Abstract
Objectives
Patients with systemic lupus erythematosus (SLE) frequently show non-compliance with their medication. We evaluated the compliance of patients with SLE in Jordan with their medication and the relationships with fetal and maternal outcomes.
Methods
We performed a retrospective cohort study of patients with SLE who had no co-morbidities or antiphospholipid syndrome; and were taking only prednisolone, hydroxychloroquine, and/or antiplatelet and anticoagulant medication.
Results
We studied 173 pregnancies. Prednisolone was administered around pregnancy in 50 (28.9%) of these. The compliance with hydroxychloroquine, prednisolone, and anticoagulant and antiplatelet medication was 87.5%, 91.4%, and 97.3%, respectively. Non-compliance with anticoagulant/antiplatelet therapy was significantly associated with pregnancy-related complications. No complications developed in participants who were non-compliant with prednisolone therapy. The mean pre-pregnancy Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) for the pregnancies was 3.7, indicating low disease activity. Pregnancies with high pre-pregnancy SLEDAI scores tended to be more likely to have preterm deliveries, intrauterine growth restriction, and stillbirth. Postpartum relapse tended to be associated with higher pre-pregnancy SLEDAI.
Conclusions
In patients with pre-conceptional low SLE activity, changes in therapeutic compliance during pregnancy are not associated with adverse outcomes. In addition, post-partum relapse is not associated with pre-pregnancy SLEDAI score in therapeutically compliant patients.
Keywords
Introduction
Population-based studies have shown that patients with systemic lupus erythematosus (SLE) frequently discontinue their medication, and particularly antimalarial drugs, during pregnancy, and these findings imply that it is important to educate women with SLE who are pregnant or planning to become pregnant regarding the benefits and risks of taking particular drugs during pregnancy.1,2 Pregnancy results in a series of changes to the immune system and kidneys that may be associated with higher risks of a disease flare-up and adverse maternal and fetal outcomes, such as preeclampsia, fetal loss, and preterm delivery. 3 Therefore, conception should be delayed until the disease is in remission, to ensure the best pregnancy outcomes. 3 Maternal disease activity and fetal well-being should be monitored closely by a multidisciplinary team, including obstetricians, rheumatologists, and nephrologists throughout pregnancy. In particular, careful attention should be paid to the dose and potential teratogenicity of the medication. 3
Pregnancies in patients with SLE are considered to be high-risk. 4 However, patients are often not well counselled regarding the importance of maintaining disease remission in advance of pregnancy, or regarding the continuation of medication during and following pregnancy. Measures such as appropriate preconception counseling, adjustment of the medication, strict disease control prior to pregnancy, and intensive surveillance during and following pregnancy are essential for optimal pregnancy outcomes. 5
In the present study, we aimed to evaluate the relationship between compliance with treatment and fetal and maternal outcomes, and that between these changes in medication and the post-partum course of the disease.
Materials and methods
We performed a retrospective study of pregnancies in patients with SLE, which had been diagnosed according to the American College of Rheumatology (ACR) criteria 6 and confirmed by a consultant rheumatologist. The participants were followed by both a consultant rheumatologist and an obstetrician. Pregnancy-related data were obtained from the participants’ medical notes, both paper-based and electronic, antenatal records, rheumatology clinic notes, and delivery and postpartum records. The data collected included the patients’ numbers and ages, the duration of their SLE, their SLE medication before pregnancy (names of the drugs, doses, and frequency of administration), and whether the disease was active or not during the 6 months prior to pregnancy, assessed using the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI).
We studied the use of hydroxychloroquine (HCQ) 200 mg, prednisolone 40 mg, and antiplatelet (low-dose acetylsalicylic acid, 100 mg orally once daily) and anticoagulant (low-molecular-weight heparin (LMWH) (enoxaparin) 40 mg subcutaneously once daily) medication because these were the most commonly administered types. The use of antiplatelet and anticoagulant medication was investigated as a single drug category because most women with high-risk pregnancies in Jordan receive these drugs in combination. Aspirin is widely used by pregnant women in Jordan, whether it is prescribed by a physician or obtained by the women, even when their pregnancies are low-risk, although this practice is not based on evidence. The use of these medications before pregnancy, during pregnancy, and postpartum was recorded, as were the changes in their use. The data were then analyzed to identify the prevalences of the changes made and non-compliance.
Compliance with these medications was confirmed by examination of prescriptions and clinical notes, and the patients’ history. We did not evaluate serum drug concentrations because most of the participants had not had these concentrations measured. Any alterations that were made were not based on the recommendations of the treating physicians or the disease course. We then evaluated the relationship between these alterations and subsequent fetal and maternal outcomes (missed miscarriage [before 24 weeks of gestation], preterm delivery, intrauterine growth restriction [IUGR] and intrauterine fetal death [IUFD; fetal death after the completion of 24 weeks of gestation]). Missed miscarriage was defined as the loss of fetal cardiac activity of an otherwise normal fetus before the gestational age of viability (24 weeks), preterm delivery was defined as spontaneous delivery before 37 weeks of gestation, IUGR was defined as a fetal mass estimated by a consultant obstetrician to be below the 10th percentile for gestational age, and IUFD was defined as fetal death in utero of an otherwise normal fetus. Every pregnant woman at our hospital is offered an early ultrasonographic examination at 12 to 14 weeks of gestation and a detailed anatomic examination at 18 to 22 weeks of gestation. The SLEDAI of each participant 6 months before pregnancy was recorded and its relationships with the fetal and maternal outcomes listed above and postpartum relapse were evaluated. We divided the pregnancies into two groups: Group A, comprising pregnancies with inactive SLE and pre-pregnancy SLEDAIs of 0 to 3; and Group B, with active SLE and pre-pregnancy SLEDAIs of 4 to 18.
The study was performed at a single center (a teaching and referral university hospital between September 2016 and January 2021). All the pregnancies of women with confirmed SLE and a complete dataset were included. We excluded women with an unconfirmed diagnosis of SLE; those with missing data; and those taking drugs other than prednisolone, HCQ, and antiplatelet and anticoagulant drugs; those with multifetal pregnancies; and those with other diseases, including diabetes mellitus, hypertension, thyroid disorders, anti-phospholipid antibody syndrome (APAS), cardiac disease, deep vein thrombosis (DVT), pulmonary embolism, and associated hematological diseases, including SLE-associated thrombocytopenia. None of the women studied had been prescribed other immunosuppressive agents. These exclusions permitted us to assess the relationships of the changes in the medication of patients with SLE alone and no confounding risk factors with adverse outcomes of pregnancy. We did not specify the clinical features (renal, joint, hematologic, or dermatologic) or serologic (anti-DNA antibodies or complement) characteristics of the participants because the changes made to medication did not necessarily relate to the clinical, biochemical, or hematologic features of the disease. Instead, the patients discontinued their medication or were otherwise non-compliant, rather than clinicians making the changes on the basis of clinical or laboratory indications. Therefore, the aim of the present study was to investigate the effects of these alterations.
SPSS version 16 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Continuous data are presented as mean ± standard deviation (SD), and categorical data are presented as count and percentage. To evaluate the relationships between SLE therapies and continuous variables, we used analysis of variance, and the relationships between SLE therapies and categorical variables were evaluated using the chi-square test. P < 0.05 was considered to indicate statistical significance. The sample size was selected to identify differences in the primary outcomes of miscarriage, preterm birth, IUFD, and IUGR between the groups.
Ethics approval was obtained by the institutional review board of Jordan University Hospital (approval number 228/2019, date 24/9/2019). The requirement for patient consent was waived because the study was a retrospective review of medical records and involved the preservation of anonymity during data collection, analysis, and manuscript writing. The study was conducted according to the principles of the World Medical Association Declaration of Helsinki. The study was registered at ClinicalTrials.gov with the unique identifier NCT05176041. The data will be made available by the corresponding author upon reasonable request. The reporting of the study conforms to the STROBE guidelines. 7
Results
We identified 223 pregnancies in 156 women with SLE, of which 173 were included in the study. The mean age of the women during these pregnancies was 31 years (interquartile range [IQR] 27 to 35 years), their mean parity was 1.8 (IQR 1 to 3), and their mean SLEDAI was 3.7 (IQR 2 to 6) (Table 1). Fifty pregnancies were excluded were because of missing data (30), multifetal gestation (8), diabetes (5), uncertain diagnosis (5), cardiac disease (1), and confirmed DVT (1).
Clinical characteristics of the pregnancies included in the study (n = 173).
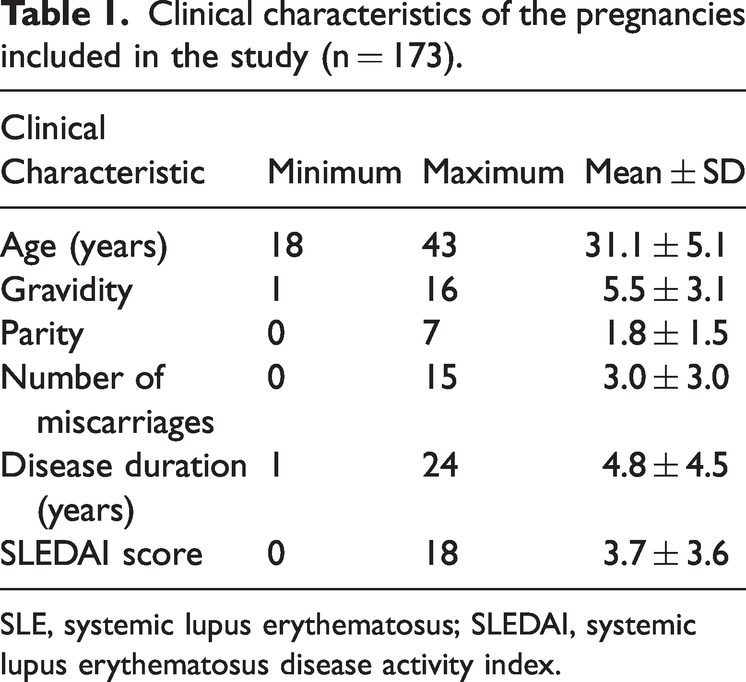
SLE, systemic lupus erythematosus; SLEDAI, systemic lupus erythematosus disease activity index.
One participant had renal involvement, indicated by substantial proteinuria. The majority of patients (52%) had urinary casts and/or hematuria, and 34% had arthritis. Approximately 20% of the participants had a combination of manifestations (headache, myositis, new-onset rash, eye symptoms, and ulcers). However, none of the participants had seizures, psychosis, organic brain syndrome, cranial nerve involvement, or cerebrovascular accidents.
Of the total number, 40 (23.1%) pregnancies were associated with HCQ administration, and most often (22 pregnancies, 12.7%) this continued throughout the pre-pregnancy, pregnancy, and postpartum periods. None of the participants commenced HCQ administration and only administered this drug during their pregnancy. However, it was initiated postpartum during eight (4.6%) pregnancies, owing to relapse (Table 2).
Alterations to the administration of therapeutic agents before and during pregnancy and postpartum associated with the included pregnancies.
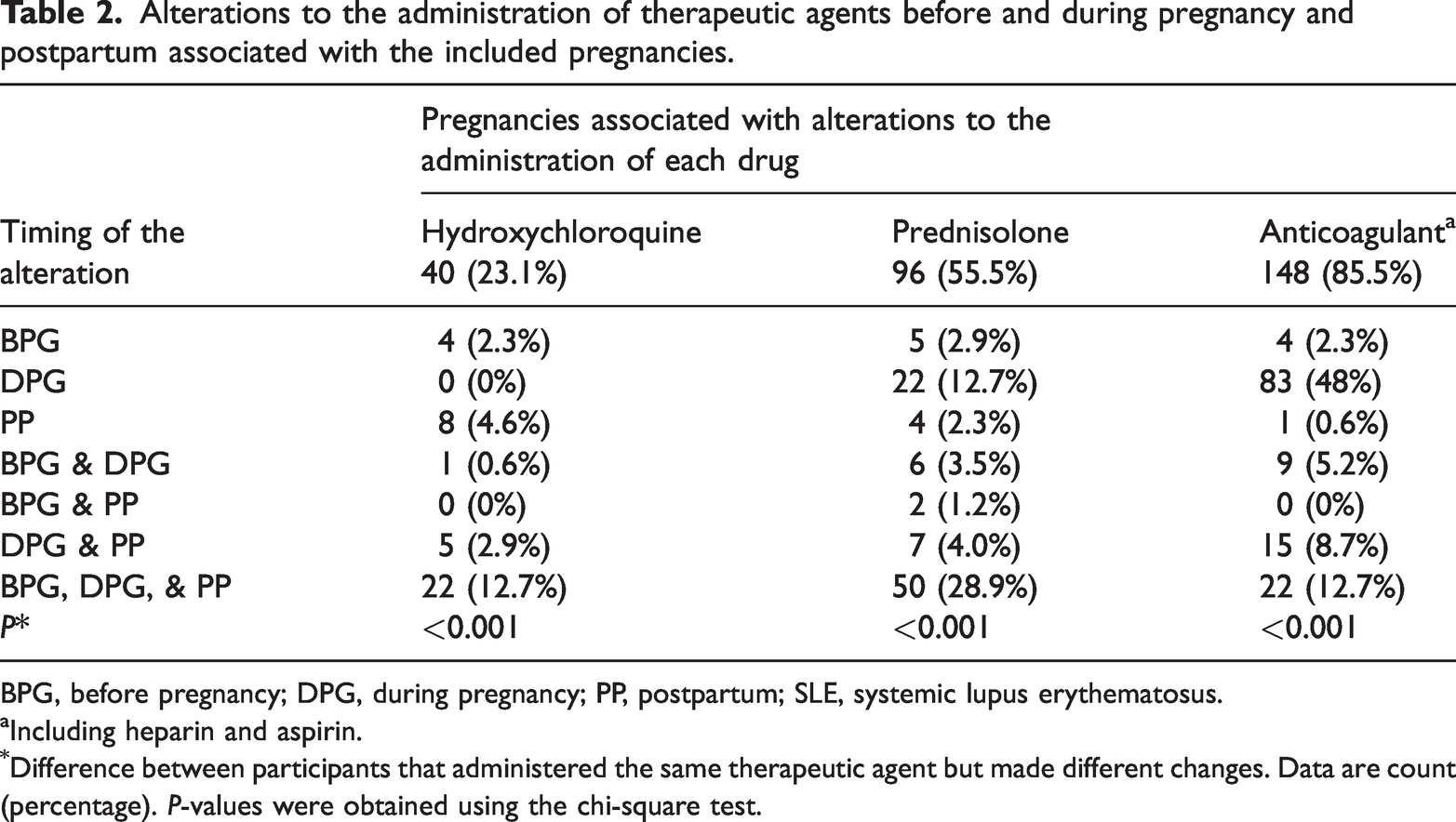
BPG, before pregnancy; DPG, during pregnancy; PP, postpartum; SLE, systemic lupus erythematosus.
Including heparin and aspirin.
Difference between participants that administered the same therapeutic agent but made different changes. Data are count (percentage). P-values were obtained using the chi-square test.
Prednisolone was administered before, during, and following pregnancy in association with 50 (28.9%) pregnancies. In approximately 13% of the total number of pregnancies (n = 22), prednisolone was administered only during pregnancy, at a mean dose of 30 mg orally daily. Its administration was also commenced postpartum at an oral dose of 80 mg daily following four pregnancies (Table 2). The participants themselves made the decision to stop or alter the administration of most of the medications during the study period, without medical advice, because they were concerned about the risks of these medications for the fetus.
All the participants who were taking antiplatelet medication (low-dose aspirin) were also taking low-molecular-weight heparin (enoxaparin), but none were taking warfarin. Anticoagulant and antiplatelet administration was started during 83 (48%) pregnancies, but these drugs were commenced before pregnancy in only four instances and postpartum in only one instance (Table 2).
Further analysis showed that approximately 62% of the non-compliance was during the first trimester of pregnancy.
Table 3 shows the timing of the use of medication during pregnancy with respect to complications of pregnancy; there were no significant associations between the incidences of the complications and the use of the three categories of medication.
Complications of pregnancy according to the therapeutic agents administered and the alterations made before and during pregnancy and postpartum.

BPG, before pregnancy; DPG, during pregnancy; IUFD, intrauterine fetal death; IUGR, intrauterine growth restriction; NA, not applicable; PG, pregnancy; PGC, pregnancy outcome; PP, postpartum; SLE, systemic lupus erythematosus.
1, Miscarriage; 2, Preterm delivery; 3, IUGR; 4, IUFD; 5, Preterm delivery and IUGR.
Including heparin, aspirin, and/or warfarin.
*P-value indicating the relationship between a complication and a specific medication.
**P-value indicating the relationship between a specific complication and a specific medication.
***P-value indicating the relationship between complications and a specific medication when different alterations were made before and during pregnancy and postpartum.
****P-value indicating the relationship between a specific complication in participants that made different alterations to this medication before and during pregnancy and postpartum. Data are count (percentage).
P-values were obtained using the chi-square test.
We also assessed the compliance with treatment of the participants, and found that most continued to administer the medication that they were administering before becoming pregnant, including HCQ, prednisolone, and anticoagulant/antiplatelet therapy (87.5%, 91.4%, and 97.3%, respectively). In addition, there was significant compliance with all three categories of medication (P < 0.001 for all) (Table 4).
Compliance with the treatment of SLE involving the administration of multiple medications.

SLE, systemic lupus erythematosus.
Including heparin, aspirin, and/or warfarin. Data are count (percentage).
*P-value indicating compliance with a specific medication before and/or during pregnancy (chi-square test).
Non-compliance with anticoagulant/antiplatelet therapy was significantly associated with the overall prevalence of pregnancy-related complications (P = 0.034), but not with the prevalences of individual complications, such as preeclampsia, probably because of the very low prevalence of non-compliance. Indeed, the compliance with aspirin treatment was very high (98.2%). Non-compliance with HCQ therapy was not found to be significantly associated with complications of pregnancy, and no complications developed in participants who were not compliant with their prednisolone therapy (Table 5).
Relationships of non-compliance with the treatment for SLE with complications of pregnancy

IUFD, intrauterine fetal death; IUGR, intrauterine growth restriction; SLE, systemic lupus erythematosus.
Including heparin, aspirin, and/or warfarin. Data are count (percentage).
*P-value indicating the relationship between all complications and non-compliance with a specific medication.
**P-value indicating the relationship between a specific complication and non-compliance with a specific medication.
P-values were obtained using the chi-square test.
The mean pre-pregnancy SLEDAI associated with the studied pregnancies was 3.7, which is indicative of low disease activity for most of the participants. Although not statistically significant compared with pregnancies associated with low scores, those associated with higher pre-pregnancy SLEDAIs resulted in higher incidences of preterm delivery, IUGR, and IUFD. In addition, postpartum relapse tended to be associated with a higher pre-pregnancy SLEDAI (Table 6). There were 115 cases of preeclampsia associated with the studied pregnancies, and 80% of these developed between 28 and 34 weeks of gestation. However, preeclampsia was not significantly associated with the levels of compliance with the drugs, In addition, there were no differences in the incidences of relapse or flare-up associated with the levels of compliance with HCQ or prednisolone therapy.
Pregnancy and delivery data for pregnancies, according to the associated SLEDAI score.
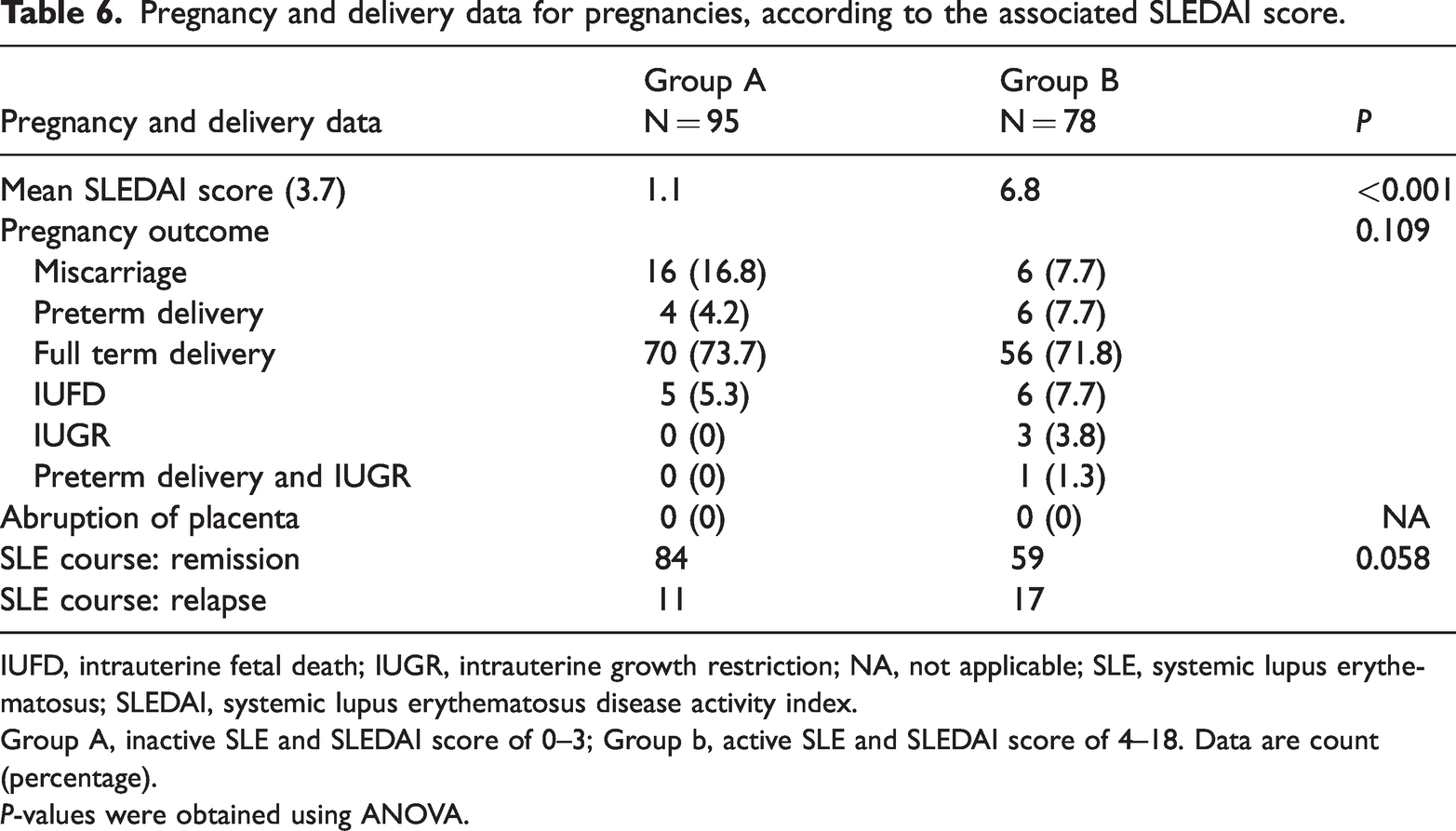
IUFD, intrauterine fetal death; IUGR, intrauterine growth restriction; NA, not applicable; SLE, systemic lupus erythematosus; SLEDAI, systemic lupus erythematosus disease activity index.
Group A, inactive SLE and SLEDAI score of 0–3; Group b, active SLE and SLEDAI score of 4–18. Data are count (percentage).
P-values were obtained using ANOVA.
Discussion
Pregnancy in women with SLE remains high-risk, with significant incidences of maternal and fetal complications. 8 However, high incidences of unplanned pregnancy and flare-ups of SLE were identified during pregnancy in patients with active SLE at conception were identified in a study conducted in Malaysia. 7 In this study of 115 pregnancies, the mean age of the participants was 29.1 years (SD 5.80) and they had a mean duration of disease of 44.63 months. 8 In the present study, the mean age of the participants was 31.1 ± 5.1 years, and they had a relatively low parity (mean 1.8 ± 1.5) compared with the expected parity for women in Jordan.
We did not investigate associations with whether the pregnancies in the present study were planned or not because such data are not usually recorded at our center. Family planning should be discussed as early as possible after a diagnosis of SLE is made, to institute measures that can reduce the risks of adverse maternal or fetal outcomes. 9 Preconception counseling is essential to ensure optimal pregnancy outcomes through careful risk stratification that takes into account disease activity, organ involvement, autoantibody profile, the use of drugs, and previous pregnancy outcomes. 10
Despite relatively similar maternal age and disease duration, the overall incidence of fetal and maternal complications in the present study was much lower than that previously reported. In one study published in 2011, 32.7% of the participants experienced SLE flare-ups, 17.3% developed preeclampsia, 20.0% experienced fetal loss, and 17.8% experienced preterm delivery. 11 In contrast, the pre-pregnancy SLEDAI of the present patients was 3.7 ± 3.6, implying that most had low disease activity.12–15
Despite most of the participants having low disease activity, HCQ was administered during 23.1% of the pregnancies in the present study, and during 87.5%, there was compliance during all the stages of the study, implying that this drug is perceived to be safe by those who used it and important to administer for the management of SLE around pregnancy. This was likely an important factor in the low incidences of fetal and maternal complications and disease flare-ups identified in the present study. HCQ use has several benefits, not only in patients with mild SLE, but also in patients with SLE and organ involvement, and in pregnant women. 16 The achievement of pre-pregnancy remission and the maintenance of HCQ administration during pregnancy have previously been shown to be important for the prevention of flare-ups. 17 Furthermore, the outcomes of pregnancy in patients with SLE can be improved by HCQ, as previously demonstrated by an approximate 90% reduction in the incidence of preeclampsia. 18 In addition, another study showed that pregnant patients with SLE tended to experience preeclampsia and preterm delivery less frequently when they were treated using HCQ. 19 However, a further study showed that HCQ treatment was safe, but did not significantly reduce the incidence of preeclampsia during pregnancy in patients with SLE. 20 A previous meta-analysis showed that HCQ treatment during pregnancy reduced the risks of preeclampsia, pregnancy hypertension, and prematurity in patients with SLE. However, the majority of these studies were retrospective and not randomized controlled trials. Nevertheless, it has been recommended that pregnant patients with SLE should be under multidisciplinary management and take HCQ, irrespective of their level of disease activity or the severity of the disease. 21 The prescription rate for HCQ in the present study was low because it seemed that the participants were concerned about its safety during pregnancy and possible effects on the fetus.
Although we excluded patients with APAS, prednisolone was administered throughout the study period in association with 28.9% of the pregnancies, and in approximately 13% of instances it was started during pregnancy. In a previous study that included patients with APAS, prednisolone was found to increase the incidence of live births, although it was also a predictor of prematurity.22,23 In addition, in patients with recurrent complications of pregnancy, an increase in heparin dose to the therapeutic level and the addition of HCQ or low-dose prednisolone during the first trimester may be considered. 24 In the present study, the participants had experienced a mean of 3.0 miscarriages (range 0 to 15). Therefore, the use of a combination of prednisolone and anticoagulants (both low-dose aspirin and LMWH) during the studied pregnancies could have contributed to the low incidence of complications.
In the present study, the administration of anticoagulants was started during pregnancy in association with 48% of the pregnancies, and in only four instances it was already being used before pregnancy and was stopped during and following pregnancy. In addition, anticoagulants were initiated postpartum in only one instance. There was 97.3% compliance with anticoagulant therapy. The results of a previous study provided support for the use of low-dose aspirin to prevent preeclampsia in patients with SLE. 25 In another previous study that compared the use of LMWH and aspirin in patients with or without APAS, it was shown that maternal and perinatal complications occurred frequently despite the use of these drugs. However, their use was associated with a significant reduction in the incidence of preterm delivery and no significant effect on hypertensive disorders of pregnancy. 26 Furthermore, another study showed that LMWH and aspirin might contribute to endothelial dysfunction in women with obstetric APAS. 26 The use of a combination of heparin and aspirin during the course of pregnancy may increase the live birth rate of women with persistent antiphospholipid antibodies versus aspirin treatment alone. 27 However, more research is needed in this area to further evaluate the potential risks and benefits of this treatment strategy, especially for women with antiphospholipid antibodies and those who have previously experienced pregnancy loss. 27 The use of aspirin and LMWH in pregnancy is widespread in Jordan, by women with both high- and low-risk pregnancies. Indeed, it is very common for primigravid women with no obstetric or medical risk factors to administer both anticoagulants.
With the exception of anticoagulants, the use or alterations in the use of the studied therapeutic agents was not found to be significantly associated with complications of pregnancy. This was principally because the comparator group consisted of participants who had low disease activity prior to pregnancy, made minor changes to their therapy, and complied well with their medication. In contrast, non-compliance with anticoagulant therapy in the present study was significantly associated with the overall incidence of pregnancy complications (miscarriage, preterm delivery, IUGR, and IUFD) (P = 0.034). Positive lupus anticoagulant tests and active disease prior to pregnancy were previously found to be strongly associated with adverse pregnancy outcomes in a multi-ethnic cohort of patients with SLE. 28 Because anticoagulant therapy is principally indicated in patients with APAS, further research is necessary regarding the effects of anticoagulants on fetal outcomes in patients who do not have APAS.
Our hospital uses the SLEDAI-2000 score to assess pre-conceptional disease activity; however, the SLE Activity Measure-Revised (SLAM-R), the Mexican SLE Disease Activity Index (MEX-SLEDAI), and the Modified SLEDAI-2000 have been found to be adequate for the assessment of SLE disease activity, and they are also less expensive than the SLEDAI-2000. 29 Although others 30 have shown that the SLE Disease Activity Score (SLE-DAS) has good construct validity and performs better than the SLEDAI-2000 for the identification of clinically significant changes in disease activity and for the prediction of damage accrual, we used the pre-pregnancy SLEDAI-2000 in the present study because the participants were relatively young and had no significant organ damage. We found a trend toward a higher incidence of pregnancy complications in participants with higher pre-pregnancy SLEDAI, but this relationship was not statistically significant, probably because the pre-conceptional disease activity of the participants was low, the participants were relatively young, and there was a high level of compliance with HCQ therapy. Postpartum disease relapses were more common in instances of high pre-pregnancy SLEDAI. Active disease at conception has previously been shown to be an independent predictor of SLE relapse during and following pregnancy, whereas older mothers and Malay ethnicity were shown to be associated with a higher incidence of flare-ups postpartum. 31 HCQ use was also shown to be significantly associated with a lower risk of flare-ups. 32 These findings are consistent with the present findings. In another study, 33 active nephritis was shown to be associated with poor pregnancy outcomes, fetal loss, and predictors of adverse maternal outcomes. The predictors of adverse fetal outcomes have been shown to comprise APAS, preeclampsia, the presence of anti-Smith antibody, a history of neuropsychiatric SLE (NPSLE), and azathioprine use.34,35 In the present study, the exclusion of patients with APAS and other antibodies could have contributed to the low incidences of complications and postpartum relapse.
The present study had several limitations. First, it was a retrospective study conducted at a single center. Second, we could not study the exact reasons for changes in the administration of medication or the recommendations of the consultant rheumatologist and the obstetricians because these changes were made by the participants themselves. Third, certain organ manifestations of SLE are associated with a high APO ratio; for instance, renal manifestations are associated with higher incidences of fetal loss and preeclampsia. 32 However, we did not characterize the specific organ manifestations of the participants. Fourth, we did not collect data regarding antibody titers related to SLE (anti-Sjögren’s Syndrome A and lupus anticoagulant) that have close relationships with adverse pregnancy outcomes.35,36 Finally, we did not investigate whether the included pregnancies were planned or not.
Conclusions
In patients with SLE and pre-conceptional low disease activity, changes in therapy around pregnancy were not found to be significantly associated with adverse fetal or maternal outcomes. In addition, postpartum relapse was not found to be significantly associated with the pre-pregnancy SLEDAI score in young patients who complied well with their medication. However, large prospective multicenter trials are needed to confirm these findings.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231225349 - Supplemental material for Therapeutic changes of systemic lupus erythematosus (SLE) patients in pregnancy and feto-maternal outcomes: a retrospective cohort study
Supplemental material, sj-pdf-1-imr-10.1177_03000605231225349 for Therapeutic changes of systemic lupus erythematosus (SLE) patients in pregnancy and feto-maternal outcomes: a retrospective cohort study by Naser Al-Husban, Diala Walid Abu-Hassan, Ro’ya Ali Abu Saleem, Lara Al-Abdallat, Alhareth Eid Alhusban and Marwan H Adwan in Journal of International Medical Research
Footnotes
Author contributions
NAH: concept, design, data analysis, discussion, writing and revision of the manuscript. DWAH: statistics, results, data analysis, and writing. RAAS: data collection. LAA: data collection and revision. AEA: data collection and revision (data analysis, results, and discussion). MHA: analysis, discussion, and writing of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
