Abstract
Rapid reduction of plasma triglycerides (TG) is believed to improve the outcome of pancreatitis in the context of hypertriglyceridaemia (HTG)-induced acute pancreatitis (HTG-AP). Previous studies have suggested that haemoperfusion (HP) with the Jafron cartridge series could be effective for reducing TG concentrations in patients with HTG-AP. However, the clearance capacity (CC) for TG removal has not been reported. This case series reports on data from three patients with HTG-AP who underwent HP with HA230 or HA330 cartridges. Blood samples were collected from both before and after the cartridge circuit every 30 min and the CC was calculated. Twelve pairs of blood samples were collected for each type of HP cartridge. The mean ± SD CC of the HA230 cartridge for TG removal in this case series was 0.009781 ± 1.117235 ml/min (95% confidence interval [CI], –0.7000762, 0.7196384 ml). The mean ± SD CC of the HA330 cartridge for TG removal in this case series was 0.344914 ± 1.412183 ml/min (95% CI, –0.5523448, 1.2421721 ml). Based on the findings of this small case series, special caution is advised when considering the use of the HA230 and HA330 cartridges for reducing blood TG concentration pending further conclusive evidence from larger studies.
Keywords
Introduction
Hypertriglyceridaemia (HTG) is an important cause of acute pancreatitis and tends to worsen the severity and related complications. 1 Rapid reduction of plasma triglyceride (TG) level is believed to improve the outcome of pancreatitis in the context of HTG-induced acute pancreatitis (HTG-AP). 2 In addition to refraining from oral food intake and administering insulin and heparin, plasma exchange is recommended as a rescue intervention for severe HTG and pancreatitis. 3 Some researchers have suggested that haemoperfusion (HP) with the Jafron HA cartridge series (Jafron Biomedical, Zhuhai, China) could be an effective treatment option for reducing TG concentrations in patients with HTG-AP.4–6 The use of HA cartridges for reducing TG in patients with HTG-AP is now quite common in some locations. 7 The effectiveness of HP depends on the specific affinity between the targeted substance and the type of cartridge used. 8 To the best of our knowledge, the extraction rate (ER) and clearance capacity (CC) of the Jafron HA cartridge series on TG removal have not been reported. This current case series followed four HP sessions from three patients with HTG-AP to determine the clearance capacity of HA230 and HA330 cartridges on TG removal.
Case reports
Case 1
In June 2023, a 48-year-old male with a previous history of HTG-AP and diabetes mellitus presented to The Third Hospital of Xiamen, Xiamen, Fujian Province, China complaining of fatigue, dizziness and vomiting. The patient was initially admitted to a Gastroenterology ward and upon admission, his fasting TG concentration was measured at 57.74 mmol/l. Treatment was initiated with 200 mg fenofibrate capsules administered orally once daily, 10 mg ezetimibe administered orally once daily and insulin (regular insulin, 0.1 units/kg per hour, administered intravenously, continuously adjusted based on monitored blood glucose levels). Additionally, a clear liquid diet was prescribed, along with 1 litre of intravenous fluid administration. The patient's condition did not improve and he developed severe upper abdominal pain. On the second day of admission, his TG concentration measured 51.24 mmol/l, leading to the patient being transferred to the intensive care unit (ICU) with a diagnosis of HTG-AP. Fasting was initiated for the patient and insulin was administered intravenously while monitoring blood glucose levels. A session of HP using an HA230 cartridge was also performed (written consent for treatment was obtained) in addition to the prescribed medication. Due to signs of hypovolaemia, 3 litres of intravenous fluid were concomitantly administered to the patient. Another session of HP using an HA230 cartridge was performed on the third day of admission. The patient's abdominal pain gradually subsided and the condition improved. The measured TG concentration on the fourth day was 13.04 mmol/l. Subsequently, the patient was transferred back to the Gastroenterology ward for further recovery. The patient made a full recovery and was discharged on the 15th day.
Case 2
In August 2023, a 27-year-old male without any previous medical conditions was admitted to a Gastroenterology ward at The Third Hospital of Xiamen, Xiamen, Fujian Province, China due to persistent severe upper abdominal pain and elevated amylase levels. The patient was kept on a fasting regimen and received intravenous fluids at a rate of approximately 3 litres per 24 h. Intravenous insulin (regular insulin, 0.1 units/kg per hour, administered intravenously, continuously adjusted based on monitored blood glucose levels) and subcutaneous heparin (50 mg enoxaparin, subcutaneously every 12 h, for 5 days) were administered due to the presence of a milky blood sample. On the morning of the second day of admission, his TG concentration was 51.8 mmol/l. Unfortunately, the patient's condition deteriorated, with an elevated C-reactive protein (CRP) level of 253.51 mg/l. On the third day, a contrast-enhanced computed tomography (CT) scan revealed necrotic pancreatitis with a Modified CT Severity Index (MCTSI) score of 9. The patient was transferred to the ICU. The patient was treated with intravenous fluids, insulin and analgesia (remifentanil hydrochloride for injection, 0.03 µg/kg per min, intravenously, continuously adjusted based on visual analogue scale [VAS] pain score and adjusted based on Behavioural Pain Scale after intubation). In order to reduce the elevated inflammation, a session of HP with an HA330 cartridge was performed (written consent for treatment was obtained) and 1150 ml of dark brown ascites was drained. On the morning of the fourth day, his TG concentration was 6.14 mmol/l, the CRP level was 226.97 mg/l and the PaO2/FiO2 ratio dropped to 210. In addition, the intra-abdominal pressure remained at 18 mmHg despite continuous ascites drainage. Consequently, the patient was intubated and sedated with midazolam (midazolam, 0.05 mg/kg/h, intravenously, continuously adjusted based on Richmond Agitation-Sedation Scale). After 14 days of treatment, the patient gradually improved and was extubated. The patient made a full recovery and was discharged on the 32nd day.
Case 3
In September 2023, a 39-year-old male with a previous history of HTG was admitted to a Gastroenterology ward at The Third Hospital of Xiamen, Xiamen, Fujian Province, China due to persistent severe upper abdominal pain and elevated amylase levels. The patient was kept on a fasting regimen and received intravenous fluids. The TG concentration was 55.09 mmol/l. Intravenous insulin (regular insulin, 0.1 units/kg per hour, administered intravenously, continuously adjusted based on monitored blood glucose levels), subcutaneous heparin (50 mg enoxaparin, subcutaneously every 12 h, for 5 days) and 0.2 mg octreotide (subcutaneously every 8 h, for 7 days) were given. In the morning of the second day of admission, the TG concentration had reduced to 10.58 mmol/l, the CRP level was 199.06 mg/l and a contrast-enhanced CT scan revealed necrotic pancreatitis with an MCTSI score of 7. As a result, the patient was transferred to the ICU. After assessing the patient’s volume status, intravenous fluid was given at a rate of 1.5 ml/kg per hour. In addition to the previous medications, the analgesia (remifentanil hydrochloride for injection, 0.03 µg/kg per min, intravenously, continuously adjusted based on VAS pain score) was increased and a session of HP with an HA330 cartridge was performed (written consent for treatment was obtained) to reduce the elevated inflammation. After 16 days of treatment, the patient gradually recovered and was discharged.
Written permission was obtained from the three patients for the findings of this study to be published in this case series. This study was approved by the Ethics Committee of The Third Hospital of Xiamen, Xiamen, Fujian Province, China (no. LLLW2023004). All patient details have been de-identified. The reporting of this study conforms to CARE guidelines. 9
In our clinical practice, the decision to initiate HP treatment is made at the discretion of the treating clinician. Vascular access is established via a double-lumen central venous catheter. HP is performed using the multiFiltrate device (Fresenius Medical Care, Bad Homburg vor der Höhe, Germany) with a synthetic resin cartridge (HA230/HA330; Jafron Biomedical). Heparin is routinely administered for anticoagulation. The blood flow is set at 150 ml/min for a duration of 3 h. After the HP process, saline is used for the return of treated blood. Throughout all four sessions of HP treatment in these three cases, blood samples were taken simultaneously from both before and after the column circuit every 30 min. TG concentration was measured using a Beckman Coulter AU2700/5400 analyzer and TG reagent OSR66118 (Beckman Coulter, Brea, CA, USA). Grossly lipaemic samples were routinely diluted 1 part sample to 4 parts saline prior to analysis and the results were multiplied by five. Estimates of precision, based on Clinical and Laboratory Standards Institute (CLSI) recommendations, 10 were consistent with typical performance. The within-run precision was <3% coefficient of variation (CV) and the total precision was <5% CV. Assays of serum pools were performed and the data were reduced following the CLSI guidelines mentioned above. The CC was calculated as follows: CC = QB (1–haematocrit) × [(CB–CA)/CB]; where QB is the blood flow through the cartridge, CB is the before cartridge plasma concentration, and CA is the after-cartridge plasma concentration. [(CB–CA)/CB] is known as the ER. 11
The TG concentrations from before and after the cartridge circuit during each of the four HP sessions at every time point for each of the three cases, as well as the corresponding ER and CC, are presented in Tables 1–4. All statistical analyses were performed using the R statistical package (R version 4.3.1; R Foundation for Statistical Computing, Vienna, Austria). The TG concentration of every sample was entered into the R statistical package, along with concomitant blood flow and haematocrit values. The ER and CC of each time point for each HP session were derived using the formula described above. The ER and CC of the two different cartridges were checked for normal distribution using the Shapiro–Wilk test and are reported as mean ± SD. 95% confidence intervals (CI) were calculated to estimate the population means. The mean ±SD ER of HA230 on TG removal from this case series was 0.0000019 ± 0.0115704 (95% CI, −0.007349529, 0.007353389). The mean ±SD CC of HA230 on TG removal from this case series was 0.009781 ± 1.117235 ml/min (95% CI, −0.7000762, 0.7196384 ml/min). The mean ± SD ER of HA330 on TG removal from this case series was 0.0034867 ± 0.0141362 (95% CI, −0.005495009, 0.012468382). The mean ± SD CC of HA330 on TG removal from this case series was 0.344914 ± 1.412183 ml/min (95% CI, −0.5523448, 1.2421721 ml/min).
Triglyceride (TG) concentration data from the first session of haemoperfusion using an HA230 cartridge in case 1.

TG ct B, TG concentration from sample taken before the cartridge circuit; TG ct A, TG concentration from sample taken after the cartridge circuit; ER, extraction ratio; BF, blood flow for haemoperfusion; HCT, haematocrit.
The ER and CC values are rounded to the three decimal places for accuracy.
Triglyceride (TG) concentration data from the second session of haemoperfusion using an HA230 cartridge in case 1.

TG ct B, TG concentration from sample taken before the cartridge circuit; TG ct A, TG concentration from sample taken after the cartridge circuit; ER, extraction ratio; BF, blood flow for haemoperfusion; HCT, haematocrit.
The ER and CC values are rounded to the three decimal places for accuracy.
Triglyceride (TG) concentration data from the session of haemoperfusion using an HA330 cartridge in case 2.
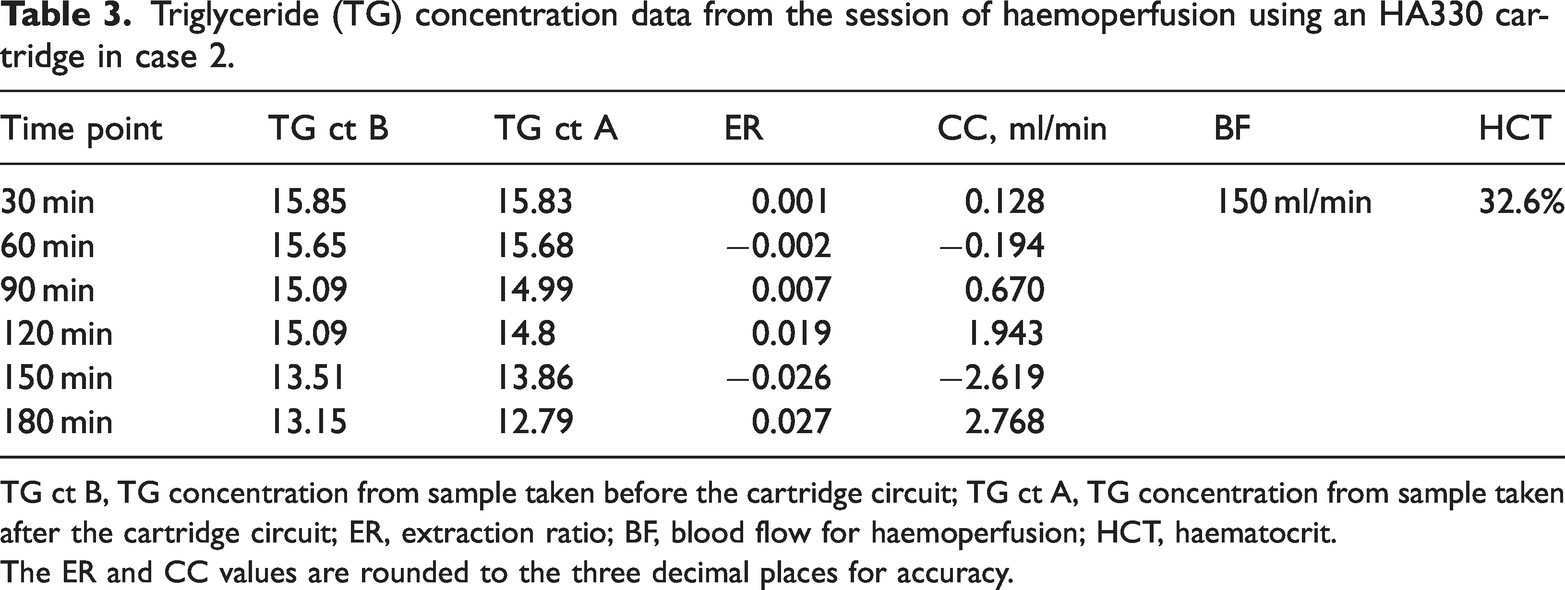
TG ct B, TG concentration from sample taken before the cartridge circuit; TG ct A, TG concentration from sample taken after the cartridge circuit; ER, extraction ratio; BF, blood flow for haemoperfusion; HCT, haematocrit.
The ER and CC values are rounded to the three decimal places for accuracy.
Triglyceride (TG) concentration data from the session of haemoperfusion using an HA330 cartridge in case 3.

TG ct B, TG concentration from sample taken before the cartridge circuit; TG ct A, TG concentration from sample taken after the cartridge circuit; ER, extraction ratio; BF, blood flow for haemoperfusion; HCT, haematocrit.
The ER and CC values are rounded to the three decimal places for accuracy.
Discussion
Data collected during the HP sessions administered to the three patients in this current case series enabled the calculation of the ER and CC of the two cartridges HA230 and HA330 that were used for TG removal. Data from two sessions of HP administered to case 1 demonstrated that the HA230 cartridge contributed minimally to TG reduction and is no longer used for this purpose in our hospital. As previous publications primarily report the effectiveness of HP in reducing TG concentrations using the HA330 cartridge,4–6 the current case series also measured the ER and CC on TG removal in two additional cases who underwent HP with the HA330 cartridge for inflammation reduction. According to the upper limit of the 95% CI for the CC of the HA330 cartridge for TG removal, a 3-h treatment can at most clear a total of 223.596 ml of blood (calculated as 1.2422 ml/min, multiplied by 60 min and 3 h), which accounts for only 4.5% of the total blood volume.
In the context of HTG-AP, TG concentrations reduce when food intake is stopped. Treatments such as intravenous hydration, analgesia and intravenous insulin infusion further accelerates the reduction of TG concentrations. 12 Assessing the effectiveness of concomitant HP by measuring the concentration of TGs before and after treatment can lead to misleading conclusions. Measuring ER and CC by comparing solute concentrations taken simultaneously from before and after the cartridge circuit is a common quantifying method used to assess the effectiveness of HP cartridges for certain materials. This approach excludes the effects of other concurrent factors. 11 As HA230/330 cartridges have already been clinically applied for TG reduction in some places, it is imperative to know their ER and CC on TG removal.
It may seem confusing as there were some negative ER and CC values in the current case series. This current study used the glycerol phosphate oxidase chromogenic method to determine the TG concentrations of the blood samples. When the blood samples had an extremely lipaemic appearance, they were routinely diluted to five-times their original volume and the results were multiplied by five. Although the within-run precision was < 3% CV and the total precision was < 5% CV, the additional procedure of dilution may have increased the measuring error to some extent. It is likely that any small real CC has been masked by the measuring noise, resulting in the negative values. Therefore, all 12 ER and CC measurements for each type of HP cartridge were used to calculate the confidence interval to estimate the possible upper limit of the ‘real’ ER and CC. There was also the possibility that deposited TG was released from the cartridge, which would then increase the TG concentration of blood samples collected after they had flowed through the cartridge. It might take an extensive laboratory study to ascertain the precise CC and to explore TG redistribution during cartridge use.
The HA230 and HA330 cartridges contain resin beads made of styrene-divinylbenzene copolymer. The HA230 cartridge is used to remove hydrophobic or protein-bound exogenous substances such as drugs, while the HA330 cartridge is used to remove pro-inflammatory cytokines and other cellular inflammatory markers found in a ‘cytokine storm’ or pancreatitis. 13 In an in vitro study aimed at evaluating the histocompatibility and absorption of the organophosphate pesticide chlorpyrifos by styrene-divinylbenzene copolymer resin, the TG concentration was elevated after 2 h of incubation. 14 However, the blood samples in this previous study were obtained from healthy volunteers with normal range TG concentrations (1.46 ± 1.35 mmol/l) and may not fully reflect the absorption potential of the styrene-divinylbenzene copolymer resin. 14 The findings of this current case series provides further data, but also raises questions about the efficacy of the HA230 and HA330 cartridges in reducing TG concentrations in patients with HTG-AP.
In conclusion, the preliminary findings of this small case series suggest that neither the HA230 nor HA330 cartridge were effective at reducing TG concentrations. Due to the unexpectedly low CC for TG and the presence of measuring errors, it is only possible to estimate the potential upper limit of TG removal using these cartridges, rather than obtaining a precise value for TG clearance or its variance over time. Special caution is advised when considering the use of the HA230 and HA330 cartridges for reducing blood TG concentration pending further conclusive evidence from larger studies.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241252607 - Supplemental material for Assessment of triglyceride clearance of haemoperfusion from three cases of hypertriglyceridaemia-induced acute pancreatitis: a case series
Supplemental material, sj-pdf-1-imr-10.1177_03000605241252607 for Assessment of triglyceride clearance of haemoperfusion from three cases of hypertriglyceridaemia-induced acute pancreatitis: a case series by Wah Kwong Yip, Bin Wu, Bao Hua Ye, Min Li Chen, Zi Yao Wu and Cheng Yi Ji in Journal of International Medical Research
Footnotes
Acknowledgements
We extend our sincere gratitude to Professor Mingwei Zhang, ICU, The First Affiliated Hospital of Xiamen University, Xiamen, Fujian Province, China. His inspiration and invaluable advice were crucial in the conception and development of this study.
Author contributions
W.Y. and B.Y. contributed equally to this study. W.Y., B.W. and B.Y. discussed and conceived the study. W.Y., C.J., Z.W. and M.C. investigated the study and collected the data. B.W. validated the study. W.Y. wrote the original draft. All authors have read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by a grant from Xiamen Third Hospital (no. 3502Z20224ZD1339). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
