Abstract
Objective
The prognosis of lung adenocarcinoma (LUAD), which is the most common type of lung cancer, remains poor. Little is known about the function and mechanism of whey acidic protein four-disulfide core domain 2 (WFDC2) in LUAD.
Methods
In this study, we used online databases to compare WFDC2 expression between LUAD and normal tissues, to analyze the relationship between WFDC2 and overall survival, and to investigate the potential roles of WFDC2.
Results
We found that WFDC2 protein and mRNA expression levels were significantly higher in LUAD tissue than in normal tissue, and high WFDC2 mRNA expression was associated with better overall survival. WFDC2 mRNA expression was correlated with the mutation status of TP53. The biological function of WFDC2 was associated with the cell cycle, and low WFDC2 mRNA expression was associated with an elevated tumor mutational burden and neoantigen levels. A negative relationship was observed between WFDC2 and immune gene expression, and high WFDC2 mRNA expression was found in patients with LUAD and low programed cell death 1 mRNA expression.
Conclusions
We propose that WFDC2 is associated with clinical benefits of immune checkpoint inhibitors in LUAD.
Keywords
Introduction
Lung cancer, which is one of the most common malignant tumors, is associated with extremely high morbidity and mortality. 1 Non-small cell lung cancer (NSCLC) accounts for approximately 80% of all lung cancers. Despite advances in the diagnosis and treatment of lung cancer, lung adenocarcinoma (LUAD), which is the most common type of NSCLC, has a poor prognosis. 1 The recent success of cytotoxic T-lymphocyte antigen-4, programmed cell death-1 (PDCD1), and PD-ligand 1 (PD-L1) inhibitors in the treatment of NSCLC has opened a new era of immunotherapy.2,3 Tumor-infiltrating lymphocytes are considered to be a specific population of lymphocytes that are highly immunologically associated with tumor cells.
Whey acidic protein four-disulfide core domain 2 (WFDC2) is a member of the whey acidic protein domain family. WFDC2 protein, also known as human epididymis 4 (HE4), is a useful biomarker for ovarian cancer, and WFDC2 mRNA is overexpressed in LUAD.4,5 WFDC2 mRNA expression in nonsmokers is significantly higher than that in smokers, 6 HE4 protein is overexpressed in lung cancer,7,8 and higher serum WFDC2 protein levels are correlated with poor outcomes in NSCLC, especially in LUAD.9,10 Therefore, investigating the function of WFDC2 may provide new ideas for future biomarkers and therapeutic targets in LUAD. This study aimed is to assess the role of WFDC2 as a potential biomarker for immunotherapy for LUAD and its association with tumor immune infiltrates.
Materials and methods
WFDC2 mRNA expression in LUAD
The GEPIA database, 11 UALCAN database, 12 and LinkedOmics 13 were applied to evaluate WFDC2 mRNA expression in LUAD. WFDC2 mRNA expression in subgroups of LUAD stratified according to sex, race, stage, and gene mutation status was also analyzed.
Overall survival analysis
GEPIA, 11 LinkedOmics, 13 OncoLnc, 14 TISIDB, 15 and TCGA portals were used to evaluate the effect of WFDC2 mRNA expression levels on overall survival (OS) in patients with LUAD. We also used CAMOIP 16 to evaluate the effect of TP53 mutation on OS in LUAD.
Enrichment analysis
CAMOIP was used to conduct Kyoto Encyclopedia of Genes and Genomes and reactome pathway analyses. 16
Analysis of predicted tumor immunogenicity
The tumor mutation burden and neoantigen loads were analyzed using CAMOIP. 16
Immune infiltration
Immune cells and immune genes were also analyzed using CAMOIP. 16 GSCA 17 was used to detect the correlation between WFDC2 mRNA expression and immune cells in LUAD.
Immunohistochemistry
WFDC2 and PDCD1 protein expression was analyzed by immunohistochemistry. Formalin-fixed paraffin-embedded tumor specimens from 19 adult patients with LUAD were analyzed. Sections (5 μm) were cut from each specimen and stained with antibodies against WFDC2 (dilution: 1:100; ab200828; Abcam, Tokyo, Japan) and PDCD1 (dilution: 1:100; ab174838; Abcam, Cambridge, MA, USA). Immunohistochemistry staining was performed as previously reported.18,19
Statistical analyses
The Mann–Whitney U-test or Wilcoxon signed-rank test was applied to compare WFDC2 protein and mRNA expression between different groups. A Kaplan–Meier analysis with the log-rank test was used to compare survival curves. A Cox regression was used in univariate analyses. All correlation analyses incorporated Spearman’s correlation tests. The χ2 test was used to analyze correlations between WFDC2 expression and clinicopathological variables. IBM SPSS version 22 (IBM Corp., Armonk, NY, USA) was used for analyses. A p value <0.05 was considered statistically significant.
Ethics statement
The study was approved by the Institutional Review Board of The Second People’s Hospital of Huai’an. The need for written informed consent was waived owing to the retrospective nature of the research.
Results
WFDC2 protein and mRNA expression
WFDC2 mRNA expression in different types of solid tumors was analyzed using GEPIA (Figure 1(a)). WFDC2 mRNA expression levels were significantly higher in LUAD tissue than in normal lung tissue (p < 0.001; Figure 1(b, c)). WFDC2 protein levels were also observed in LUAD tissue than in normal lung tissue (p < 0.001; Figure 1(d)). No difference in WFDC2 expression in patients with LUAD was observed in the subgroup analysis based on sex and race (Figure 1(e–f)), except for the stage subgroup (p = 0.0023; Figure 1(g)). WFDC2 mRNA expression was correlated with the mutation status of TP53, epidermal growth factor receptor (EGFR), and serine/threonine kinase 11 (STK11) (all p < 0.05, Figure 1(h–k)). The TP53 mutation was common in patients with low WFDC2 mRNA expression (Figure 2).

(a) WFDC2 mRNA expression in different cancers (GEPIA). (b) WFDC2 mRNA expression in LUAD (GEPIA). (c) WFDC2 mRNA expression in LUAD (UALCAN, p = 1.62E-12). (d) WFDC2 protein expression in LUAD (UALCAN, p = 8.32E-12). (e) WFDC2 mRNA expression in LUAD based on sex (UALCAN, p = 0.316). (f) WFDC2 mRNA expression in LUAD based on race (UALCAN, p = 0.339). (g) WFDC2 mRNA expression in LUAD based on stage (GEPIA, p = 0.0023). (h) WFDC2 mRNA expression in LUAD based on the TP53 mutation status (LinkedOmics, p = 1.275e-05). (i) WFDC2 mRNA expression in LUAD based on the EGFR mutation status (LinkedOmics, p = 8.841e-03). (j) WFDC2 mRNA expression in LUAD based on the STK11 mutation status (LinkedOmics, p = 1.271e-02) and (k) gene expression between driver-mutated samples (red) and not mutated (gray) samples (TCGA portal). WFDC2, whey acidic protein four-disulfide core domain 2; LUAD, lung adenocarcinoma; WT, wild type; MUT, mutant; EGFR, epidermal growth factor receptor; STK11, serine/threonine kinase 11.

Mutation landscape according to WFDC2 expression. WFDC2, whey acidic protein four-disulfide core domain 2; TSG, tumor suppressor gene.
WFDC2 mRNA levels predict OS
The Kaplan–Meier analysis showed that the OS time in patients with LUAD in the high WFDC2 mRNA expression group was significantly longer than that in the low WFDC2 mRNA expression group (all p < 0.05, Figure 3(a–e)). These results suggested that WFDC2 could serve as a prognostic biomarker in patients with LUAD.

Overall survival curves of WFDC2 in LUAD from different databases. (a) GEPIA, (b) LinkedOmics, (c) OncoLnc., and (d) TISIDB. WFDC2, whey acidic protein four-disulfide core domain 2; LUAD, lung adenocarcinoma; TPM, transcripts per million; HR, hazard ratio; CI, confidence interval.
TP53 mutation predicts the anti-PDCD1 response
A retrospective analysis based on TCGA data showed that OS was similar between TP53 mutant and TP53 wild-type LUAD (Figure 4(a, b)). Figure 4(c) shows that progression-free survival was significantly longer in patients with the TP53-mutant (TP53-MUT) receiving anti-PDCD1 therapy than in those with the TP53 wild type (p = 0.017). Univariable Cox regression showed the same results as those in Figure 4(c) (p < 0.05, Figure 4(d)).

(a) OS curve of TP53 mutation in LUAD. (b) Univariable Cox regression analysis of OS in LUAD. (c) PFS curve of TP53 mutation in patients with LUAD who received anti-programmed cell death-1 therapy and (d) Univariable Cox regression analysis of PFS in patients with LUAD who received anti-programmed cell death-1 therapy. LUAD, lung adenocarcinoma; MT, mutant; WT, wild type; CI, confidence interval; HR, hazard ratio; OS, overall survival; PFS, progression-free survival.
Enrichment analysis
WFDC2 and TP53 mutations had the same enrichment in the cell cycle (Figure 5(a–d)), especially in reactome pathway analysis. The main Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis showed that WFDC2 was involved in spliceosomes, RNA transport, ribosome biogenesis in eukaryotes, the cell cycle, chemical carcinogenesis, the mRNA surveillance pathway, ubiquitin-mediated proteolysis, peroxisomes, human T cell leukemia virus 1 infection, and regulation of the actin cytoskeleton.

(a, b) Enrichment analysis according to WFDC2 expression and (c, d) the TP53 mutation status.
Tumor immunogenicity
The tumor mutational burden level was significantly higher in TP53-MUT tumors than in wild-type TP53 tumors (p < 0.05, Figure 6(a)). Accordingly, the neoantigen loads were also significantly higher in TP53-MUT tumors than in wild-type TP53 tumors (p < 0.05, Figure 6(b)), which indicated that TP53-MUT was associated with enhanced tumor immunogenicity. Because of the correlation between WFDC2 mRNA expression and TP53 mutation, enhanced tumor immunogenicity was observed in low WFDC2-expressing LUAD tumors (Figure 6(c, d)).

(a, b) Tumor immunogenicity analysis according to WFDC2 expression and (c, d) the TP53 mutation status. ****p < 0001. WFDC2, whey acidic protein four-disulfide core domain 2; LUAD, lung adenocarcinoma; TMB, tumor mutational burden.
Immune cell infiltration
We found an association between WFDC2 mRNA expression and CD4 T cell infiltration (p < 0.05, Figure 7(a)). Accordingly, the same result was observed in TP53-MUT tumors (p < 0.05, Figure 7(b)). Strong infiltration of CD4+ T cells was observed in low WFDC2-expressing LUAD tumors (Figure 7(c, d)).

(a) Immune cell infiltration according to WFDC2 expression and (b) the TP53 mutation status. (c, d) Association between WFDC2 expression and CD4+ T cell infiltration. *p < 0.05, **p < 0.015, ***p < 0.001, ****p < 0.0001. MT, mutant; WT, wild type; ns, not significant; LUAD, lung adenocarcinoma; WFDC2, whey acidic protein four-disulfide core domain 2.
Treg (r = 0.12, p < 0.001) and Th17 (r = 0.23, p = 0.0001) cells were positively related to the expression of WFDC2 (data from GSCA).
Immune genes
The expression of CD274, PDCD1, and CTLA4 was higher in the TP53-MUT group (Figure 8(a)). Accordingly, the same result was observed in the low WFDC2 expression group (Figure 8(b)).
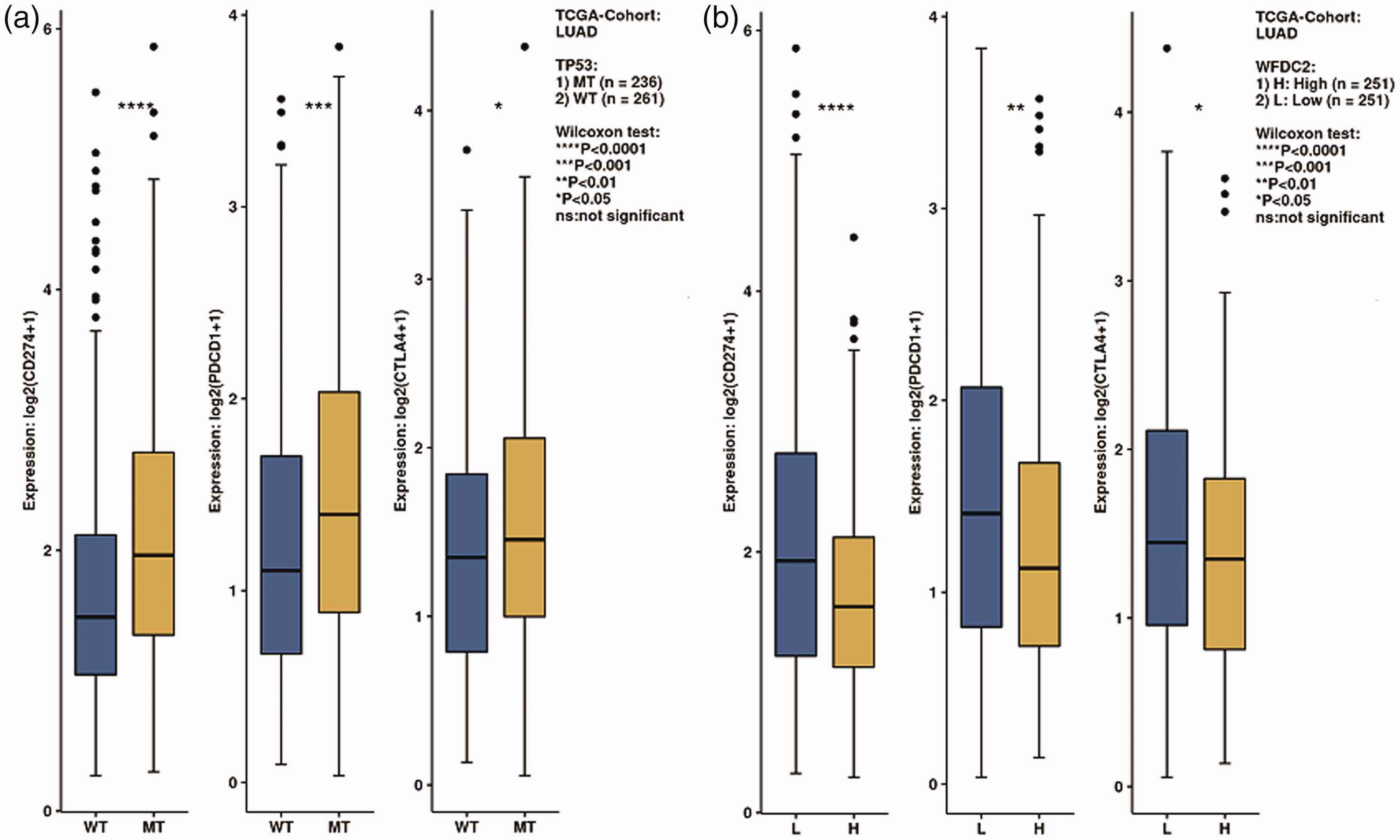
(a) Immune gene expression according to WFDC2 expression and (b) the TP53 mutation status.
Immunohistochemistry
We examined samples from 12 (63.2%) men and 7 (36.8%) women, with a mean age of 61.0 years old (range: 46–72 years old). WFDC2 protein was highly expressed in 9 of 19 (47.4%) samples (Figure 9(a–b)). A total of 52.6% of tissues exhibited high PDCD1 immunostaining (Figure 9(c–d)). There was a significant trend toward higher WFDC2 expression in patients with low PDCD1 expression (p = 0.01).

Immunohistochemistry of WFDC2 and PDCD1 protein expression in LUAD. WFDC2, whey acidic protein four-disulfide core domain 2; PDCD1, programed cell death 1.
Discussion
Despite advances in lung cancer treatment, it remains one of the most common forms of cancer and a leading cause of cancer-related death. Adenocarcinoma, which is the most common histological type of lung cancer, comprises 40% of all lung cancer cases. LUAD is often diagnosed at an advanced stage involving disseminated metastatic tumors, with an approximate 15% 5-year survival rate. Treatment of LUAD remains a challenge for clinicians.
The WFDC2 gene encodes a secreted glycosylated protein called HE4, which belongs to the WFDC (previously named whey acidic protein) family and is located on human chromosome 20. 20 WFDC2 protein was first found in distal epididymal epithelial cells and was originally predicted to inhibit proteases. 21 HE4 is overexpressed in several cancers.22,23 Our study showed that WFDC2 mRNA levels were higher in LUAD tissue than in normal tissue. Significantly elevated serum HE4 concentrations have been detected in patients with LUAD. 24 Low expression of WFDC2 mRNA is associated with poor OS. However, serum HE4 levels are a prognostic biomarker in lung cancer. 25 Despite the abundance of studies on WFDC2 in lung cancer, little is known regarding the function of WFDC2, specifically its role in the malignant biological behaviors of LUAD. We found that many immune cells in the microenvironment were associated with WFDC2 mRNA expression. Treg and Th17 cells were positively related to the expression of WFDC2. The main Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis showed that WFDC2 was involved in spliceosomes, RNA transport, ribosome biogenesis in eukaryotes, the cell cycle, chemical carcinogenesis, the mRNA surveillance pathway, ubiquitin-mediated proteolysis, peroxisomes, human T cell leukemia virus 1 infection, and regulation of the actin cytoskeleton.
In recent years, several molecular alterations that affect the treatment and prognosis of NSCLC have been identified, such as EGFR, KRAS, TP53 and STK11.26–28 LUAD is classified into three tumor immune profiles, and TP53, EGFR, and STK11 mutations are the major determinants of the tumor immune microenvironment for immune checkpoint inhibition. 29 Our study showed that WFDC2 mRNA expression was associated with the TP53, EGFR, and STK11 mutation status. A negative relationship was also observed between WFDC2 and immune genes. TP53 mutation is the most common mutation in patients with low WFDC2 expression. Low expression of WFDC2 was found in TP53 mutation in LUAD in our analysis, while TP53-MUT tumors enriched in Tax-interacting protein 1 have been previously found to be characterized by PD-L1 expression. 29 Our study showed high WFDC2 expression in EGFR- or STK11-mutated tumors. STK11 or EGFR mutations are associated with lower expression of PD-L1. 29 STK11, EGFR, and TP53 mutations are the major determinants of the expression of PD-L1 in LUAD. 29 The expression of WFDC2 may be a robust parameter to identify potential responders to immune checkpoint inhibitors.
The role of WFDC2 in LUAD should be interpreted with caution because of several limitations to our study. First, only TCGA cohort data were included in this study. Second, the main ethnic population in this meta-analysis was Caucasian. Third, our study was limited by a lack of access to data on treatment regimens. Fourth, TP53 mutations were not validated in patients with low WFDC2 expression. Fifth, protein expression of WFDC2 and PDCD1 was analyzed in a small sample size (19 adult patients). Finally, the cutoff values of WFDC2 expression were different among some databases, which showed different results. WFDC2 expression should be defined as high or low based on a uniform standard. Therefore, further studies are required to confirm our findings.
In conclusion, by determining the potential effects of WFDC2 in lung adenocarcinoma, this study provides evidence that WFDC2 expression is associated with the TP53 mutation status and immune gene expression, and WFDC2 is also a prognostic biomarker. Therefore, WFDC2 could be a robust biomarker to identify potential responders to immune checkpoint inhibitors.
Footnotes
Author contributions
All authors made a considerable contribution to the work, including the conception of the study, study design, execution, acquisition of the data, analysis, and interpretation. All authors participating in drafting, revision or critical review of the manuscript. All authors gave final approval of the version to be published, agreed on the journal to which the article has been submitted, and agree to be accountable for all aspects of the work.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the online databases.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sector.
