Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection can trigger autoimmune inflammation in the liver, leading to acute autoimmune hepatitis (AIH). We herein report a case involving a 39-year-old woman with a 23-day history of yellow skin and urine. Using the revised original scoring system of the International AIH Group, we definitively diagnosed the patient with acute severe AIH (AS-AIH). She began treatment with 80 mg/day intravenous methylprednisolone, which was gradually reduced and followed by eventual transition to oral methylprednisolone. The patient finally achieved a biochemical response after 30 days of therapy, and liver transplantation was avoided. Clinicians should be aware that the onset of AS-AIH after SARS-CoV-2 infection differs from the onset of conventional AIH with respect to its clinical and pathological features. Early diagnosis and timely glucocorticoid treatment are crucial in improving outcomes.
Introduction
Acute severe autoimmune hepatitis (AS-AIH) is a unique subtype of AIH, and its severity lies between that of acute AIH and acute liver failure. The major clinical manifestations of AS-AIH include icterus and coagulation dysfunction, which can quickly progress to acute liver failure. 1 The major histopathological features of AS-AIH are acute lobular inflammation with central lobular necrosis. 2 Early diagnosis and timely glucocorticoid treatment are crucial for a satisfactory outcome, and effective treatment can avoid liver transplantation. 3
The spike glycoprotein (S protein) of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has peptide homology with at least 26 human proteins. After SARS-CoV-2 infection, the immune response against SARS-CoV-2 may include cross-reaction with host proteins, leading to autoimmune injury. 4 The liver, as a special immune organ of the body, can also trigger autoimmune inflammation. 5 We herein report a case of AS-AIH after SARS-CoV-2 infection. We present an analysis of the patient’s clinical data and discuss the entire diagnosis and treatment process.
Case presentation
A 39-year-old woman presented and was admitted to our hospital with a 23-day history of yellow skin and urine. The patient had developed yellow skin and urine approximately 3 weeks after recovery from SARS-CoV-2 infection. A local hospital had diagnosed her with acute icteric hepatitis, and she was given diammonium glycyrrhizinate and ursodeoxycholic acid for more than 20 days. However, her yellow skin and urine did not significantly improve. Thus, on 6 February 2023, the patient was admitted to our hospital with icterus of unknown origin.
The patient’s history was as follows. On 18 December 2022, she was diagnosed with SARS-CoV-2 infection based on an antigen test. Another antigen test was negative on 25 December 2022. The patient had been inoculated twice with the SARS-CoV-2 vaccine (CoronaVac; Sinovac Biotech, Beijing, China) on 31 June 2021 and 1 December 2021. She had no history of chronic hepatitis, blood transfusion, alcohol abuse, or use of hepatotoxic drugs (including herbal and dietary supplements).
Her vital signs on presentation were as follows: body temperature, 36.8°C; heart rate, 83 beats/minute; and blood pressure, 116/75 mmHg. Her physical examination revealed severely yellow skin and sclera.
Liver function tests showed the following results: total bilirubin, 201.82 µmol/L; direct bilirubin, 164.34 µmol/L; alanine aminotransferase, 612 U/L; aspartate aminotransferase, 2305 U/L; alkaline phosphatase, 250 U/L; γ-glutamyl transpeptidase, 131 U/L; albumin, 35.4 g/L; and globulin, 32.5 g/L. Immunological tests showed the following results: immunoglobulin G (IgG), 17.57 g/L; IgG4, 0.55 g/L; antinuclear antibody (ANA), 1:100 (speckle type, subtract: human hep-2 cell); and positivity for anti-Sjögren’s syndrome type A antibody, anti-extractable nuclear antibody, and perinuclear antineutrophil cytoplasmic antibody. Thyroid function tests showed positivity for thyroid peroxidase antibody and thyroglobulin antibody. Coagulation function tests showed a prothrombin time of 16.7 s and an international normalized ratio (INR) of 1.56. The patient was negative for hepatitis A to E, Epstein–Barr virus, cytomegalovirus, coxsackie virus, herpes zoster virus, Treponema pallidum, and human immunodeficiency virus.
Contrast-enhanced upper abdominal computed tomography and magnetic resonance imaging showed a normal liver volume with no signs of cirrhosis. Magnetic resonance cholangiopancreatography showed no signs of extrahepatic bile duct obstruction.
A liver biopsy was performed, and the pathological examination showed that the main periportal change was moderate interface hepatitis and plasma cell infiltration (Figure 1 (a)), while the main lobular changes were hepatic rosette formation (Figure 1(b)), bridging, and massive lobular necrosis (Figure 1(c)). The overall inflammation was graded as level 4. According to the revised original scoring system of the International AIH Group, 6 if the total score is >16 before glucocorticoid therapy, a definitive diagnosis of AIH can be made. The total score of this patient was 21; therefore, she was definitively diagnosed with AS-AIH.
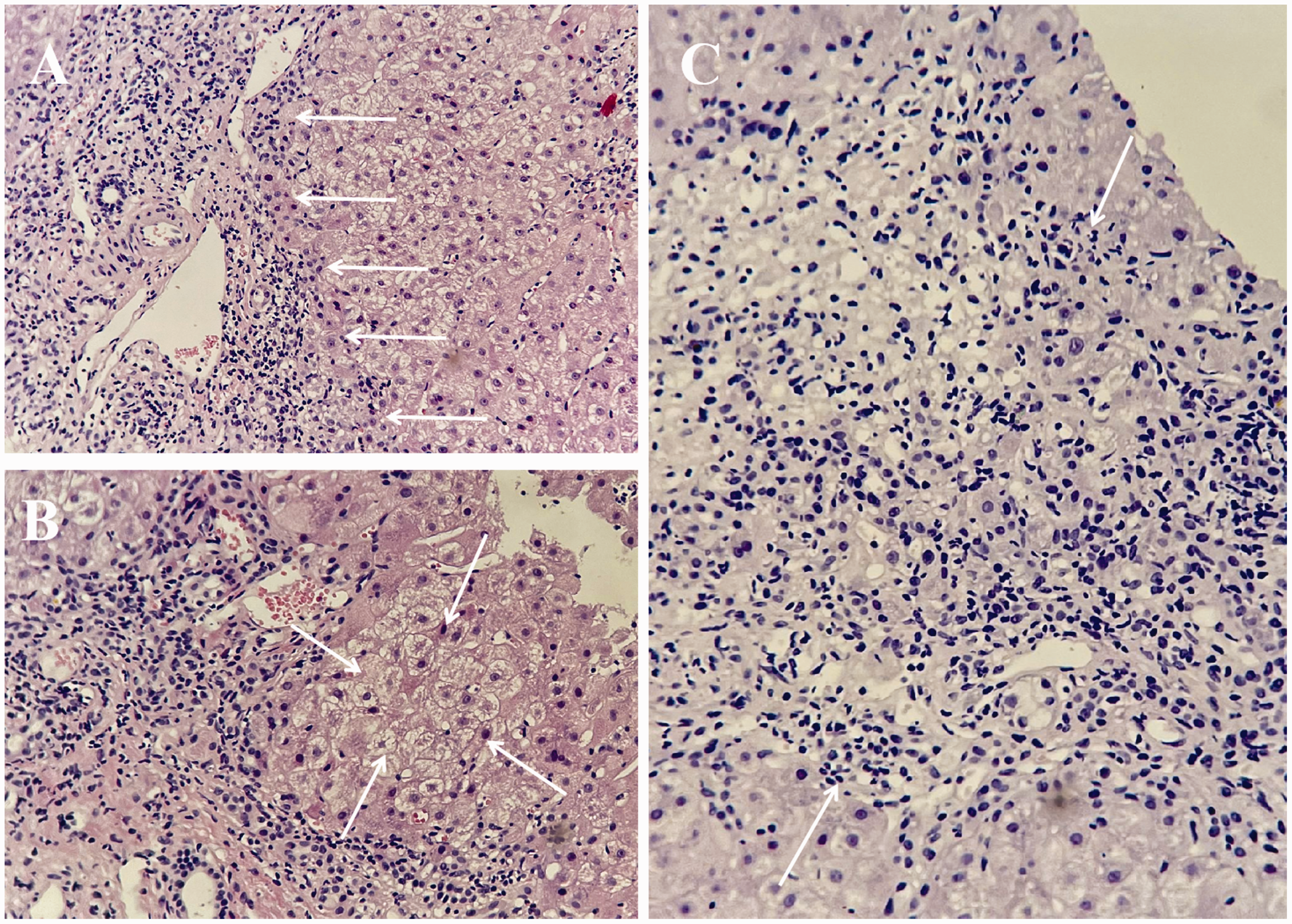
Histological characteristics of liver biopsy. (a) Moderate interface hepatitis and plasma cell infiltration (white arrows) were visible. Hematoxylin–eosin (HE) staining, 200× magnification. (b) Typical sign of rosette formation (white arrows). HE, 200× magnification and (c) bridging and massive lobular necrosis (white arrows). HE, 200× magnification.
The Model for End-stage Liver Disease (MELD) score was 14 after admission. Liver function and immunological indicators were monitored every 3 to 5 days (Figure 2). On 8 February 2023 (day 3 after admission), 80 mg/day methylprednisolone was intravenously administered, and this treatment was continued for 6 days. On the day 7 of methylprednisolone therapy, the MELD score had decreased to 11. Moreover, the patient’s liver function was significantly improved (total bilirubin, 111.02 µmol/L; alanine aminotransferase, 354 U/L; aspartate aminotransferase, 118 U/L), and immunological indices had declined to normal levels (IgG, 11.33 g/L). The dose of methylprednisolone was reduced to 60 mg/day for 3 days and then to 40 mg/day for 3 days. On 20 February, 32 mg/day methylprednisolone was administered orally, and the dose was then reduced to 4 mg weekly. The patient had a good response to methylprednisolone and reached biochemical remission with no relapse. This case is reported in line with the CARE guidelines. 7
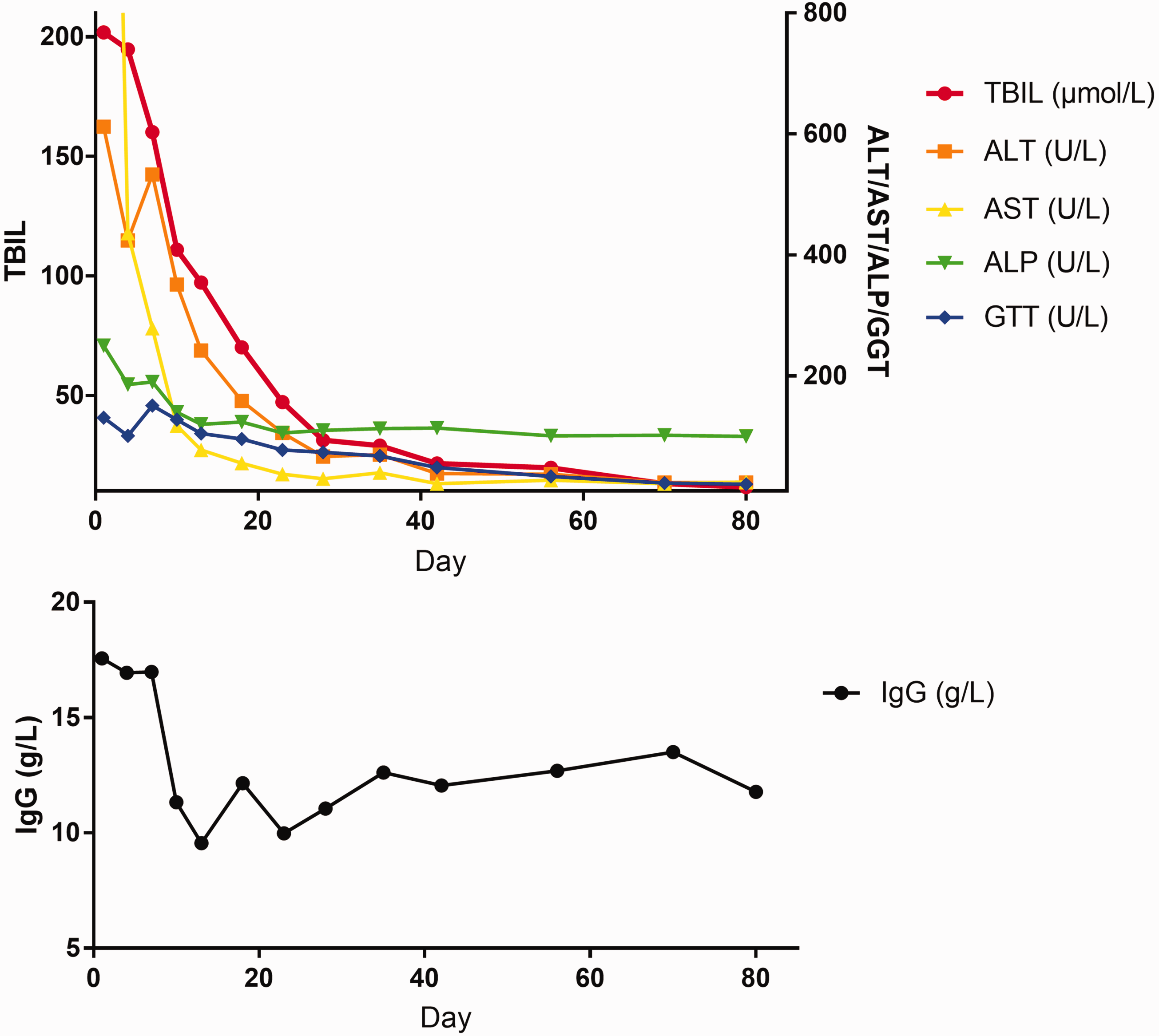
Changes in main liver function and immunological indexes. TBIL, total bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; GGT, gamma-glutamyl transferase; IgG, immunoglobulin G.
Discussion
This report describes a case involving a 39-year-old woman with acute onset of yellow skin and urine approximately 3 weeks after recovery from SARS-CoV-2 infection. With the help of laboratory and pathological tests, AS-AIH was finally diagnosed. Early diagnosis and timely glucocorticoid treatment significantly improved the patient’s condition and prevented liver transplantation.
One prominent feature of this case is that the onset of AS-AIH coincidently occurred 3 weeks after SARS-CoV-2 infection. Studies focusing on the relationship between AIH and SARS-CoV-2 infection are summarized below.
Some research has focused on SARS-CoV-2 infection and the presence of new autoantibodies, especially ANA. Pascolini et al. 8 prospectively studied 33 patients with SARS-CoV-2 infection and found that 45% of the patients tested positive for at least one autoantibody, including 11 (33%) who were positive for ANA, 8 (24%) for anticardiolipin antibodies (IgG and/or IgM), and 3 (9%) for anti-β2 glycoprotein antibodies (IgG and/or IgM). No antineutrophilic cytoplasmic antibody reactivity was detected.
Other studies have examined the underlying mechanism of AIH after SARS-CoV-2 infection. Four major themes can be identified among such studies. The first is molecular mimicry and immune cross reactivity. Antigen cross reactivity plays an important role in natural SARS-CoV-2 infection, which is characterized by autoinflammatory dysregulation with consequent tissue damage mediated by viral spike protein. 9 Moreover, a shared heptapeptide has been recognized between the human proteome and the viral spike glycoprotein. 10 Thus, it is reasonable to believe that spike-directed antibodies induced by SARS-CoV-2 recognize cross-reacting self-antigens or shared antigenic protein sequences. This molecular mimicry can trigger autoimmune conditions in the liver of predisposed individuals, eventually leading to the onset of AIH. 11 The second major research theme is epitope spreading. Some epitopes are not exposed or are expressed at low levels. Lymphocytes specific to autoantigens may escape negative selection during the development of central immune organs and appear in the mature lymphocyte pool. 12 After SARS-CoV-2 infection, epitope spreading occurs through the diversification of epitopes recognized by the immune system, and it can appear to both B and T cells. Endocytic processing, antigen presentation, and somatic hypermutation contribute to prolongation and worsening of autoimmune damage in the liver, 13 eventually leading to the onset of AIH. A third area of focus among these studies is the X chromosome and female susceptibility. Because of the stimulatory effect of estrogens on the immune system, overexpression of X-linked genes provokes a stronger post-vaccination autoimmune reaction in female than male patients. To some extent, sceptical hepatotropism of SARS-CoV-2 may account for such autoimmune phenomena. 14 Finally, bystander hepatitis and autoreactive lymphocytes has been examined in past studies. Activation of systemic cytokines or chemokines can trigger bystander hepatitis. 15 The release of certain cytokines, such as tumor necrosis factor α, interferon γ, and interleukins 1β, 6, and 10, may also occur after SARS-CoV-2 infection and the subsequent activation of autoreactive B and T cells, leading to autoimmune liver injury. 16 SARS-CoV-2–induced spike-specific CD8 T cells were found in the peripheral blood along with extensive CD8 liver infiltration in a patient with post-SARS-CoV-2 vaccine AIH, 17 supporting the idea that autoreactive lymphocytes contribute to the pathogenesis of AIH.
Based on the above evidence, we do not believe that our patient incidentally developed AS-AIH after SARS-CoV-2 infection. Instead, SARS-CoV-2 infection is likely an important triggering factor for an imbalance of the immune system, leading to autoimmune injury of certain organs.
Moreover, most cases of SARS-CoV-2 vaccination-induced AIH are moderate in severity and occur within 2 to 4 weeks after vaccination. 18 By contrast, although the patient in the present case had a history of vaccination 1 year previously, a long period of time had passed between the last SARS-CoV-2 vaccination and AIH. Thus, the evidence for a cause-and-effect relationship between vaccination and AIH is weak in this case. We propose that SARS-CoV-2 infection was the real triggering factor in our patient. Furthermore, our patient followed a rapidly progressive course and was eventually diagnosed with AS-AIH, a rare subtype of conventional AIH. 19 AS-AIH after SARS-CoV-2 infection differs from conventional AIH.
The AIH comprehensive diagnostic scoring system has a significant advantage over the AIH simplified scoring system in diagnosing AS-AIH and can significantly improve the detection rate of AS-AIH.20,21 Our case met all the above criteria, and the diagnosis was confirmed.
The histopathological manifestations of AS-AIH after SARS-CoV-2 infection include intralobular inflammation and central necrosis.22,23 Aizawa et al. 24 demonstrated that centrilobular zonal necrosis might be a unique subtype marker of acute AIH. 25 The bridging and massive lobular necrosis in the present case were consistent with the classic acute onset of AIH and were in agreement with previous reports in the literature.
Patients with AS-AIH have a variable response to corticosteroids. Studies have shown that a MELD score of ≤28 or a United Kingdom Model for End-stage Liver Disease score of ≤57 on admission, absence of massive hepatic necrosis on histology, and improvement in the bilirubin concentration and INR within 4 days after treatment are associated with a high response rate to corticosteroids. 26 Our patient was admitted without hepatic encephalopathy, with a MELD score of 14, and with an INR of 1.56, suggesting a possible good response to glucocorticoids. After 6 days of 80 mg methylprednisolone intravenous therapy, the patient responded well with a decrease in her MELD score from 14 to 11, suggesting a good response to corticosteroid therapy and avoiding liver transplantation. 27
As mentioned above, few studies have examined the connection between SARS-CoV-2 infection and AIH. To our knowledge, this is the first case report of AS-AIH, a rare subtype of AIH, occurring after SARS-CoV-2 infection. In this case, timely treatment with glucocorticoids successfully avoided the need for liver transplantation.
Conclusion
We have herein reported a case of AS-AIH triggered by SARS-CoV-2 infection, with clinical and pathological features that differed from those of conventional AIH. Early diagnosis and timely treatment with glucocorticoids are crucial for improving outcomes, especially avoiding liver transplantation. The main limitations of this case are that the follow-up time was relatively short and an immunosuppressant was not added. Further research on the relationship between SARS-CoV-2 infection and AIH is needed.
Footnotes
Acknowledgements
The authors thank the anonymous reviewers and the editor for their valuable comments.
Author contributions
YZ: Study design, drafting and detailed writing of the article, data analysis/interpretation, critical revision of the article, approval of the article, data collection.
QJ: Study design, drafting and detailed writing of the article, data analysis/interpretation, critical revision of the article, approval of the article, data collection.
CW: Study design, drafting and detailed writing of the article, data analysis, critical revision of the article, approval of the article.
XZ: Study design, data analysis, drafting of the article, critical revision of the article, approval of the article.
HL: Data interpretation, data collection, pathological guidance, approval of the article.
JB: Data interpretation, data collection, approval of the article, availability of data and materials.
Data availability statement
The corresponding author can be contacted with data requests.
Declaration of conflicting interests
The authors declare that there is no conflict of interest regarding the publication of this paper.
Ethics statement
All studies involving human participants were reviewed and approved by the research ethics committee of Hangzhou Xixi Hospital (approval no. 2023-00205829). Written informed consent for publication of her clinical details and clinical images was obtained from the patient.
Funding
This work was supported by the Zhejiang Provincial Medical and Health Science Project (No. 2023KY197) and the Zhejiang Provincial TCM Science and Technology Project (No. 2022ZB293).
