Abstract
Stylocarotid artery syndrome (SAS) is a rare variant of Eagle’s syndrome that may lead to transient ischemic attack or stroke. The underlying pathophysiological mechanism involves compression of the internal carotid artery by an elongated styloid process (ESP), potentially resulting in vascular occlusion or dissection. An ESP exceeding 2.5 cm is deemed elongated, with a length of 3.0 cm considered clinically significant. Although the prevalence of ESP ranges from 4.0% to 7.3%, symptomatic cases are rare; symptoms are present in only approximately 4.0% of individuals with an ESP. Unlike the typical symptoms of Eagle’s syndrome, SAS may not cause pharyngeal discomfort, the sensation of a foreign body in the throat, dysphagia, or facial pain. This absence of characteristic symptoms as well as the development of central nervous system symptoms often leads patients to seek care from neurologists instead of otolaryngologists, increasing the likelihood of misdiagnosis or underdiagnosis. We herein report a unique case of ischemic stroke caused by SAS and present a literature review on cases of SAS-associated ischemic stroke published in the past decade. The reporting of this study conforms to the CARE guidelines.
Keywords
Background
Stylocarotid artery syndrome (SAS) is a rare variant of Eagle’s syndrome (ES) that may lead to transient ischemic attack (TIA) or stroke. The underlying pathophysiological mechanism involves compression of the internal carotid artery (ICA) by an elongated styloid process (ESP), potentially causing vascular occlusion or dissection. An ESP exceeding 2.5 cm is deemed elongated, with a length of 3.0 cm considered clinically significant. 1 Although the prevalence of ESP ranges from 4.0% to 7.3%, 1 symptoms are rare, presenting in only approximately 4.0% of individuals with an ESP. 2 Unlike the typical symptoms of ES, SAS may not cause pharyngeal discomfort, the sensation of a foreign body in the throat, dysphagia, or facial pain. This absence of characteristic symptoms coupled with the development of central nervous system symptoms often leads patients to seek care from neurologists instead of otolaryngologists, increasing the likelihood of misdiagnosis or underdiagnosis. We herein report a unique case of ischemic stroke caused by SAS and present by a literature review on reports of SAS-associated ischemic stroke published in the past decade. The reporting of this study conforms to the CARE guidelines. 3
Case presentation
A man in his early 30s, previously healthy and an avid badminton player, presented to the neurology department with recurrent episodes of speech indistinctness of approximately 1 hour in duration. He had experienced two transient aphasia episodes triggered by head flexion. Upon hospitalization, the aphasia recurred along with the development of transient memory loss. Clinical examination showed stable vital signs, clear consciousness, slurred speech, diminished responsiveness, memory impairment (including an inability to recall his father’s name), normal limb strength, absence of extrapyramidal signs, and no pathological reflexes. His National Institutes of Health Stroke Scale (NIHSS) score was 3. An initial head computed tomography (CT) scan revealed no hemorrhage or low-density areas. After obtaining written informed consent from the patient, we initiated intravenous thrombolysis using alteplase. This treatment led to rapid improvement in his language skills, although his responsiveness remained somewhat blunted. His limb strength remained normal. Subsequent head magnetic resonance imaging revealed multiple acute infarcts in the left frontal lobe, insula, periventricular region, and centrum semiovale. Neck CT angiography showed bilateral ESPs and significant stenosis at the origin of the left ICA. Considering the patient’s clinical history, we suspected SAS. Throat examination was unremarkable, showing no mucosal congestion or palpable masses in the tonsillar fossae. Neck magnetic resonance imaging and digital subtraction angiography revealed a dissection in the proximal segment of the left ICA at the level of C1. The dissection was approximately 27 mm in length and was causing notable luminal narrowing. Because he was concerned about the risks of styloidectomy, the patient opted for a regimen of clopidogrel, aspirin, and atorvastatin instead of surgery. Four months post-discharge, the patient demonstrated excellent recovery with an NIHSS score of 0 and no further ischemic episodes. The patient’s imaging findings are presented in Figures 1 to 4.
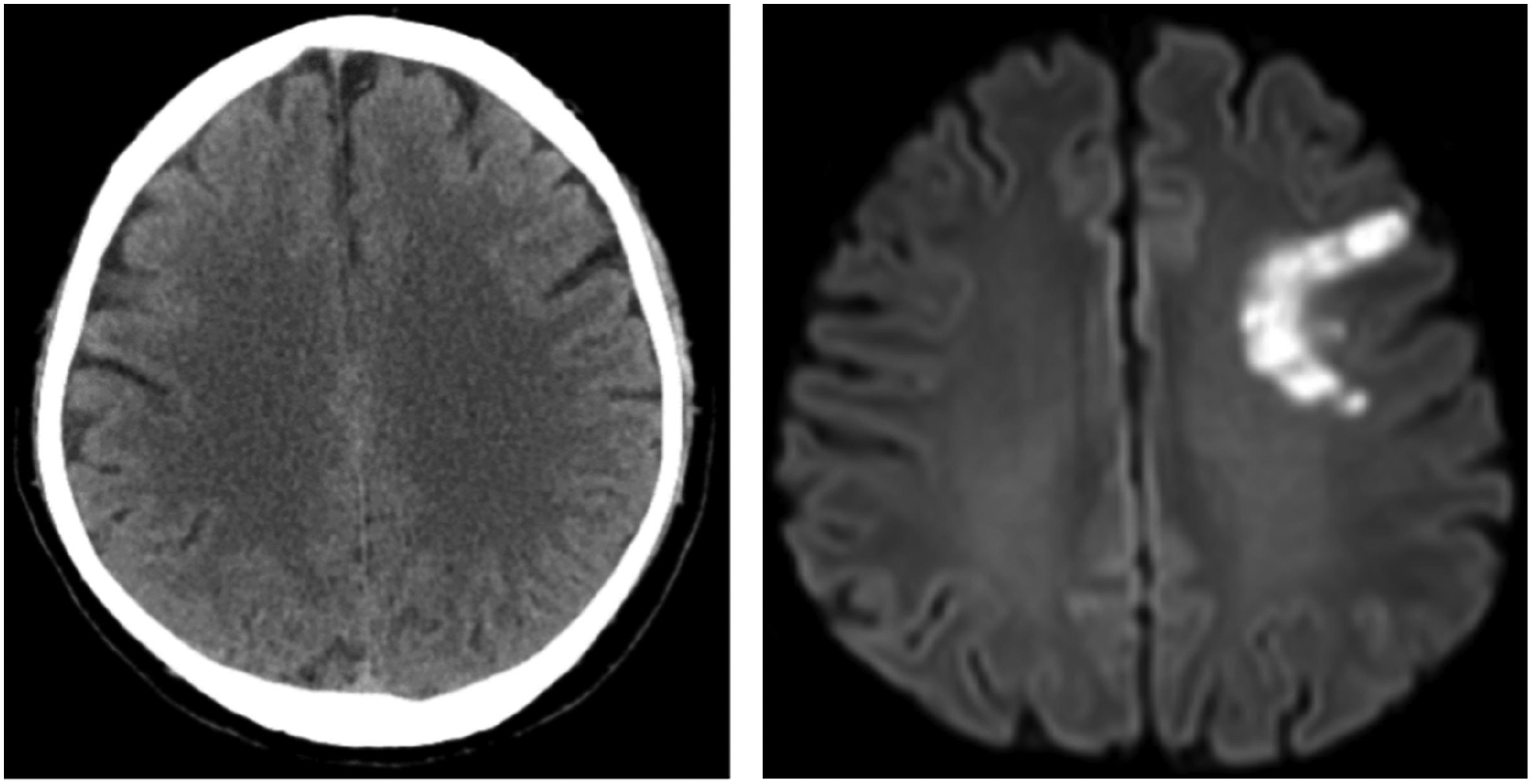
Left panel: computed tomography scan taken before intravenous thrombolysis showed no lesions. Right panel: diffusion-weighted magnetic resonance imaging taken 1 day after symptom onset revealed acute infarcts in the left frontal lobe and centrum semiovale.
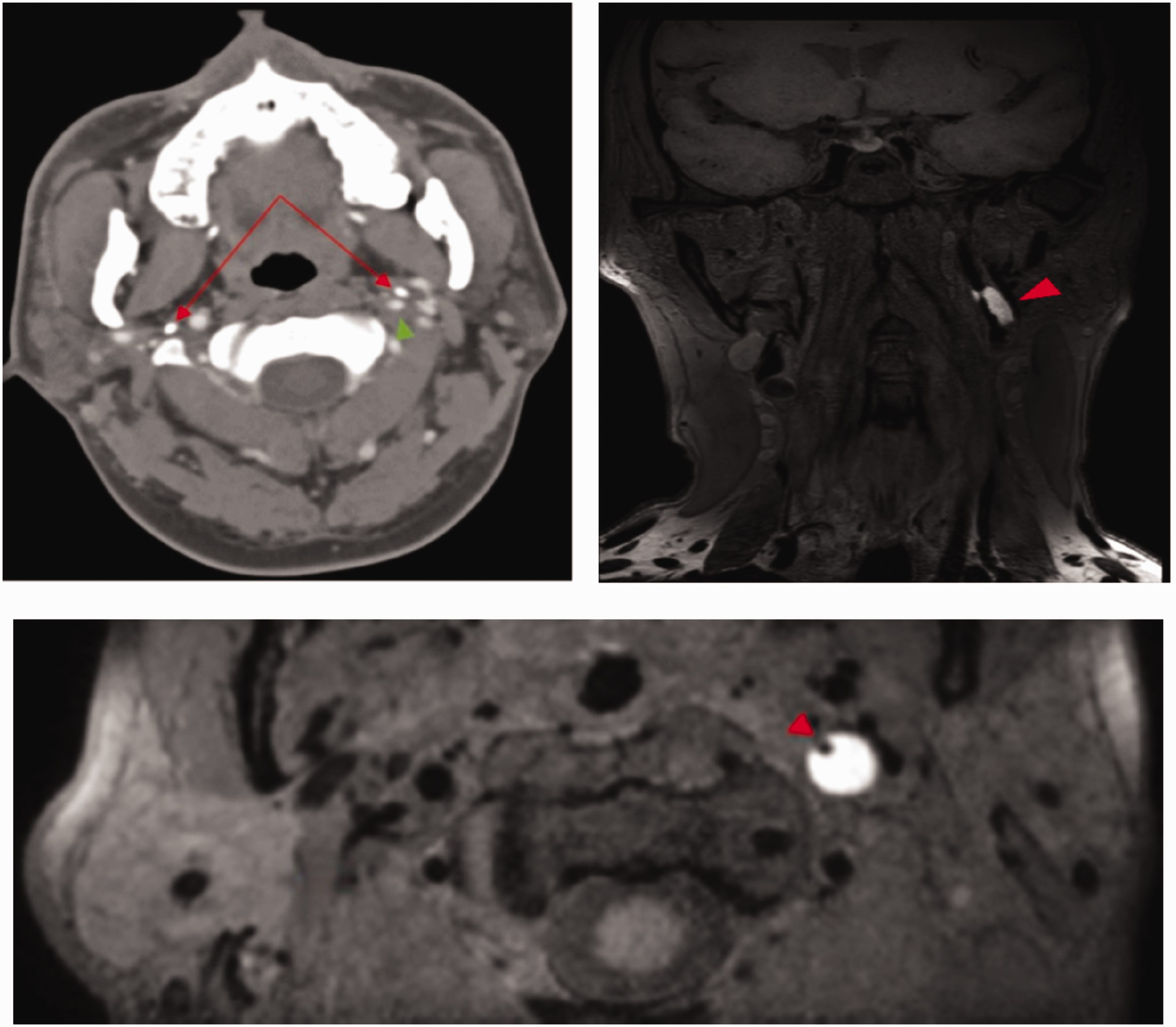
The red arrow indicates the styloid process, which is abnormally adjacent to the left internal carotid artery, as shown by the green triangle in the left-hand image. The red triangle marks a dissection in the left internal carotid artery, which is associated with severe stenosis, as evident in the images to the right and beneath.
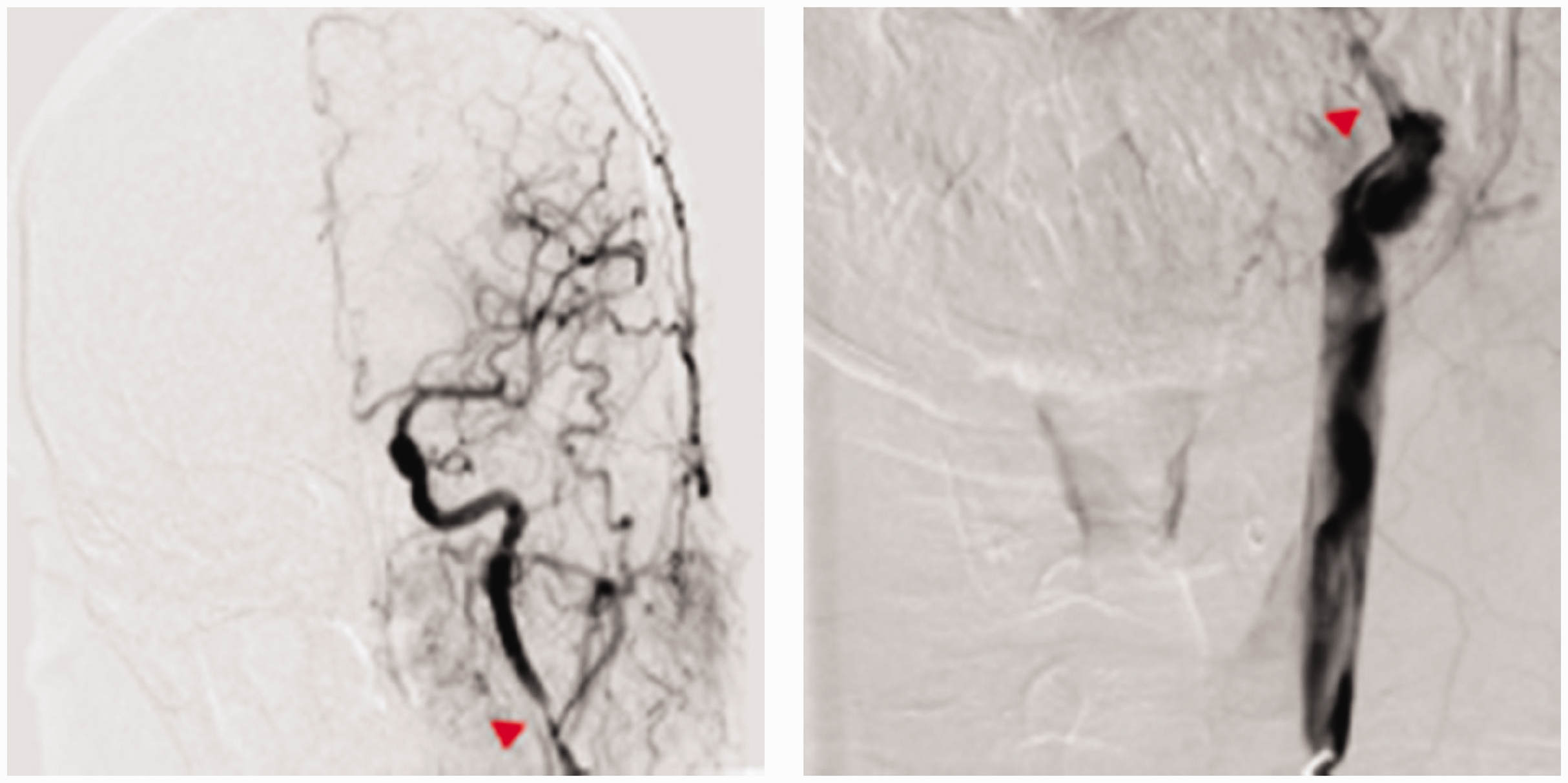
Digital subtraction angiography revealed a dissection in the left internal carotid artery. The red triangle highlights the area of severe stenosis.

Three-dimensional computed tomography reconstruction revealed medial deviation of the left styloid process at 19.93° and the right at 35.27°, as shown in the left panel. The left styloid process measures 57.4 mm in length, with an anterior inclination of 31.03°, depicted in the right panel.
Review of ischemic stroke associated with SAS
A PubMed search using the terms ((ischemic stroke) OR (cerebral infarction)) AND ((Eagle syndrome) OR (stylocarotid artery syndrome)) yielded 23 cases of ischemic stroke attributable to SAS from 2014 to 2024. The patients’ mean age at onset was 48.45 ± 2.88 years (range, 26–85 years). The comprised 19 (82.6%) men and 4 (17.4%) women. The presenting symptoms were hemiplegia (n = 20), aphasia (n = 17), sensory deficits (n = 6), facial paralysis (n = 5), and headache (n = 3). Less common symptoms included consciousness disturbances, neck pain, and visual impairments (n = 2 each) as well as neglect, tinnitus, and palpitations (n = 1 each). The mean NIHSS score was 10.82 ± 2.75 among 11 patients. An ESP was noted on the left side in 5 (21.7%) patients, on the right side in 1 (4.3%), and bilaterally in 17 (73.9%). The mean lengths of the left and right styloid processes were 4.24 ± 0.99 cm (n = 16) and 4.29 ± 1.31 cm (n = 15), respectively. Infarctions occurred most frequently in the middle cerebral artery territory (n = 6), followed by the frontal and parietal lobes (n = 5 each). They occurred less frequently in the temporal lobes, insula, and periventricular regions (n = 3 each), with basal ganglia infarctions occurring in two patients. Carotid artery dissections were observed on the left side in eight (34.8%) patients, on the right side in three (13.0%), and bilaterally in six (26.1%). Styloidectomy was performed in nine (39.1%) patients, with no further cerebrovascular incidents reported thereafter (Table 1).
Review of ischemic stroke associated with stylocarotid syndrome (2014–2024).
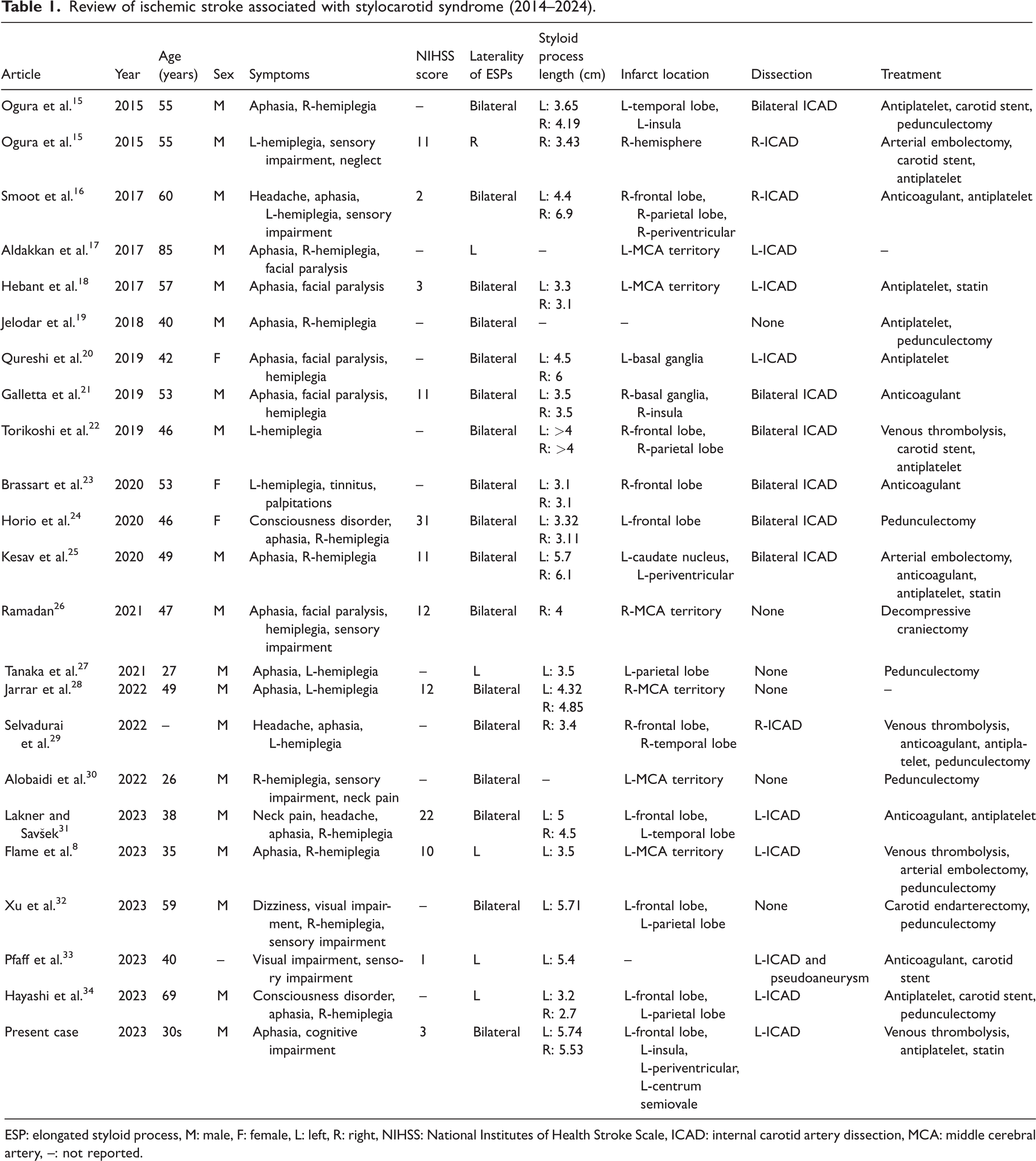
ESP: elongated styloid process, M: male, F: female, L: left, R: right, NIHSS: National Institutes of Health Stroke Scale, ICAD: internal carotid artery dissection, MCA: middle cerebral artery, –: not reported.
Discussion
The styloid process, a slender and cylindrical projection, forms part of the temporal bone. It originates at the junction of the petrous base and the mastoid process of the temporal bone, extending anteroinferiorly. Positioned between the internal and external carotid arteries, the styloid process typically measures approximately 2.5 cm in length. Nearby structures include the internal jugular vein and cranial nerves V, IX, X, and XII.
ES encompasses a range of clinical manifestations attributed to an abnormally long or malpositioned styloid process, initially characterized by Eagle 4 in 1937. A styloid process exceeding 3 cm is indicative of an ESP. However, not every case involving an ESP progresses to ES because the angulation of the process and the adjacent anatomical features also affect the outcome. 5 The precise correlation between the angulation of the styloid process—both anterior inclination and medial inclination—and the development of ES remains undefined. The standard inclination of an adult’s styloid process deviates from the vertical by approximately 25° anteriorly and medially. Angles beyond 40° or below 20° are deemed atypical. 6
ES is typically divided into two forms: the classical type and SAS. 7 The former is mainly characterized by compressive neurological symptoms such as pharyngeal pain, dysphagia, neck pain upon rotation, facial pain, and earache, often initially identified in otolaryngology settings. SAS, a rarer condition with an estimated incidence of 4 to 8 per 100,000 individuals, 8 is frequently linked to neck massage, extreme head and neck movements, or mechanical injuries that can lead to carotid artery damage or dissection, potentially causing TIA or ischemic stroke.
Flame et al. 8 reported a case of SAS involving dissection and occlusion of the left ICA, resulting in two ischemic stroke episodes affecting the left middle cerebral artery territory within a span of 3 months. The patient experienced no further ischemic events after surgical removal of the ESP. David et al. 9 documented a case of SAS in a patient presenting with recurrent right-sided limb weakness and syncope triggered by turning the head right or yawning. Shi et al. 10 described an incident in which prolonged head shaking due to ecstasy consumption led to bilateral ICA dissections from mechanical stress caused by an ESP.
In contrast to these cases, our patient developed symptoms following a badminton session and a neck massage. This sequence of events led to multiple TIA episodes within a short period. We hypothesize that excessive neck movement resulted in compression of the C1 segment of the ICA by the left styloid process. The subsequent neck massage likely exacerbated this compression, leading to carotid dissection, significant stenosis, and ultimately an ischemic stroke.
Three-dimensional CT is recognized as the definitive standard for the diagnosis of ES. Reconstructed three-dimensional CT imaging reveals the morphology of the styloid process and its spatial relationship with adjacent structures, enabling accurate measurements of its length and angulations. Additionally, CT angiography is instrumental not only in directly visualizing carotid artery stenosis and dissection but also in delineating the spatial correlation between the styloid process and the vasculature. 11 A three-dimensional CT scan is highly advised in patients with suspected ES.
Styloidectomy, a definitive therapeutic intervention for ES, encompasses two primary surgical techniques: the intraoral and extraoral approaches. 12 Postoperative outcomes typically include significant symptomatic alleviation for the majority of patients. For those who decline surgical intervention, pharmacological management with agents such as nonsteroidal anti-inflammatory drugs, pregabalin, carbamazepine, and amitriptyline, among others, may be prescribed.13,14 Importantly, however, conservative medical treatments may be associated with a heightened risk of symptom recurrence.
Given its symptomatic variability, ES is frequently undiagnosed in clinical settings. Patients who develop symptoms after extensive head and neck motion, notably those with carotid ischemic presentations, should undergo detailed physical evaluations alongside comprehensive cervical CT imaging. Current literature on ischemic stroke attributed to SAS suggests a higher prevalence in men than in women, with left ICA dissections surpassing those on the right. A deeper investigation of a larger cohort is warranted to discern potential patterns in these findings.
Footnotes
Acknowledgment
The authors thank the patient for allowing the publication of his case details.
Author contributions
YW conceived the study; BL wrote the manuscript; QL and YL collected the data; and JC, HJ, and YZ performed the digital subtraction angiography procedure.
Data availability statement
The patient provided verbal consent for publication, and his personal details were de-identified for privacy. The data from this study are accessible and will be provided publicly upon request. For access to the raw data, please contact the corresponding author at
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
This study did not involve human or animal subjects; therefore, ethical approval was not required. The study protocol adhered to the CARE guidelines.
Funding
This study was funded by the Specialty Feature Construction Project of Health Commission of Pudong New District, Shanghai (PWZzb2022-18).
