Abstract
Among the various manifestations of COVID-19, the neurological implications of SARS-CoV-2 infection are of significant concern. Marchiafava–Bignami disease (MBD), a neurodegenerative disorder, exhibits a clinical spectrum ranging from mild progressive dementia in its chronic form to states of acute coma and varied mortality rates. Acute MBD primarily occurs in chronic alcoholics and malnourished individuals and is characterized by sudden loss of consciousness, seizures, confusion, and psychosis. We herein report a case of MBD presenting as acute loss of consciousness after the development of COVID-19. The patient presented with a history of fever and upper respiratory infection and was diagnosed with SARS-CoV-2 infection. He developed a neurological syndrome characterized by altered consciousness and convulsions, and brain magnetic resonance imaging revealed abnormal signals in the corpus callosum and frontoparietal lobes. Considering his alcohol intake history and the absence of other differential diagnoses, we diagnosed him with acute MBD triggered by COVID-19. After high-dose vitamin B1 and corticosteroid therapy, his clinical symptoms improved. In this case, we observed a temporal sequence between the development of COVID-19 and acute exacerbation of MBD. This case adds to the mounting evidence suggesting the potential effect of SARS-CoV-2 on the neurological system.
Keywords
Introduction
The COVID-19 pandemic has had a profound impact on global public health and healthcare systems.1–3 Among its various manifestations, the neurological implications of SARS-CoV-2 infection are of significant concern. 4 A previous study showed that neurological manifestations could be identified in one-third of patients with COVID-19, predominantly during the early and middle stages of the illness. 5 For some patients, neurological signs were even the primary presenting symptoms. Further complicating matters, COVID-19 may precipitate certain neurological conditions such as stroke, particularly in individuals with pre-existing neural damage. 6 The mechanisms by which COVID-19 induces neurological disorders are under active investigation, and much remains unknown. However, accumulating observational data underscore the multi-systemic impacts of COVID-19 and hint at the possibility of concurrent neurological complications. Prompt consultation with healthcare professionals for accurate assessment and appropriate treatment is essential for patients with COVID-19 experiencing neurological symptoms.7,8
Marchiafava–Bignami disease (MBD), a neurodegenerative disorder, exhibits a clinical spectrum ranging from mild progressive dementia in its chronic form to states of acute coma and varied mortality rates. Acute MBD primarily occurs in chronic alcoholics and malnourished individuals and is characterized by sudden loss of consciousness, seizures, confusion, and psychosis.9,10 The etiology likely involves alcohol-induced neurotoxicity combined with a deficiency of vitamin B1. Other possible factors include ketoacidosis secondary to diabetes or alcoholism, malnutrition following gastric bypass surgery, carbon monoxide poisoning, sepsis, cerebral malaria, sickle cell disease, and sudden serum osmotic fluctuations induced by cardiac cancer surgeries.11–14 MBD is characterized by corpus callosum degeneration, potentially affecting other brain areas and leading to disrupted interhemispheric communication. This results in cognitive and motor impairments, with involvement of additional brain regions often indicating a worse prognosis. Understanding these neuroanatomical abnormalities is crucial for managing the diverse symptoms and complications of MBD. 15
We herein report a case of acute MBD following the development of COVID-19. The patient’s condition was determined after ruling out differential diagnoses such as viral encephalitis and autoimmune encephalitis. We managed the patient with high-dose vitamin B1 and corticosteroids, ultimately observing both radiological improvements and alleviation of clinical symptoms.
Case report
A 55-year-old man with a 1-week history of fever and upper respiratory symptoms was diagnosed with SARS-CoV-2 infection through nucleic acid testing. After symptomatic treatment including antipyretics, the patient’s condition stabilized. Two days later, however, he developed a neurological syndrome characterized by unprovoked altered consciousness and convulsions in all four limbs. Following an episode of convulsions that self-terminated after 1 minute without incontinence or tongue biting, the patient presented with delirium and prominent hallucinations. His consciousness deteriorated further 2 hours later, necessitating intensive care unit admission. The patient had no history of preceding head injury, involuntary movements, rash, arthralgia, oral ulcers, photosensitivity, reduced urination, dyspnea, diarrhea, intravenous drug abuse, recurrent blood transfusions, developmental delay, incontinence, hypertension, cardiac disease, diabetes, cerebrovascular disease, hepatitis, tuberculosis, or malaria. He admitted to a decade-long history of alcohol intake, averaging 250 g of distilled spirits per day without cessation. He denied any cranial nerve or sensory impairment.
Upon physical examination, the patient was semi-comatose (Glasgow Coma Scale score of E2V2M2), had a blood pressure of 110/58 mmHg, and had a pulse rate of 65 beats per minute.16,17 His bilateral pupils were equal in size and both directly and indirectly reactive to light. The patient showed increased muscular tension in all four limbs, but he did not cooperate with the muscular strength examination. Hoffmann’s and pathological signs were both negative bilaterally. The sensory system, cerebellar signs, and gait could not be assessed, but other system examinations were unremarkable. Routine blood tests, liver and kidney function tests, electrolyte levels, blood glucose, lipids, and cardiac injury markers were all within normal limits.
Chest digital radiography revealed thickening of the lung texture with small patchy opacities (Figure 1). Brain magnetic resonance imaging (MRI) showed multiple patchy abnormal signals in the corpus callosum, appearing as high signals on fluid-attenuated inversion recovery imaging and diffusion-weighted imaging (DWI) and as low signals on the apparent dispersion coefficient map (Figure 2(a), (b)). No evident enhancement was observed in the corpus callosum lesion area on enhancement scanning. In addition, the perfusion in the lesion area of the corpus callosum showed unevenly increased activity on perfusion-weighted imaging. Intracranial arteries showed no evident stenosis on magnetic resonance angiography. Furthermore, scattered dot-like, nodular, and patchy abnormal signals were observed bilaterally in the centrum semiovale and frontoparietal lobes.
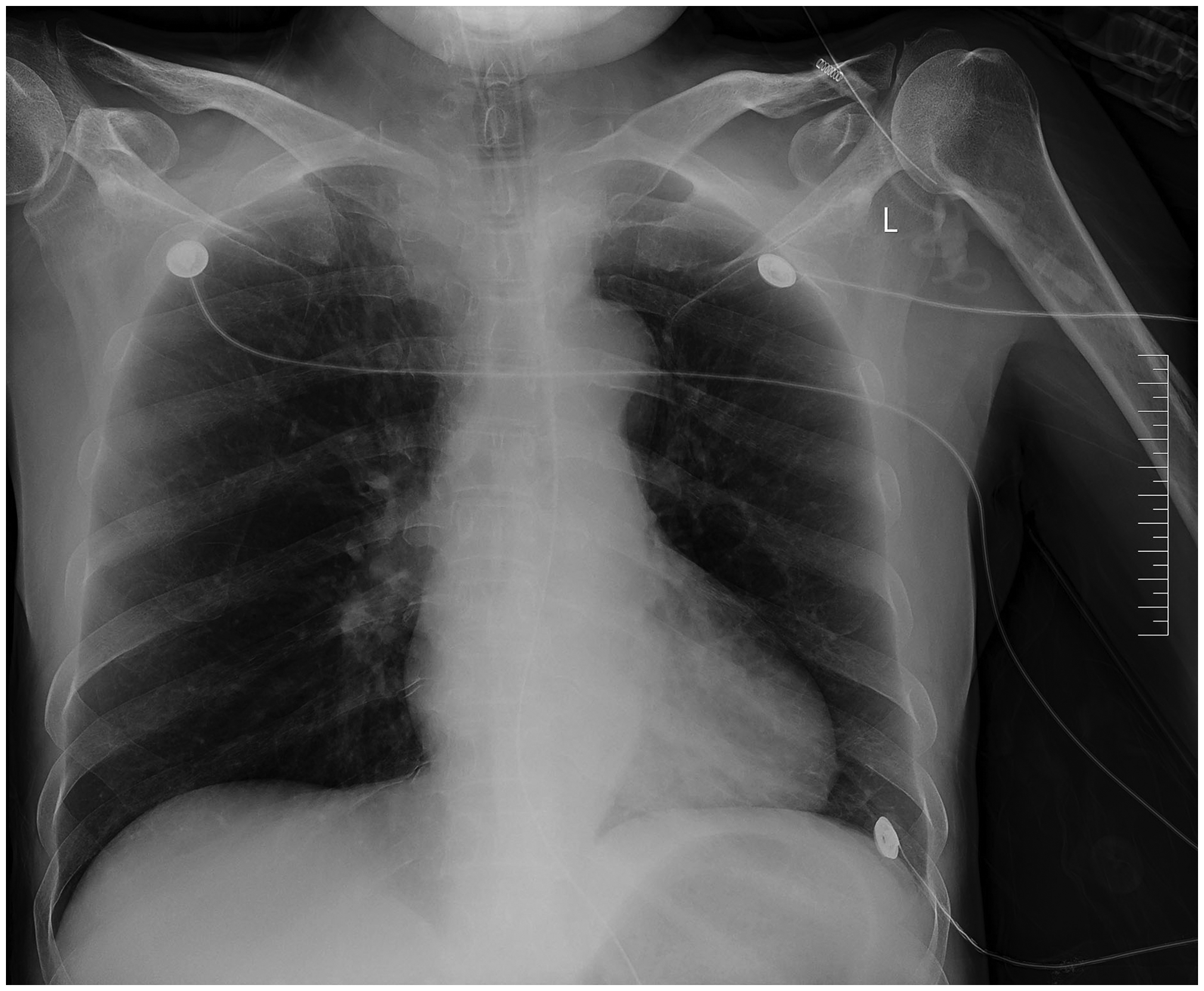
Chest digital radiograph showing small patchy opacities.

Magnetic resonance imaging findings. (a) An array of patchy hyperintensities can be observed on fluid-attenuated inversion recovery imaging within a multitude of regions encompassing the basal ganglia, corpus callosum, bilateral subcortical regions of the frontal and parietal lobes, corona radiata, and centrum semiovale. (b) Diffusion-weighted imaging of the patient’s brain showed high signals, and the apparent diffusion coefficient map of the patient’s brain showed low signals. (c) Fluid-attenuated inversion recovery imaging of the corpus callosum showed resolution of the abnormal signals in most of the original lesions and (d) diffusion-weighted imaging and the apparent diffusion coefficient map with improved symptoms after treatment.
The patient was continuously monitored with electrocardiography and received treatments to maintain his fluid and electrolyte balance, resulting in gradual improvement of consciousness. Lumbar puncture was performed following another episode of generalized tonic-clonic seizures on the second day of admission. The patient was negative for autoimmune encephalitis antibodies. Because of his history of ingesting 300 to 400 mL of homemade alcohol per day for 180 days prior to the hospital visit, methanol poisoning and Wernicke’s encephalopathy were considered as differential diagnoses. He was treated with an increased dosage of thiamine (500 mg three times a day), vitamin B12, and multivitamin supplements. Acute disseminated encephalomyelitis secondary to methanol abuse or occupational toxin exposure was likely; therefore, the patient also received methylprednisolone pulse therapy (1 g once a day for 3 days). After further symptom improvement, electromyography was performed. Peripheral neuropathy was ruled out because the results of a nerve conduction velocity test were within normal limits. Electroencephalography showed diffuse slowing of the brain waves with no other abnormalities.
Given the absence of neuroimaging features of methanol poisoning, Wernicke’s encephalopathy, acute disseminated encephalomyelitis, and adult leukodystrophy in our patient, we diagnosed him with acute MBD triggered by COVID-19. However, genetic testing was not conducted, so the possibility of hereditary adult leukodystrophy could not be completely ruled out. After completion of hormone therapy on the eighth day of hospitalization, his clinical symptoms improved. He continued treatment with oral steroids (1 mg/kg), thiamine, and multivitamin supplements. His clinical condition improved significantly except for mild gait impairment. He was discharged on the 12th day of hospitalization with a regimen involving 6 weeks of steroid tapering and oral thiamine supplements. At 4 weeks post-discharge, repeat DWI showed mild resolution of the edema and radiological findings compared with the previous scans (Figure 2(c), (d)). Three months after discharge, the patient showed gradual resolution and eventual disappearance of his symptoms. Follow-up head MRI showed resolution of the abnormal signals in most of the original lesions (Figure 2(c), (d)). Despite clinical improvement, the radiological features persisted.
All above-mentioned treatments were performed with the consent of the patient and his family. The review board waived the requirement for ethics approval because this study constitutes a case report involving a single patient whose personal information has been anonymized, and no significant intervention was performed. The reporting of this study conforms to the CARE guidelines, and written consent was obtained from the patient to share his clinical data. 18
Discussion
MBD is a rare severe neurodegenerative complication of chronic alcoholism and is predominantly characterized by demyelination of the corpus callosum. Its clinical manifestations are distinct, primarily including psychiatric symptoms such as cognitive impairment, an altered mental state, behavioral anomalies, and psychomotor agitation. The typical pattern of cognitive impairment in patients with MBD is characterized by significant memory impairments accompanied by varying degrees of executive dysfunction and attentional deficits, which contribute to its multifaceted and intricate clinical manifestations. 19 Neurological motor impairments often involve elevated muscle tone, incoordination, muscle weakness, and spasticity. Ocular abnormalities, such as nystagmus, involuntary eye movements, gaze deviation, and ophthalmoplegia, are not uncommon. The defining characteristic of MBD, namely demyelination of the corpus callosum, can be detected through neuroimaging.20,21
On the basis of the bilateral symmetric involvement and demyelination of the corpus callosum in our patient, we pursued a diagnosis of MBD. He had been a chronic alcohol consumer for the past decade and presented with acutely altered consciousness following the development of COVID-19. The diagnosis of acute MBD was driven by MRI findings: bilateral symmetric high signal intensities and edema in the corpus callosum, involving the body, splenium, and callosal edema. The signals extended to the posterior limbs of the internal capsules and the white matter of the bilateral frontal and parieto-occipital lobes. High signals were also observed in the bilateral cerebral peduncles, central tegmental tracts, and white matter of the frontal and temporal lobes. Diffusion restriction in the corpus callosum, as shown on DWI and corresponding apparent diffusion coefficient sequences, solidified the diagnosis of MBD. This evidence ruled out differential diagnoses such as post-vaccination acute disseminated encephalomyelitis, Wernicke’s encephalopathy, multiple sclerosis, and methanol toxicity. The prognosis of acute MBD with cortical involvement is generally poor; however, the literature suggests that patients with MBD often respond favorably to intravenous thiamine, corticosteroids, oral folate, and other vitamin B complex supplements as well as maintenance of stable plasma glucose levels. Our patient exhibited improvements in his clinical symptoms following 5 days of pulsed corticosteroid treatment and escalated doses of thiamine supplementation along with proactive nutritional management. With the exception of a mild gait disorder, no significant neurological signs remained, thus validating the effectiveness of the treatment and supporting the MBD diagnosis.9–11
Currently, there is no conclusive evidence supporting a direct causative or aggravating link between COVID-19 and primary corpus callosum degenerative disease. Nevertheless, we observed a temporal sequence between the development of COVID-19 and acute exacerbation of MBD in this case. Possible mechanisms might involve immune dysregulation: COVID-19 may cause inappropriate immune responses and inflammation, processes that are implicated in the development of corpus callosum degenerative diseases. Consequently, COVID-19 could trigger or exacerbate existing degenerative diseases of the corpus callosum, either by disrupting normal immune function or provoking additional inflammatory responses. Additionally, COVID-19 might directly induce neurological damage as suggested by reported neurological symptoms such as headaches and loss of smell and taste, suggesting potential direct or indirect impacts on the central nervous system.22,23 If such effects extend to the corpus callosum, they could induce neuronal damage and degeneration, thereby worsening symptoms of degenerative diseases of the corpus callosum. For example, disruption of the blood–brain barrier, a known consequence of severe COVID-19, may allow the entry of harmful inflammatory mediators into the central nervous system, further compromising the integrity of the corpus callosum.24,25 It is important to underscore that these hypotheses currently lack sufficient scientific evidence, necessitating further research to ascertain whether a direct or indirect link exists between COVID-19 and corpus callosum degenerative diseases. This requires a multidisciplinary approach that integrates clinical, immunological, and neuroscientific perspectives to elucidate the complex dynamics at play. Furthermore, because COVID-19 is a severe disease potentially causing numerous other health issues, we strongly recommend adherence to public health guidelines, maintenance of good hygiene practices, and timely consultation with healthcare professionals for accurate information and advice.
Conclusion
In this case, we observed a temporal sequence between the development of COVID-19 and acute exacerbation of MBD. This case adds to the current evidence suggesting the potential effect of SARS-CoV-2 on the neurological system, extending beyond its well-known respiratory effects.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241234555 - Supplemental material for COVID-19-induced acute loss of consciousness in Marchiafava–Bignami disease: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605241234555 for COVID-19-induced acute loss of consciousness in Marchiafava–Bignami disease: a case report by Jie Tian, Hai-li Wang, Shuang-jie Li and Chang-he Shi in Journal of International Medical Research
Footnotes
Acknowledgement
We would like to express our gratitude to the patient described in this report.
Author contributions
CS contributed to the conception or design of the study. JT and HLW contributed to the acquisition and analysis of the data. JT, SJL, and CS contributed to drafting and substantially revising the manuscript. All authors read and approved the final manuscript.
Data availability statement
The data are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
This work was supported by the Scientific and Technological Research Projects in Henan Province [grant number 222102310033 to Dr. Jie Tian] and the Key Research Project in Higher Education Institutions of Henan Province [grant number 22B320022 to Dr. Jie Tian].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
