Abstract
Objective
To present a novel method that uses an epigenetic fingerprint to measure changes in plasma concentrations of cardiac-specific cell-free DNA (CS-cfDNA) as a marker of myocardial cell death.
Methods
This prospective, analytic, observational comparative study included patients with heart disease or multiple risk factors for heart disease undergoing major noncardiac, mostly vascular surgery, requiring an arterial-line, and at least 24 h hospitalization in the post anaesthesia care unit or critical care unit after surgery. Blood samples were collected at least four times per patient to measure troponin-T (via high-sensitivity troponin-T test) and CS-cfDNA pre- and postoperatively.
Results
A total of 117 patients were included (group 1, 77 patients [66%] with low preoperative and postoperative troponin-T; group 2, 18 patients [15%] with low preoperative but increased postoperative troponin-T; group 3, 16 patients [14%] with high troponin-T both preoperatively and postoperatively; and group 4, six patients [5%] with elevated preoperative troponin-T that decreased postoperatively). The increase in CS-cfDNA after surgery was statistically significant only in group 2, which correlated with an increase in troponin-T in the same group.
Conclusions
CS-cfDNA increased early postoperatively, particularly in patients with silent postoperative troponin elevation, and was correlated with an increase in troponin-T. These results may suggest that, in the subgroup of patients with postoperative elevated troponin, cardiomyocyte death indeed occurred.
Keywords
Introduction
Troponin is composed of three protein subunits regulating the contraction of skeletal and cardiac striated muscle, with this contraction mediated by calcium ions. Troponin has a significant role in the regulation of muscle stimulation and contraction. 1 There are three troponin isoforms (T, I, and C) with varying specificity for different tissues. Assays for cardiac troponin (isoforms I and T) are based on monoclonal antibodies interacting with specific epitopes found only on the surface of cardiac troponin. 1 This interaction gives rise to the test's high specificity. Typically, most troponin is tightly bound to contracting proteins, such as actin and myosin, but 2–6% of total troponin can be found in the cell cytoplasm. This ‘free’ troponin explains the claim of some researchers and clinicians that troponin can ‘leak’ from muscle cells following mild ischaemia and cause damage to cell membranes without actual irreversible damage to muscle cells. This theory may explain minor or mild blood troponin elevations that are above the normal range in healthy individuals. 2
The diagnosis of myocardial infarction requires an increase (and later decline) in troponin above normal values with accompanying symptoms (usually angina pectoris) and electrocardiogram changes. 3 One cause of troponin elevation above normal levels without clinical symptoms of ischaemia is silent troponin elevation (STE). STE is common in patients undergoing major surgery, hospitalized in the intensive care unit (ICU), or experiencing sepsis, trauma, or renal failure, etc. This phenomenon poses a diagnostic and therapeutic challenge, as previous studies have shown that elevated troponin predicts short- and long-term morbidity and mortality with or without symptoms suggestive of myocardial infarction, 4 and that most troponin elevations in patients undergoing major surgery or sepsis are asymptomatic.4–6
Regarding the analysis of cardiac-specific cell-free DNA (CS-cfDNA) in peripheral blood, an accurate method to identify the tissue source of these short DNA fragments in peripheral blood has been developed at the Hebrew University Medical School. This method is based on different methylation patterns in each tissue. In a recent paper, 7 the researchers showed that CS-cfDNA levels in patients hospitalized in the ICU with sepsis are highly increased and are similar to those of patients with ST-elevation myocardial infarction. They also found that an increase in CS-cfDNA predicts increased mortality in patients with sepsis (Figure 1). However, no correlation between troponin levels and CS-cfDNA was found. This finding might be explained by the differences in secretion kinetics of each of these markers. Because of the lack of evidence and clarity regarding the clinical and therapeutic significance of STE, there is a need for studies examining whether STE represents significant myocyte destruction.

Correlation between cell-free DNA and survival between septic and nonseptic patients (reproduced directly without changes from Zemmour et al. Nat Commun 2018; 9: 1443, under the Creative Commons Attribution 4.0 International License).
In a recent meta-analysis by Tan et al., 8 652 studies were analysed, and 17 were included in the results, which showed that plasma cfDNA emerges as a dependable indicator of myocardial injury, though the breadth of inferences drawn from current findings is constrained due to methodological variations.
The present authors hypothesized that increased CS-cfDNA in the blood is correlated with an increased level of troponin, and that it may be possible to define clinical circumstances and a threshold for troponin levels, from which elevated troponin has clinical significance. Another significance of the present study was to analyse if a correlation between cfDNA and troponin is well reflected by the complexity of surgical procedures.
The aim of the present study was to examine whether there is a correlation between perisurgical troponin and CS-cfDNA in patients undergoing noncardiac surgery, and to assess CS-cfDNA levels following complicated surgeries.
Patients and methods
This prospective, analytic observational comparative study was authorized by the institutional ethics committee of the Hadassah Medical Centre, The Hebrew University of Jerusalem (IRB approval licence No. 0442-16). The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2013. Written informed consent was obtained from all participants, and all patient details were de-identified. This manuscript adheres to the applicable EQUATOR guidelines, and the reporting of this study conforms to STROBE guidelines. 9
Study population
This study, conducted at Hadassah – Hebrew University Medical Centre, Jerusalem, Israel, included samples that were collected from sequential patients with known heart disease or multiple risk factors for heart disease (Revised Cardiac Risk Index [RCRI] > 2) who were candidates for major noncardiac surgery between April 2019 and October 2019. Additional criteria comprised: patients receiving predominantly vascular surgery and requiring an intra-arterial line for haemodynamic monitoring; and at least 24 h of hospitalization planned in the post-anaesthesia care unit or critical care unit. Exclusion criteria were as follows: paediatric patients (aged < 18 years); patients with RCRI ≤ 2; patients undergoing cardiac surgery without arterial cannula and with <24 h hospitalization time postoperatively.
Sample collection
Blood samples were collected from an arterial line at predefined times to quantify troponin T (via high-sensitivity troponin-T test conducted in the clinical laboratory of Hadassah – Hebrew University Medical Centre) and CS-cfDNA. Blood samples were collected prior to surgery, at 1 h postoperatively, and every 6 h thereafter until the morning after surgery or the removal of the arterial line (minimum of four samples per participant). No other element of patient care was changed or influenced by the researchers. Anaesthesia techniques for the procedures were chosen by the staff anaesthesiologist in charge of the case, and investigators did not influence this decision. As the study aim was to find the correlation between CS-cfDNA and troponin T levels, details of the anaesthesia technique and surgical technique, and minor complications, were not analysed and included in the results.
Analysis of cfDNA
Cell death is followed by the release of short fragments of DNA (usually approximately 163 base pairs) into the circulation 15–60 minutes before these fragments are cleared through the liver. Researchers at the Yuval Dor lab, Hebrew University Medical School, developed a method to identify tissue-specific cell death using a blood test. 7 The method is based on variability in DNA methylation, which is specific for each tissue. Methylation of DNA is used by cells to silence genes with unwanted activity in a specific tissue, while genes that are expressed do not undergo methylation. Measuring cfDNA enables not only detection of the cfDNA source, but also its absolute and relative quantification. Researchers have shown that cardiac cfDNA levels in healthy subjects are nonexistent to minimal, while in patients with myocardial infarction, cardiac cfDNA levels are increased and linearly correlate with cardiac troponin levels taken at the same time. Therefore, measuring cardiac cfDNA enables determination of the presence and extent of cardiac cell death. For example, the number of cardiac cfDNA fragments per ml of blood may be used to approximate the number of actual cardiomyocytes destroyed.
For the present study, blood samples were collected into ethylenediaminetetra-acetic acid tubes and centrifuged at 1500 g for 10 min at 4°C within 2 h of collection. Plasma was removed and recentrifuged at 3000 g for 10 min at 4°C to remove any remaining cells. Plasma was then stored at −80°C until assay. In the Dor lab (YD), cell-free DNA was extracted using the QIAsymphony SP instrument and its dedicated QIAsymphony Circulating DNA Kit (Qiagen, Ann Arbor, MI, USA) according to the manufacturer’s instructions. The DNA concentration was measured using the Qubit® dsDNA HS Assay Kit (Thermo Fisher Scientific, Waltham, MA, USA). cfDNA was treated with bisulfite using EZ DNA Methylation-Gold™ (Zymo Research, Irvine, CA, USA), and polymerase chain reaction (PCR) was performed with primers specific for bisulfite-treated DNA but independent of methylation status at the monitored CpG sites. Treatment with bisulfite led to degradation of 60–90% of the DNA (on average, 75% degradation). Note that while DNA degradation does reduce assay sensitivity (since fewer DNA molecules are available for PCR amplification), it does not significantly harm assay specificity as methylated and unmethylated molecules are equally affected. Primers were barcoded using TruSeq Index Adapters (Illumina, San Diego, CA, USA), allowing the mixing of samples from different individuals when sequencing PCR products using a NextSeq sequencer (Illumina). Sequenced reads were separated by barcode, aligned to the target sequence, and analysed using custom scripts written and implemented in R. Reads were quality filtered based on Illumina quality scores and identified by having at least 80% similarity to target sequences and containing all the expected CpGs in the sequence. CpGs were considered methylated if CG was read, and were considered unmethylated if TG was read. The fraction of unmethylated molecules in a sample was multiplied by the total concentration of cfDNA in the sample to assess the number of cardiac genome equivalents per ml of plasma. The concentration of cfDNA was measured prior to bisulfite conversion, rendering the assay robust to potential intersample fluctuations in the extent of bisulfite-induced DNA degradation.
Statistical analyses
Categorical variables are presented as n (%) prevalence, and quantitative variables are presented as mean ± SD. A need for 96 matched blood samples (troponin and cfDNA) was calculated in order to apply Student’s paired t-test with a significance of 5% and statistical power of 80%. We allowed for a 10% deviation between the ratio of positive troponin and cfDNA samples. Preoperative and postoperative troponin and cfDNA for each study group was compared using Student’s paired t-test. The correlation between each troponin and CS-cfDNA group was assessed using Spearman’s rank correlation coefficient (nonparametric correlation test). Statistical analyses were performed using IBM SPSS Statistics software (Armonk, NY, USA) and a P-value <0.05 was considered statistically significant.
Results
A total of 137 patients were assessed for the study, of whom, 20 were excluded. Twelve of the excluded patients did not meet the inclusion criteria, five declined to participate, and three were excluded due to loss of data. Thus, a total of 117 patients were included in the study: 76 (65%) were male and 41 (35%) were female; 80 patients (68%) underwent vascular surgery (carotid endarterectomy = 24, carotid artery stenting = 15, thoracic endovascular aneurysm repair = 10, other = 31); 11 underwent kidney transplantation; one underwent liver transplantation; one underwent combined liver and kidney transplantation; four underwent Whipple surgery; seven underwent colectomy, oesophagectomy, or explorative laparotomy; three underwent radical prostatectomies; two underwent radical cystectomy; one underwent partial nephrectomy; five underwent radical neck dissection or mandibulectomy/laryngectomy; one underwent lumbar spine surgery; and one underwent lung segmentectomy. Maximum estimated blood loss was 3200 ml from the combined liver and kidney transplantation.
The 117 included patients were divided into 4 groups based on their pre- and postoperative troponin T results (Figure 2). Group 1 included 77 patients (66%) with low preoperative and postoperative troponin levels. Group 2 included 18 patients (15%) with low preoperative but increased postoperative troponin levels. Group 3 included 16 patients (14%) with high troponin both preoperatively and postoperatively. Group 4 included six patients (5%) with elevated preoperative troponin that decreased postoperatively.

Troponin levels before and after surgery in 117 patients who underwent noncardiac surgery (Group 1, low preoperative and postoperative troponin levels; Group 2, low preoperative but increased postoperative troponin levels; Group 3, high preoperative and postoperative troponin levels; Group 4, elevated preoperative troponin levels and decreased postoperative troponin levels). T1, before surgery; T2, 1 h after surgery; T3, 6 h after surgery; and T4, 1 day after surgery. Normal cut-off, 33 ng/l. Data presented as mean ± SD.
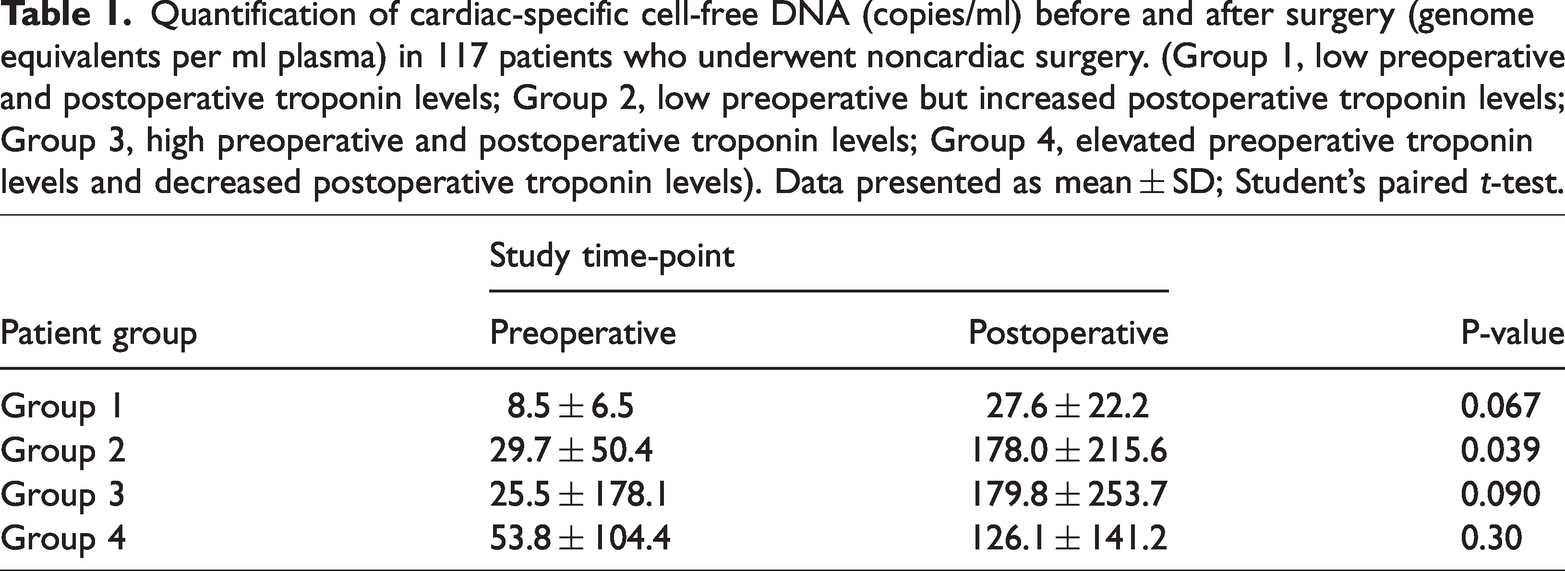
The increase in CS-cfDNA after surgery was statistically significant only in group 2 (P = 0.039), which was correlated with an increase in troponin in the same group (Table 1). Furthermore, in the three other groups, the CS-cfDNA results were increased postoperatively, but the increases were not statistically significant (P > 0.05). These insignificant results were also not correlated with the troponin results.
Quantification of cardiac-specific cell-free DNA (copies/ml) before and after surgery (genome equivalents per ml plasma) in 117 patients who underwent noncardiac surgery. (Group 1, low preoperative and postoperative troponin levels; Group 2, low preoperative but increased postoperative troponin levels; Group 3, high preoperative and postoperative troponin levels; Group 4, elevated preoperative troponin levels and decreased postoperative troponin levels). Data presented as mean ± SD; Student’s paired t-test.
Preoperative and postoperative CS-cfDNA for the entire cohort was compared using Student’s paired t-test. Postoperative CS-cfDNA was higher than preoperative CS-cfDNA, which was statistically significant (P = 0.0063; Figure 3).

Cardiac-specific cell-free (cf)DNA measured preoperatively and 24 h postoperatively in 117 patients who underwent noncardiac surgery. Data presented as mean ± SD.
The correlation between troponin and CS-cfDNA of each group was tested using Spearman’s rank correlation coefficient, and the correlation was only found to be statistically significant in Group 2 (P = 0.0339; Figure 4).

Correlation between postoperative increase in troponin and cardiac-specific cell-free DNA (CS cfDNA) levels in 117 patients who underwent noncardiac surgery. (Group 1, low preoperative and postoperative troponin levels; Group 2, low preoperative but increased postoperative troponin levels; Group 3, high preoperative and postoperative troponin levels; Group 4, elevated preoperative troponin levels and decreased postoperative troponin levels). Spearman’s rank correlation coefficient.
Discussion
Cardiac-specific troponins have long served as the gold standard for diagnosing myocardial damage. Nonetheless, it is essential to acknowledge their limitations. One primary challenge lies in distinguishing whether the release of a cytoplasmic protein, such as troponin, into the bloodstream indeed signifies cardiomyocyte death or rather reversible cellular injury. 10 Notably, elevated troponin levels have been documented in healthy individuals following strenuous exercise,11,12 as well as in certain critically ill patients within surgical or medical ICUs. In these cases, the origins of troponin elevations remain uncertain, leaving us to ponder whether they stem from myocardial cell death or reversible myocardial injury.13,14 Moreover, the presence of renal dysfunction may significantly slow the clearance of troponin, thereby complicating the interpretation of troponin elevations, particularly in patients with comorbidities, such as heart disease and renal impairment. 15
Adding to the complexity, we face the challenge of quantifying the number of injured cardiac cells in various clinical scenarios, given the uncertainty surrounding the quantity of protein molecules released from each damaged cardiomyocyte. The Dor laboratory at Hadassah Medical School, Hebrew University of Jerusalem, introduced a novel approach for identifying the origins of cfDNA by analysing tissue-specific methylation patterns within cfDNA.16–18 This method allows us to infer cell death in tissues that share a genome identical to that of the host. Consequently, a cfDNA biomarker for cardiomyocyte death presents exciting prospects for the diagnosis and monitoring of cardiac pathologies, as well as for advancing our understanding of normal human cardiac physiology and development. 7
The present study yields compelling evidence that CS-cfDNA levels increase postoperatively, particularly among patients in the present group 2, who exhibited low preoperative but increased postoperative troponin levels. Remarkably, in this subgroup of patients, a significant correlation emerged between troponin levels and CS-cfDNA, suggesting that elevated postoperative troponin levels, observed in approximately 15% of patients in the present study, indeed signify cardiomyocyte cell death. This discovery not only sheds light on a long-standing debate in the field, but also prompts a fresh set of inquiries concerning the factors driving cardiac cell death following surgery and its biological and clinical significance.
Cardiac cfDNA measurements have the potential to serve as a novel, highly specific marker for myocardial damage, possibly complementing the existing array of markers. One notable advantage of cfDNA is its rapid clearance, estimated to occur within 15–60 minutes, a considerably shorter timeframe than troponin. The time to first increase of cfDNA concentrations has been described as within 2 h from the onset of chest pain (similar to hs-cardiac troponin assays),7,19 whereas conventional cardiac troponin assays only detect rises 4–10 h after chest pain onset. 20 This implies that cardiac cfDNA may provide insights into the rate of cardiomyocyte death close to the time of sampling, complementing troponin's ability to indicate longer-term cardiac damage. Notably, cfDNA measurements hold promise in dynamic scenarios, such as cases involving sequential or ongoing myocardial insults.
As our understanding of methylation biology expands and DNA analysis technology advances, we anticipate significant enhancements in cardiac cfDNA measurements, including faster results, heightened specificity, and enhanced sensitivity.
Recognizing the time-sensitive nature of diagnostic applications in cardiology, a ddPCR version of the cardiac cfDNA assay has been developed, offering a turnaround time of 6.5–7 hours from blood collection to result retrieval. 7 Further optimization efforts could further reduce this time, and emerging technologies, such as nanopore sequencing, 21 hold the potential to enable near real-time measurement of cardiac cfDNA. Future investigations will delve into methylation signatures of cardiac subdomains and cellular subtypes, providing a means of noninvasively detecting damage in specific regions of the heart. In terms of sensitivity, while the current cfDNA assay appears slightly less sensitive than troponin, the possibility of multiplexing independent cardiac methylation markers may substantially boost sensitivity, increasing the likelihood of successfully capturing heart-derived DNA fragments.
In 2022, Brusca et al. 22 showed that circulating cfDNA is elevated in patients with pulmonary arterial hypertension, correlates with disease severity, and predicts worse survival.
The cardiac cfDNA assay promises to find extensive utility in advancing our understanding of human heart biology. For instance, it offers the opportunity to establish baseline levels of cardiac cell death throughout the human lifespan, from birth to old age, 23 facilitating more accurate models of human heart development.
The results of the present study may be limited by several factors. The study included a relatively small sample of 117 patients, which might affect the generalizability of the findings to a broader population. Also, the study was conducted at a single centre (Hadassah Medical Centre), which may limit the generalizability of the findings to other medical institutions with different patient demographics and practices. While the study found a correlation between troponin levels and CS-cfDNA in certain groups, the clinical significance of this correlation is not fully explored. More detailed clinical correlation and outcomes data would enhance the study's impact. Finally, the present study focused on short-term postoperative outcomes. Investigating the long-term implications of elevated CS-cfDNA levels would provide a more comprehensive understanding of its clinical significance. While these limitations exist, they don't necessarily diminish the value of the study but rather provide areas for consideration in future research and clinical applications.
Conclusion
With the findings of the present study, we may conclude that CS-cfDNA increases early after surgery, particularly in patients with silent postoperative myocardial troponin elevation. An increase in CS-cfDNA was also correlated with an increase in hs-troponin T results in patients with low hs-troponin T and low CS-cfDNA preoperatively. This suggests that the subgroup of patients with elevated troponin postoperatively experienced cardiomyocyte death as a consequence of surgery. More studies with larger patient populations are needed to determine if major surgeries indeed cause cardiac cell death, and to understand the biological mechanisms underlying the phenomenon and the clinical significance, as well as to ascertain whether cardiac cfDNA analysis can evolve into a useful biomarker.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241229638 - Supplemental material for The correlation between high-sensitivity troponin-T and cell-free cardiac DNA in the blood of patients undergoing noncardiac, predominantly vascular surgery
Supplemental material, sj-pdf-1-imr-10.1177_03000605241229638 for The correlation between high-sensitivity troponin-T and cell-free cardiac DNA in the blood of patients undergoing noncardiac, predominantly vascular surgery by Tural Alekberli, Braha Lea Ohana, Hai Zemmour, Rudy Khader, Ruth Shemer, Yuval Dor and Giora Landesberg in Journal of International Medical Research
Supplemental Material
sj-zip-2-imr-10.1177_03000605241229638 - Supplemental material for The correlation between high-sensitivity troponin-T and cell-free cardiac DNA in the blood of patients undergoing noncardiac, predominantly vascular surgery
Supplemental material, sj-zip-2-imr-10.1177_03000605241229638 for The correlation between high-sensitivity troponin-T and cell-free cardiac DNA in the blood of patients undergoing noncardiac, predominantly vascular surgery by Tural Alekberli, Braha Lea Ohana, Hai Zemmour, Rudy Khader, Ruth Shemer, Yuval Dor and Giora Landesberg in Journal of International Medical Research
Footnotes
Acknowledgement
In memory of an outstanding mentor, a determined and meticulous researcher, a visionary cardiovascular anaesthesiologist, a devoted family man, a great team player, and a wonderful friend – Professor Giora Landesberg.
Author contributions
TA: study design, data collection, statistical analysis, writing manuscript, BLO: cfDNA analysis, identification and quantification of cardiac cfDNA, HZ: cfDNA analysis, identification and quantification of cardiac cfDNA, RH: data collection, consent form preparation and collection, RS: data analysis, supervision of the findings, YD: data analysis, supervision of the findings, editing the manuscript, GL: idea presentation, study design, data collection, sample size calculation, statistical analysis, supervision of the project. We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us. We understand that the corresponding author is the sole contact for the editorial process. He is responsible for communicating with the other authors about progress, submissions of revisions, and final approval of proofs.
Data accessibility statement
The data supporting the findings of this study are available within the article and in supplemental material.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
