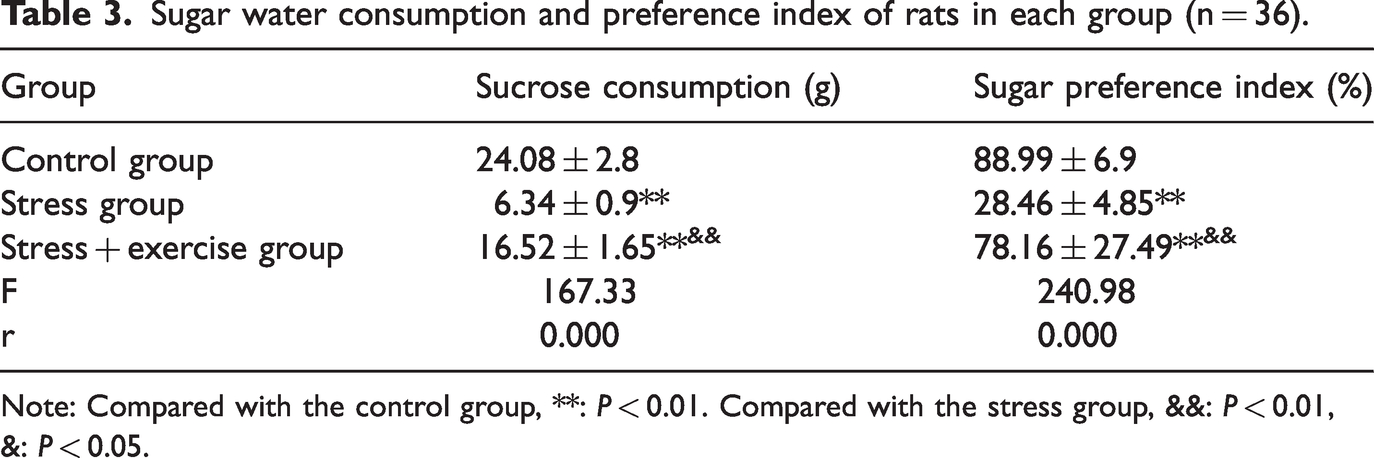
Note: Compared with the control group, **: P < 0.01. Compared with the stress group, &&: P < 0.01, &: P < 0.05.
The forced swimming test results showed that compared with the control group, the immobility time of rats in the stress group was significantly increased (P < 0.01). Compared with the stress group, the immobility time of rats in the stress exercise group was significantly decreased (P < 0.01) (Table 4).
Forced swimming immobility time (s) and the numbers of horizontal and vertical movements in the open field test compared among the groups (n = 36).

Note: Compared with the control group, **: P < 0.01. Compared with the stress group, &&: P < 0.01, &: P < 0.05.
The open field test results showed that compared with the control group, the horizontal and vertical movements of rats in the stress group were significantly decreased (P < 0.01). Compared with the stress group, the horizontal and vertical movements of rats in the stress exercise group were significantly increased (P < 0.01, P < 0.05) (Table 4).
Comparison of the expression levels of the inflammatory factors IL-1β, IL-6, TNF-α, IL-4, and IL-10 in the hippocampus of rats in each group
Comparisons of the expression levels of IL-1β, IL-6, and TNF-α in the three rat groups revealed statistically significant differences (P < 0.01) according to analysis of variance. In the stress group, the expression levels of IL-1β, IL-6, and TNF-α in the rat hippocampus were significantly increased compared with those in the normal control group (P < 0.01). In the stress exercise group, the expression levels of IL-1β, IL-6, and TNF-α in the hippocampus were significantly decreased compared with those in the stress model group (P < 0.01). Comparison of the expression levels of IL-4 and IL-10 in the three groups also showed statistically significant differences (P < 0.01) according to analysis of variance. In the stress model group, the levels of IL-4 and IL-10 in the rat hippocampus were significantly decreased compared with those in the normal control group (P < 0.01). In the stress exercise group, the expression levels of IL-4 and IL-10 in the hippocampus were significantly increased compared with those in the model group (P < 0.01) (Table 5).
Comparison of expression levels of the hippocampal inflammatory factors IL-1β, IL-6, TNF-α, IL-4, and IL-10 in different rat groups (n = 36).

Note: Compared with the normal control group, **: P < 0.01, *: P < 0.05; Compared with the stress group, &&: P < 0.01, &: P < 0.05.
IL, interleukin; TNF, tumor necrosis factor.
Comparison of hippocampal KYNA and QUIN levels among rat groups
The analysis of variance results indicated significant differences in KYNA levels among the three rat groups (P < 0.01). The hippocampal KYNA level in the stress group was significantly lower than that in the normal control group (P <0.01), while the hippocampal KYNA level in the stress exercise group was significantly higher than that in the stress model group (P < 0.05). The analysis of variance results also showed significant differences in QUIN levels among the three rat groups (P = 0.001). The hippocampal QUIN level of rats in the stress model group was significantly higher than that in the normal control group (P < 0.01), while the hippocampal QUIN level of the stress exercise group was significantly lower than that in the stress model group (P < 0.01) (Table 6).
Comparison of KYNA and QUIN in the hippocampus of rats in each group (n = 36).

Note: Compared with the normal control group, **: P < 0.01, compared with the stress group, &&: P < 0.01,
&: P < 0.05.
KYNA, kynurenic acid; QUIN, quinolinic acid.
Comparison of relative expression levels of KMO, 3-HAO, and KAT mRNA in the hippocampus of each rat group
The relative expression levels of KMO and 3-HAO mRNA in the hippocampus of each rat group were compared, and the differences were statistically significant (P < 0.01). The model group showed increased levels compared with those in the control group (P < 0.01), while the stress exercise group showed decreased levels compared with those in the stress model group (P < 0.05, P < 0.01). The relative expression level of KAT mRNA in the hippocampus of each group was compared, and the differences were statistically significant (P < 0.01). The model group showed a decreased level compared with that in the normal control group (P < 0.01), while the stress exercise group showed an increased level compared with that in the stress model group (P < 0.01) (Table 7).
Comparison of relative expression levels of KMO, 3-HAO, and KAT mRNA in the hippocampus of rats in each group (n = 36).

Note: Compared with the normal control group, **: P < 0.01, compared with the stress group, &&: P < 0.01,
&: P < 0.05.
KMO, kynurenine-3-monooxygenase; 3-HAO, 3-hydroxyanthranilate-3, 4-dioxygenase; KAT, kynurenine aminotransferase.
Discussion
Depression is one of the most common psychological disorders; however, treatment with antidepressant drugs has many drawbacks. Exercise can not only treat but can also prevent depression. Even a very small amount of exercise, such as walking for 20 minutes a day or gardening for 20 minutes, can have a significant effect on preventing depression. 24 This study combined physiological chemistry with applied behavioral tests, and the results showed that 4 weeks of voluntary wheel running exercise could significantly reduce the levels of the pro-inflammatory cytokines IL-1β, IL-6, and TNF-α in the hippocampus of depressed model rats while increasing the levels of the anti-inflammatory cytokines IL-4 and IL-10. This exercise also significantly increased the KYNA level and decreased the QUIN level. The relative expression levels of KMO and 3-HAO mRNA decreased, while that of KAT mRNA increased, indicating that 4 weeks of voluntary wheel running exercise can alter the abnormal TRP metabolism in depressed rats. This may also be a mechanism by which exercise intervention exerts its antidepressant effects.
The KP is composed of different active metabolites and related enzyme systems. First, TRP is oxidized by tryptophan 2,3-dioxygenase (TDO) and two types of indoleamine 2,3-dioxygenase (IDO-1 and IDO-2) to form the intermediate metabolite KYN. 25 Under normal physiological conditions, IDO activity is low, and TRP is mainly metabolized by TDO in the liver, where TDO is the rate-limiting enzyme of basic KP metabolism. When the body exhibits an immune response or stress state, TDO activity is induced by pro-inflammatory cytokines, and the isoenzyme IDO is overactivated, becoming the rate-limiting enzyme of KYN synthesis, leading to an increase in KYN levels in peripheral and central nervous tissues.26,27 Subsequently, depending on the cell type, KYN is further metabolized into neurotoxic products, mainly including QUIN, and neuroprotective products, mainly including KYNA. In central and peripheral tissues, KYN is mainly converted into 3-hydroxy-kynurenine, 3-hydroxyanthranilic acid, and QUIN3 by KMO and 3-HAO. 28 QUIN promotes neurodegeneration by inducing excitotoxicity in neurons through ionotropic glutamate receptors (iGluRs), N-methyl-D-aspartate receptors (NMDARs), alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptors (AMPARs), kainate receptors, and alpha-7-nicotinic acetylcholine receptors (α7-nAChRs) and further activates iGluRs by promoting glutamate (Glu) release in neurons. This inhibits Glu reuptake by astrocytes and blocks Glu synthetase expression, thereby disrupting the dynamic balance of the Glu-glutamine-gamma-aminobutyric acid cycle and exacerbating the excitotoxicity caused by Glu. 29 QUIN also induces immune imbalances and inflammation, leading to the production of various pro-inflammatory cytokines, thereby activating IDO. KMO is also upregulated under inflammatory conditions, leading to a vicious cycle of the KP and further promoting its neurotoxic metabolites.
In astrocytes of the central nervous system and skeletal muscle cells of the peripheral nervous system, KYN is catalyzed by KAT to synthesize KYNA, with KAT II synthesizing most of the KYNA in the central nervous system. In contrast to the neurotoxic activity of QUIN, KYNA is a neuroprotective metabolite that is a broad-spectrum iGluR (AMPARs, NMDARs, and kainate receptors) antagonist and a potent negative allosteric modulator of α7-nAChRs, thereby blocking the excitotoxicity of iGluRs and α7-nAChRs induced by QUIN. KYNA can also clear reactive oxygen and nitrogen species and extracellular Glu in neurons, exerting a neuroprotective effect. 30
The KP and depression
Increasing evidence suggests that abnormalities in KP metabolism are associated with the pathophysiological mechanisms of depression.4,31,32 Previous studies have found that depression patients exhibit dysregulation in KP metabolism, with abnormal levels of the neurotoxic metabolite QUIN and the neuroprotective metabolite KYNA. 33 A recent meta-analysis comparing the concentrations of KP pathway metabolites between patients with depression and healthy controls found significantly lower KYNA and KYN levels and KYNA/QUIN ratios in patients with depression. 34 Animal experiments have also confirmed the close relationship between KP pathway metabolites and the occurrence of depression. For example, a commonly used depression model, Flinders Sensitive Line rats, had lower KYNA levels in the prefrontal cortex than those in control Flinders Resistant Line rats. 35 In addition, a mouse model of peripheral nerve injury showed increased mRNA and protein levels of the enzymes KMO and QUIN, as well as a decreased KYNA level in the contralateral hippocampus. Another study found increased QUIN levels in the hippocampus of rats intraperitoneally injected with lipopolysaccharide, along with increased IL-1β, IL-6, and TNF-α levels in the plasma and hippocampus. 36 Similarly, rats treated with interferon showed significantly increased immobility times in the open field and forced swimming tests, along with increased QUIN levels in the hippocampus. After treatment with the antidepressant imipramine, the symptoms significantly improved. 37 Therefore, the results of these animal model studies suggest a close association between KP metabolism abnormalities and the development of depression. The results of this study indicated that in a stress model, the level of the neuroprotective metabolite KYNA in the hippocampus was significantly decreased, while the QUIN level was significantly increased. The pro-inflammatory cytokines IL-1β, IL-6, and TNF-α were significantly elevated, while the anti-inflammatory cytokines IL-4 and IL-10 were significantly decreased. The relative expression levels of KMO and 3-HAO mRNA were significantly upregulated, while the relative expression level of KAT mRNA was significantly downregulated, consistent with previous research findings.
Exercise and the KP
Research has shown that exercise can stimulate the KP. 38 Agudelo et al. found that 8 weeks of voluntary wheel running exercise in mice significantly increased the mRNA expression levels of PGC-1α, KAT1, KAT3, and KAT4 in skeletal muscle while decreasing the expression levels of TDO1 and KMO mRNA. 39 Plasma KYNA levels significantly increased and were significantly correlated with the gene expression levels of KAT1, KAT3, and KAT4 in skeletal muscle. Joisten et al. demonstrated that acute endurance exercise and resistance exercise training in 24 healthy adult males had a significant time effect on the KYN/TRP ratio in blood samples, and endurance exercise caused acute changes in human KP outcomes. 38 Specifically, acute resistance and acute endurance exercise significantly reduced the TRP/QUIN/KYNA ratio in the blood of healthy adult men while the KYNA and KYNA/KYN ratio was significantly increased, and the KAT4 mRNA expression level in peripheral blood mononuclear cells was significantly upregulated. Strasser et al. showed that TRP concentrations significantly decreased and KYN levels increased in trained athletes after acute exercise. 40 In addition, studies have reported an increased metabolic flux of KYNA after acute exercise. Mudry et al. observed increased plasma KYNA and decreased KYN levels in both normoglycemic men and men with type 2 diabetes after acute exercise, as well as decreased plasma TRP levels and an increased [KA] * 1000/[KYN] ratio during recovery in both groups. 41 Similarly, Joisten et al. observed the same results in the blood of healthy males after participating in acute exercise. 38 Although there are heterogeneities in the study populations, exercise modalities, measurement time points, and samples, the above studies all suggest that exercise can affect KP metabolism. The results of this study indicate that 4 weeks of voluntary wheel running exercise significantly increased KYNA levels and decreased QUIN levels in the hippocampal tissue of depressed model rats while downregulating the relative expression levels of KMO and 3-HAO mRNA and upregulating the KAT mRNA expression level. These findings confirm that exercise can alter KP metabolism abnormalities in the central nervous system of depressed model rats.
Exercise and depression
Physical activity is considered an excellent non-pharmacological strategy for preventing and treating various diseases. It is characterized by low cost, no complications, and non-invasiveness and is believed to effectively alleviate depression.42–44 Gao et al. found that women who participated in square dancing interventions for 3 consecutive months had a lower depression rate than those who did not participate. Square dancing also significantly reduced depressive symptoms in perimenopausal women. 45 Li et al. demonstrated that lower levels of physical activity were associated with more severe depressive symptoms. 46 Philippot et al. conducted physical activity interventions for adolescents hospitalized because of depression and anxiety. The results showed that physical activity significantly reduced the Hospital Anxiety Depression Scale scores in depressed adolescents. 47 Kim et al. examined the relationship between regular exercise and depressive symptoms in 5,379 Korean adults aged ≥55 years. 48 The results revealed a significant association between a lack of regular exercise and increased frequency of depressive symptoms in older adults. In addition, hand grip strength may increase the effect of regular exercise on depressive symptoms in individuals aged 65 years and older. Mitsue et al. conducted a self-rating depression scale questionnaire survey on 20 healthy young adults and assessed their movement quality using the Body Awareness Assessment Scale-Movement Quality and Experience assessment, administered by qualified physical therapists. 49 The Body Awareness Assessment Scale-Movement Quality and Experience scores were higher in the group with low scores on the self-rating depression scale questionnaire than in the high-scoring group for all items. The present study demonstrated that 4 weeks of voluntary wheel running significantly increased horizontal and vertical movements in the open field test, sucrose consumption, and the sucrose preference index and significantly decreased immobility time in the forced swimming test, consistent with previous research findings.
Voluntary wheel running exercise may improve depressive symptoms in a rat model of depression by regulating abnormal TRP-KYN metabolism
Chronic unpredictable stress combined with social isolation is currently widely recognized as a method for replicating depression models by researchers both domestically and internationally. In this experiment, after 21 days of stress, the horizontal and vertical activity in the rats decreased compared with that in the control group, indicating successful replication of the depression model in rats. Voluntary wheel running training increased horizontal and vertical activity in chronically stressed rats, demonstrating the antidepressant effects of exercise. The experiment showed that chronic stress can lead to a decrease in KYNA content and an increase in QUIN content in the hippocampal region of rats. Chronic stress also resulted in an increase in the relative expression levels of KMO and 3-HAO mRNA and a decrease in the relative expression of KAT mRNA. These findings suggest that chronic stress can over activate the neurotoxic TRP-QUIN branch of the TRP-KP while inhibiting the neuroprotective TRP-KYNA branch. This result indicates a close relationship between the depressive behavior induced by chronic stress in rats and the disruption of the central nervous system caused by disorder of the TRP-KP.
Studies have shown that KP metabolism is influenced by different biological environments. Under normal physiological conditions, the KP tends to favor the neuroprotective TRP-KYNA branch. However, when the body exhibits an immune response state, the activity of the first rate-limiting enzyme of the KP (IDO) and other metabolic enzymes (such as KMO and 3-HAO) is increased by proinflammatory cytokines such as interferon-γ, TNF-α, and prostaglandins, leading to the neurotoxic TRP-QUIN branch of the KP. 27 Furthermore, research has shown that exercise has anti-inflammatory effects on individuals of different ages or with chronic diseases such as obesity, type 2 diabetes, Parkinson’s disease, and Alzheimer’s disease. Specifically, after appropriate exercise, the expression levels of pro-inflammatory cytokines (such as IL-1, IL-2, IL-6, and TNF-α) in the peripheral and central nervous systems significantly decrease, while the expression levels of anti-inflammatory cytokines (such as IL-4 and IL-10) significantly increase. 50 The results of this study showed that in the voluntary wheel running exercise group of rats, the levels of the pro-inflammatory cytokines IL-1β, IL-6, and TNF-α significantly decreased, while the levels of the anti-inflammatory factors IL-4 and IL-10 significantly increased. The KYNA content significantly increased, while the QUIN content significantly decreased. The relative expression levels of KMO and 3-HAO mRNA decreased, while the relative expression level of KAT mRNA increased. These findings indicate that 4 weeks of voluntary wheel running exercise can alter the abnormal TRP-KYN metabolism in depressed rats, and this may also be a mechanism by which exercise intervention exerts its antidepressant effects.
Summary
Depression is closely related to KP abnormalities. Exercise may alleviate depression, may change KYN metabolism abnormalities, has no significant adverse effects on the body, and thus is safe, effective, and worth further promotion in clinical practice. However, this study did not directly detect changes in the neuronal structure and related indicators in the rat hippocampus and may not strongly show the impact of abnormal KYN metabolism on central nervous system damage in depression model rats. In future studies, relevant data are needed to further clarify the anti-depression mechanism of exercise. In addition, KYN can cross the blood-brain barrier, and peripheral factors are involved in the impact of stress on brain KYN metabolites. However, the lack of KYN measurement is a limitation of this study.
In addition, it is necessary to expand the sample size and focus on the delayed effects of exercise (1 month or longer) on depression.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241261986 - Supplemental material for The improvement effect of exercise on rats with chronic stress-induced depression-like behavior: a discussion based on the kynurenine pathway
Supplemental material, sj-pdf-1-imr-10.1177_03000605241261986 for The improvement effect of exercise on rats with chronic stress-induced depression-like behavior: a discussion based on the kynurenine pathway by Lingling Zhao, Minqiang Du and Ruihong Zhang in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to express our sincere gratitude to all individuals and institutions that contributed to this research project.
Author contributions
LLZ: writing-original draft preparation and data curation. MQD: data curation and writing-review & editing. RHZ: data collection and writing-review & editing. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was funded by Shanghai Educational Science research general project No. JJW2021729 and JJW20221160.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
