Abstract
Spontaneous internal carotid artery dissection (ICAD) is a rare disease and an important cause of stroke in young patients. The presentation of ICAD is variable and atypical. We have used three-dimensional T1-weighted volume isotropic turbo spin-echo acquisition to help diagnose ICAD. ICAD should be considered in young patients presenting with relevant symptoms in an emergency setting. We herein report ICAD with an unexplained onset in two patients. Neither had a history of large or small cervical trauma, but both had a history of hypertension. The first patient was a 33-year-old man who presented with upper extremity numbness and mobility impairment, and the second patient was a 40-year-old man with onset of visual impairment. There were no obvious triggers in either case. Both patients were diagnosed with ICAD by blood vessel examination. However, their prognoses differed. The first patient recovered after endovascular therapy, whereas the second patient was successfully managed with medical treatment. ICAD can cause different symptoms depending on where the dissection occurs. Clinicians must be aware of the different clinical manifestations of ICAD, make the correct diagnosis in a timely manner, make appropriate treatment plans according to the patient’s condition, and strive for timely recanalization of the blood vessels.
Keywords
Introduction
Spontaneous internal carotid artery dissection (ICAD) is a rare disease with an incidence rate of about 2.6 to 2.9 per 100,000 individuals. 1 However, it accounts for up to 5% to 22% of strokes in patients under the age of 45 years. 2 With the rapid development of vascular imaging technology, the detection rate of ICAD has gradually increased. Three-dimensional T1-weighted volume isotropic turbo spin-echo acquisition (3D T1-VISTA) magnetic resonance imaging (MRI) provides a high signal-to-noise ratio and can provide information on vascular plaques and vascular walls that cannot be evaluated by traditional vascular imaging technologies.
The presentation of ICAD is variable. Approximately 50% to 59% of patients experience symptoms of cerebral or retinal ischemia (only 3% of patients have amaurosis, while 3% have ischemic optic neuropathy [ION]), 44% to 69% of patients report headache, up to 50% develop Horner syndrome, and about 12% develop cranial nerve palsy. However, 50% of patients have an atypical presentation, and approximately 5% are asymptomatic.2–4
At present, the treatment of ICAD is based on the treatment of related clinical symptoms. Thrombolysis, anticoagulation, and antiplatelet therapy should be considered for the prevention and treatment of stroke. However, no randomized study has been performed to evaluate the efficacy and safety of endovascular interventional therapy or surgery in the treatment of ICAD. Because of the lack of evidence-based medicine, treatment of ICAD is based on clinical experience and chosen at the physician’s discretion.
Case presentations
The reporting of this study conforms to the CARE guidelines. 5
Case 1
A 33-year-old man was admitted to the hospital with a 4-hour history of numbness and dyskinesia in the left upper limb without an obvious trigger. The patient had a 1-year history of hypertension, and his blood pressure was 129/88 mmHg on admission. Physical examination revealed hypoalgesia on the left side of the body along with left hemiplegia (muscle strength: grade 0 in the left upper extremity and grade 3 in the left lower extremity). The patient had positive pathological signs on the left side, dysarthria, and a shallow left nasolabial fold.
After admission, digital subtraction angiography (DSA) revealed a massive intramural hematoma in the C1 segment of the right internal carotid artery (ICA) (Figure 1(a)). The patient’s blood vessels were recanalized, and his symptoms improved significantly after catheter thrombus aspiration and stent thrombectomy; however, the right ICA vessel wall was still not smooth (Figure 1(b)). Four days after endovascular therapy (EVT), 3D T1-VISTA MRI sequences showed a hyperintensity (indicating an intramural hematoma) in the vessel wall of the right ICA (Figure 1(c)).

Comparison of Case 1 before and after treatment. (a) Digital subtraction angiography shows a massive mural thrombus in the C1 segment of the internal carotid artery (circle). (b) After treatment, the vessel wall of the internal carotid artery is still not smooth (circle) and (c) Three-dimensional T1-weighted volume isotropic turbo spin-echo acquisition shows the intramural hematoma in the right internal carotid artery (arrow).
One month after antiplatelet therapy (oral aspirin at 100 mg once daily and oral clopidogrel at 75 mg twice daily), the patient recovered with only mild persistent facial paralysis.
Case 2
A 40-year-old man developed transient amaurosis and intermittent mild headache of unknown cause and achieved no relief after taking anti-cold medicine. These symptoms occurred intermittently for 2 weeks, followed by worsening before admission to the hospital. Upon admission, the patient had only mild light perception in the central field of the left eye and complete loss of vision in the peripheral field (described as a “tubular visual field”). Two hours after admission, he experienced transient complete vision loss in the affected eye. The patient had a history of hypertension, and his blood pressure was 141/81 mmHg on admission; however, he denied a history of diabetes and alcohol or tobacco consumption.
DSA showed that the end of the left common carotid artery and almost the entire length of the ICA were occluded (Figure 2(a)). The right ICA and basilar artery were supplying blood to the left anterior circulation through the anterior and posterior communicating arteries (Figure 2(b)). 3D T1-VISTA showed arterial narrowing and occlusion from the end of the left common carotid artery to the intracranial segment of the ICA (C1–C6) (Figure 2(c), (d)). After 2 weeks of conservative treatment (oral aspirin at 100 mg once daily and oral atorvastatin calcium tablets at 20 mg once daily), the patient’s clinical symptoms stabilized without further aggravation. The first day after admission, MRI showed ischemic stroke in the left frontal and temporal lobes (Figure 3(a)). Three months later, the thickened arterial wall became thinner and the degree of lumen stenosis was reduced (Figure 3(b)). Eight months later, the hematoma was stabilized and the artery demonstrated mild stenosis (Figure 3(c)).

Preoperative imaging findings in Case 2. (a, b) On digital subtraction angiography, the internal and external carotid arteries are not visible; however, the anterior and middle cerebral arteries are visible and (c, d) Sagittal oblique and coronal sections on three-dimensional T1-weighted volume isotropic turbo spin-echo acquisition show a hyperintense and continuous hematoma.

Comparison of Case 2 before and after treatment. (a) On fluid-attenuated inversion recovery imaging, the left frontal and temporal cerebral lobes show hyperintensity and (b, c) On computed tomography angiography 3 and 8 months after treatment, the artery demonstrated slight stenosis (circle).
Discussion and conclusion
ICAD is generally considered to originate from a tear in the intima of the artery. Because of the arterial blood pressure, blood enters the subintima to form a hematoma between the vessel walls. The lumen of the vessel gradually narrows with the persistent increase in the size of the hematoma and formation of a thrombus at the lesion site.
In previous reports, the typical presentation of carotid dissection was pain on one side of the head, face, or neck and partial Horner syndrome (sympathetic ophthalmoplegia), followed by cerebral or retinal ischemia hours or days later. However, this classic triad is found in less than one-third of affected patients, and only approximately one-fifth of patients develop ischemic stroke with no warning signs.
2
Neither of our patients had the typical clinical symptom of pain. In Case 1, the symptoms were mainly those of ischemic stroke, while in Case 2, the symptoms were mainly decreased vision with intermittent mild headache. Therefore, the diagnosis of arterial dissection was difficult in both patients.
DSA is considered the gold standard diagnostic technique for arterial dissection. However, because it is an invasive procedure, DSA is hardly the first choice for making this diagnosis in the emergency setting. Although the changes in the lumen and blood flow can be adequately displayed using classic vascular imaging techniques such as DSA, computed tomography angiography, and magnetic resonance angiography, the pathological changes in the vessel wall cannot be fully displayed. Therefore, we used an unconventional examination, namely 3D T1-VISTA, in both of our patients. This examination can make up for the shortcomings of the traditional vascular imaging methods that cannot show the vessel wall, and it can reveal the existence of a vascular wall hematoma much earlier, even when the vascular lumen has not significantly narrowed. Thus, 3D T1-VISTA is helpful for the achievement of a timely diagnosis.
In Case 1, the patient mainly presented with ischemic stroke, and emergency computed tomography showed cerebral infarction. However, we also needed to exclude diseases that can cause hemiplegia, such as hemorrhagic stroke, hypoglycemia, and nervous system tumors (Figure 4(a)). In Case 2, the patient developed ION when the blood supply to the ophthalmic artery was blocked (Figure 4(b)), and ICAD is one of the rare causes of ION. In a study by Biousse et al., 7 only 4 (3.6%) of 110 patients with ICAD showed ION; however, the appearance of ION may indicate a serious condition and poor prognosis. ION must be differentiated from optic neuritis. In Case 2 of the present report, the possibility of optic neuritis was excluded because of the normal appearance in optic nerve imaging and because the patient had no exercise- or heat-related vision deterioration (Uhthoff’s phenomenon). 8 In addition, acute monocular vision impairment can occur in patients with retinal migraine, which is an extremely rare form of migraine described in only isolated case reports. 9 Our patient had no migraine history, and normal sight was not restored after admission. Accordingly, the diagnosis of retinal migraine was also ruled out.

Imaging details in the two cases. (a) Emergency computed tomography shows cerebral infarction on the right side and (b) Magnetic resonance angiography shows that the right ophthalmic artery is visible (circle), but the left is not.
In view of the lack of evidence-based guidelines, the clinical treatment methods are based on the treatment principles of related diseases. Because of the limited extent of dissection lesions, the natural history of most cases of ICAD is self-healing within 3 to 6 months, during which time remodeling and recanalization of the artery are completed. 10 Antiplatelet and anticoagulation therapies are the first choice for ICAD with the aim of preventing local thrombus and embolic complications. 11 However, some scholars have pointed out that the primary treatment for dissection of proximal intracranial arteries is EVT, especially for patients with persistent ischemic symptoms. 12 In Case 1, the ICA occlusion resulted from secondary thrombus formation of the luminal stenosis secondary to the dissection. Because the lesion site was in the main ICA and the thrombus was limited, the patient received EVT in a timely manner. After receiving subsequent antiplatelet therapy, he gradually recovered.
Anatomically, the ophthalmic artery usually originates from the C6 segment of the ICA 9 and provides a blood supply to the optic nerve and retina. The intramural hematoma extending from the end of the common carotid artery to the ophthalmic artery explains the loss of left eyesight in Case 2 (Figure 5). To the best of our knowledge, such an extensive lesion (intramural hematoma) is unprecedented in previous case reports. This patient exhibited symptoms of optic nerve ischemia, and the lesion extended to the ophthalmic artery, suggesting a poor prognosis. 7 Early aggressive surgical treatment is particularly important for revascularization and prevention of complications. Unfortunately, some patients choose conservative treatment to avoid the risks of a difficult surgery.
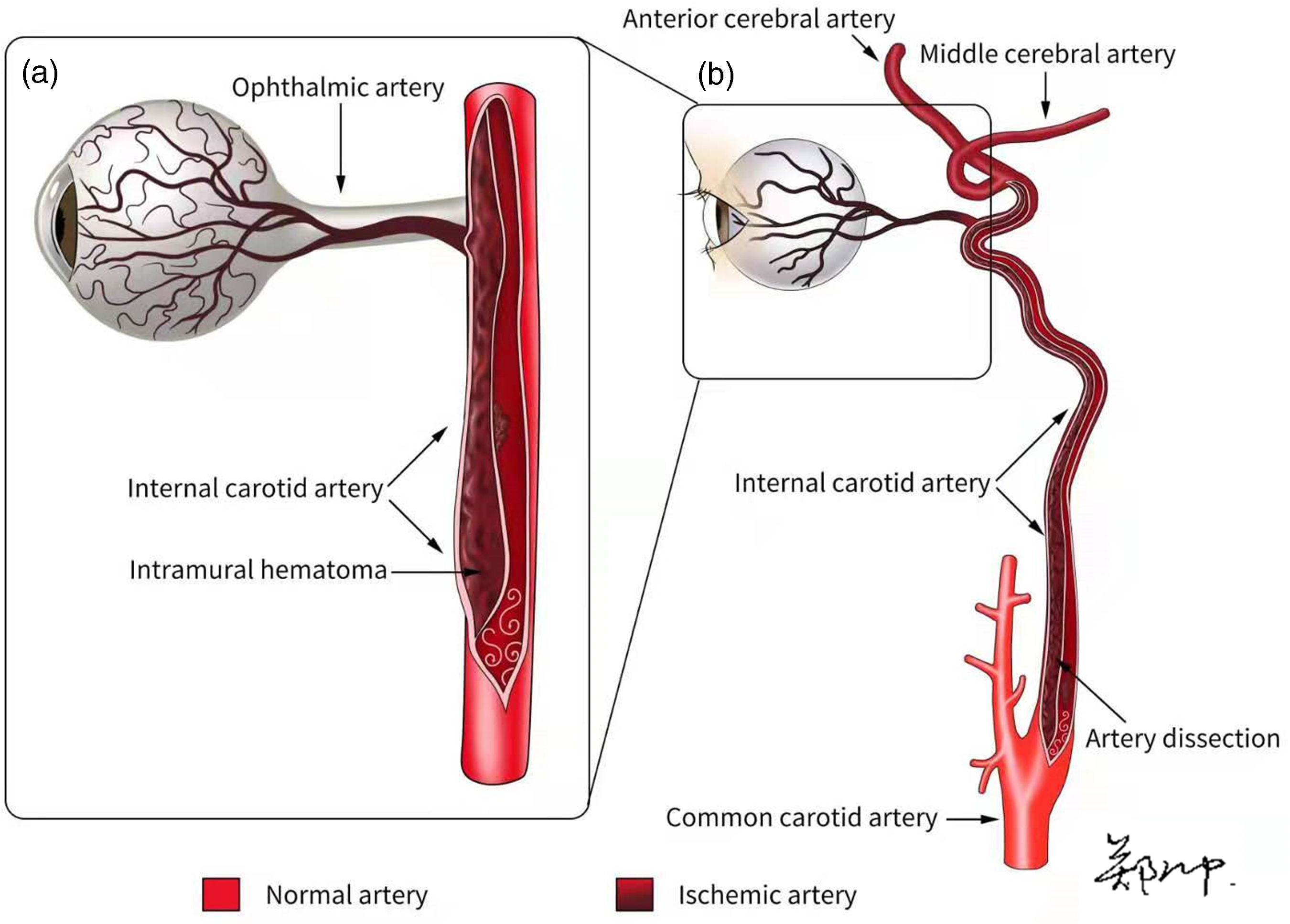
Pathogenesis. (a) Intramural hematoma occluding the origin of the ophthalmic artery and (b) Schema demonstrating the full length of the artery dissection.
Based on the severity of the lesions in our two patients, the doctors chose different treatment methods, and the outcomes were also very different. When clinicians are faced with different clinical symptoms, using a variety of imaging methods to diagnose ICAD in a timely and accurate manner can help in choosing an appropriate treatment plan as soon as possible and reduce complications. Notably, ICAD involving distal branch arteries presents a great challenge in the performance of EVT.
ICAD is an important cause of stroke in young patients, and the symptoms are often nonspecific, which can hinder the diagnosis. Thrombolytic therapy is currently recommended for patients with ICAD who meet the treatment criteria and have no contraindications, and both antiplatelet and anticoagulant agents have shown similar efficacy. The failure of medical management and contraindications to the use of anticoagulation are the most common indications for EVT.
11
Footnotes
Acknowledgement
We are very grateful to Professor Hongwei Zhou for selflessly providing valuable case information that not only broadened our horizons but also exercised our writing ability.
Author contributions
The corresponding author, Professor Tiefeng Ji, provided the information regarding these cases and conceived, planned, and revised the article. The first author, Zheng Chong, drafted, designed, wrote, and revised the article, and drew the illustration in ![]() . The remaining authors individually contributed to the completion of this paper.
. The remaining authors individually contributed to the completion of this paper.
Data availability statement
All data related to this case report are documented within this manuscript.
Declaration of conflicting interests
All the authors declare that there is no conflict of interest.
Ethics statement
This study was approved for publication by the Ethics Committee of the First Hospital of Jilin University (approval no. AF-IRB-032-06). The patients provided written informed consent for treatment and publication, and the patients’ identifying details have been removed from the figures.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Finance of Jilin Province, China (Project number: 3D5205164428). The funding body played no role in the collection, analysis, and interpretation of the data or the writing of this manuscript.
