Abstract
Background and aim
Acute kidney injury (AKI) affects a significant number of patients and the prognosis for this condition remains poor. The aim of this study was to assess adherence to KDIGO clinical practice guidelines and identify areas for improvement.
Methods
For this retrospective study, data were extracted from the medical database of the University Hospital Brandenburg, for patients who had been diagnosed with AKI from January to March 2021. Implementation rates of eight KDIGO AKI therapeutic measures were analyzed in relation to several AKI severity/risk categories.
Results
Data from 200 patients were included in the study. Three specific measures were commonly implemented: hyperglycemia control (100%), volume therapy (82%), and fluid balance management (65%). Nephrotoxic medications were discontinued in 51% patients, while iodinated contrast media was used in 35% patients. Patients with an increased risk of complications, such as those requiring ICU therapy or with sepsis, received these measures more frequently.
Conclusions
While some 2012 KDIGO recommended measures were implemented for a substantial number of affected individuals, others were not. Our study highlights the need for improvement in the quality of care for patients with AKI.
Introduction
Acute kidney injury (AKI) affects a significant number of patients receiving in-hospital treatment, with reported incidences of up to 30%.1,2 The risk is further increased in intensive care unit (ICU) patients, as approximately 50% experience a sudden decline in kidney function during therapy. 3 The in-hospital mortality rate associated with AKI varies from approximately 10% to over 60%. 2 Importantly, patients with hematological cancer, sepsis caused by chemotherapy, and AKI requiring kidney replacement therapy have been reported to have an almost 100% mortality rate. 4
Currently, the diagnosis of AKI is based on the 2012 revised Kidney Disease Improving Global Guidelines (KDIGO) for AKI, which considers changes in serum creatinine and/or urine output as measures of severity. 5 It is hoped that new diagnostic criteria will be published soon, which may include new biomarkers of kidney damage, such as neutrophil gelatinase-associated lipocalin (NGAL) and kidney injury molecule-1(KIM-1).6–8
The 2012 guidelines recommend preventive and therapeutic measures for patients with emerging or established AKI. 5 These measures aim to either reduce the risk or attenuate the course of the disease. However, their effectiveness relies heavily on the frequency of their implementation in daily critical care practice and the awareness of them by practicing physicians. Interestingly, in a cross-sectional survey conducted in 2020 at Omdurman Military Hospital in Sudan, data showed that only 5% of the 169 physicians had reasonable AKI-related practices. 9 Similarly, data from another study, conducted in Nigeria in 2017, showed that only 1% of 82 physicians had good knowledge of AKI. 10 The poor compliance with the 2012 guidelines is worldwide. For example, in a study conducted in 2014 at two large acute teaching hospitals in the UK serving a combined population of over 1.5 million people, only 26% of all in-hospital physicians were aware of the local AKI guidelines. 11 In addition, data from a UK study conducted in 2013 that analysed outcome and healthcare quality variables for approximately 1600 patients with AKI who were admitted to a single centre university hospital found that AKI was common in hospitalized patients and associated with a significant increase in hospital stay and mortality. 19 Alarmingly, AKI was unrecognized in 24% patients, two-thirds of whom were discharged without resolution of renal function. Moreover, fluid therapy documentation was inadequate and 40% cases did not have nephrotoxic medications discontinued.
In the current investigation, we aimed to evaluate the frequencies by which individual preventive/therapeutic measures according to KDIGO guidelines were implemented in our institution. 5 The purpose of the analysis was to identify gaps in the quality of care for patients with AKI, with the goal of enhancing the quality of future overall management.
Methods
This was a single centre, retrospective study. All data were obtained from the medical database of the University Hospital (MEDICO CGM®) Brandenburg, Germany for patients who had been diagnosed with AKI from January to March 2021. Patients were consecutively included if they met either criterion 1 or 2 of the 2012 KDIGO guidelines for AKI (i.e., serum creatinine concentration increases by ≥0.3 mg/dl within 48 hours or, creatinine concentration increases by ≥1.5-fold within 7 days) and were aged 18 years or over. 5 Patients with stage 5 chronic kidney disease were excluded.
According to the 2012 KDIGO guidelines for AKI, eight therapeutic measures that should be implemented in patients who are at risk of, or, have established AKI are as follows: (1) medication dosage adjustment based on renal function; (2) hemodynamic monitoring; (3) volume administration; (4) hyperglycemia control; (5) avoidance of iodinated contrast media; (6) discontinuation of nephrotoxic drugs; (7) subclavian access if central venous line is indicated; (8) fluid balance control.
In our current investigation, a specific measure was considered fulfilled (i.e., a ‘YES’ response) if the following criteria were met:
the dosage of at least one type of drug was adapted according to kidney excretory function; patients were provided with an intra-arterial blood line for continuous blood pressure (BP) measurement; intravenous infusion of at least 500 ml of a crystalloid fluid preparation within 24 hours after AKI onset; subcutaneous or intravenous administration of fast-acting insulin in response to elevated plasma glucose levels at least once after AKI onset; administration of any type of iodinated contrast media in a systemic fashion after AKI onset; discontinuation of the following substances: aminoglycosides, non-steroidal anti-inflammatory drugs (NSAIDs), vancomycin; subclavian access as necessary. documentation of fluid intake and output taken on a daily basis.
If a certain measure was not mandatory, (e.g., the insertion of a subclavian venous line or nephrotoxic drug discontinuation), the measure was judged as not applicable (N/A).
The implementation rates of the eight KDIGO AKI therapeutic measures were analyzed in relation to six AKI severity/risk categories. These six categories were: AKI stage according to KDIGO; requirement for ICU therapy; development of sepsis during in-hospital stay; requirement for kidney replacement therapy (KRT); pre-existing heart failure; pre-existing diabetes mellitus.
The reporting of this study conforms to STROBE guidelines. 12 The study obtained formal approval from the Ethics Committee of the Brandenburg Medical School. (August 5, 2022, reference number E-01-20220621). Written informed consent was not required due to the retrospective design of the study and patient data were anonymized prior to analysis.
Statistical analysis
The analysis was performed using WIZARD application for MacOS (version 2.0.11, developed by Evan Miller) and P < 0.05 was considered statistically significant. Descriptive statistics were used to describe the demographics and clinical characteristics of the included patients. All categorical variables were analysed using Pearson's χ2 test.
Results
Patients
In total, 200 patients were included in the study, of whom 39% (78) were female and 61% (122) were male (Table 1). Mean ± SD age of the group was 75 ± 11 years and duration of in-hospital treatment was 16 ± 11 days. According to the KDIGO criteria, the identified stages of AKI were as follows: stage 1, 36%; stage 2, 27%; stage 3, 38%. The most common individual cause of AKI was sepsis (21%), followed by pre-renal AKI (15%). In 41% of the patients, AKI occurred due to a combination of two or more causes. During hospital stay, 46 (23%) patients received at least one kidney replacement therapy (KRT) session. At discharge from the hospital, 23 (12%) patients still required KRT.
Demographic data.

Data are expressed as mean ± standard deviation, or n (%).
Abbreviations: AKI, acute kidney injury; COPD, chronic obstructive pulmonary disease; ICU, intensive care unit; KRT, kidney replacement therapy.
Analysis of data from the eight therapeutic measures from the 2012 KDIGO recommendations that were implemented across the 200 patients showed the following: medication dosage adjustment (yes 68%, no 33%); hemodynamic monitoring (yes 39%, no 61%); volume administration (yes 82%, no, 19%); hyperglycemia control (N/A 56%, yes 44%); avoidance of iodinated contrast media (yes 35%, no 65%); discontinuation of nephrotoxic drugs (yes 51%, no 37%, not applicable, 13%); subclavian access if indicated (yes 5%, no 35%, N/A 61%); fluid balance control (yes 65%, no 35%) (Figure 1). Since all patients requiring glucose normalization were already receiving insulin, the measure ‘hyperglycemia control' was not included in the final analysis.
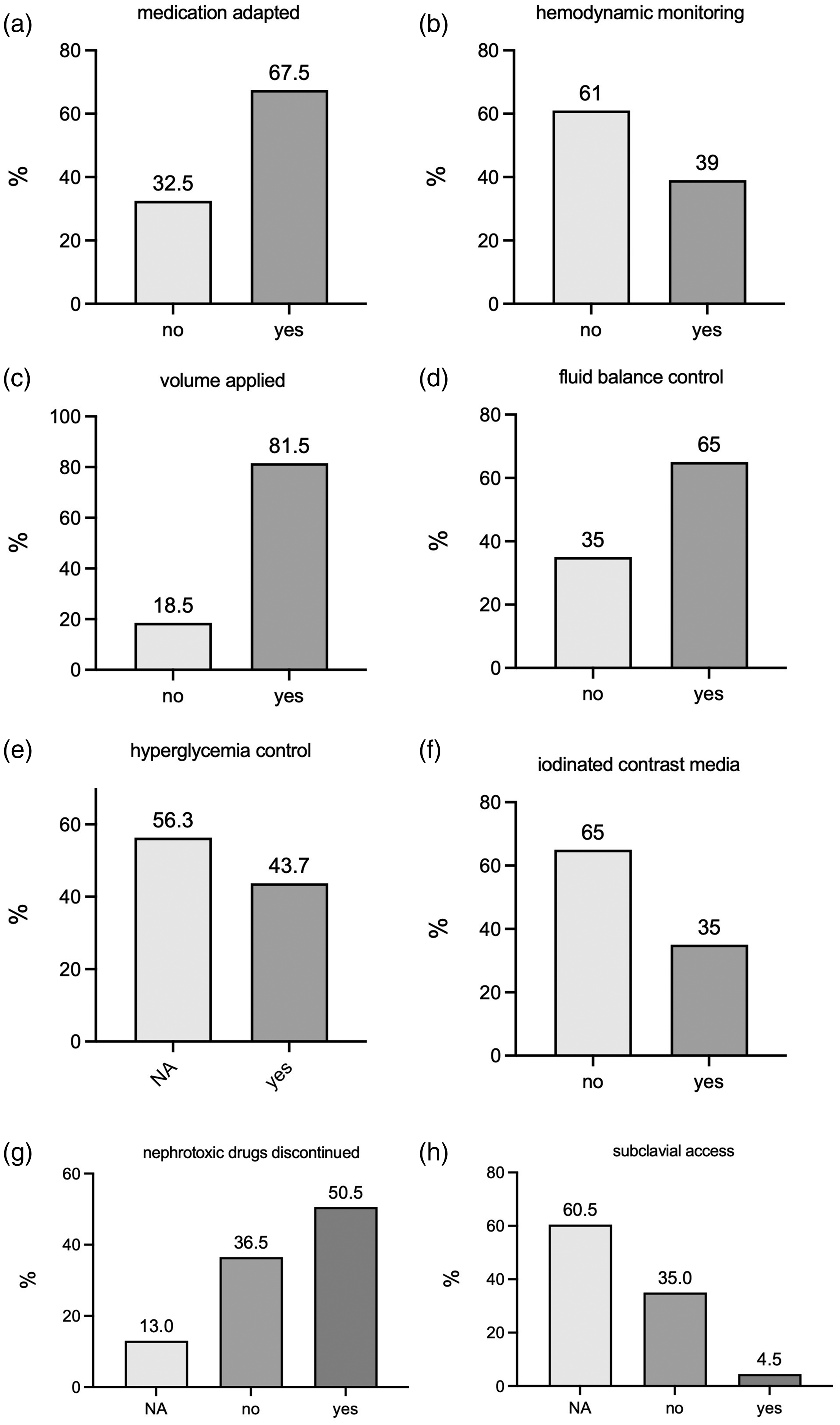
Implementation rates of eight measures listed in the 2012 Kidney Disease Improving Global Guidelines (KDIGO) for acute kidney injury (AKI).
Implementation rates of the final seven KDIGO AKI measures were assessed in relation to six AKI severity/risk categories (Table 2). According to the KDIGO AKI stages, four measures showed increasing prevalence from stage 1 to 3. These were: hemodynamic monitoring (P = 0.018); volume administration (P = 0.04); discontinuation of nephrotoxic drugs (P = 0.002); subclavian access if indicated (P = 0.03).
Implementation of seven KDIGO AKI preventive/therapeutic measures according to six risk/severity categories.

Abbreviations: AKI, acute kidney injury; ICU, intensive care unit; KDIGO, Kidney Disease Improving Global Guidelines; KRT, kidney replacement therapy ns, not statistically significant.
Sepsis that developed in hospital.
For patients in ICU (n = 71), except for dosage adaptation and discontinuation of nephrotoxic drugs, all measures were significantly more implemented in these patients than in non-ICU patients. For patients with sepsis that developed in hospital (n = 52), four measures were implemented significantly more than in non-sepsis patients. These were: hemodynamic monitoring (P < 0.001); volume administration (P = 0.007); fluid balance control (P = 0.016); subclavian access if indicated (P = 0.005) (Table 2).
Regarding patients who required KRT (n = 46), only two measures were established more often than in non-KRT patients: dosage adaptation (P = 0.013) and discontinuation of nephrotoxic drugs (P = 0.006). For patients who had pre-existing heart failure (n = 111), with the exception of volume administration (P = 0.04) which was used less often in this group than in patients without heart failure, there were no difference in the implementation categories. In diabetic patients (n = 90), two measures were implemented more frequently than in non-diabetic patients; these were fluid balance control (P = 0.005) and discontinuation of nephrotoxic drugs (P = 0.002) (Table 2).
We conducted a comprehensive analysis comparing the impact of implementing the seven KDIGO AKI measures on three important outcome variables: in-hospital death, the need for kidney replacement therapy (KRT), and KRT at discharge (Table 3). Surprisingly, we found that in-hospital death was high in patients who had received hemodynamic monitoring (44% death vs. 20% survived; P < 0.001) and in those who received volume administration (34% vs. 8%; P = 0.002). Furthermore, the need for KRT was significantly higher in patients whose drug therapy was adapted (28% need vs. 12% no need; P = 0.013) and in those who had nephrotoxic drugs discontinued (32% vs. 11%; P = 0.001) Finally, patients who had nephrotoxic drugs discontinued were more likely to require KRT at discharge (16% vs. 6%; P = 0.034).
According to the implemented KDIGO measures, the proportion of patients who died in hospital, required KRT or KRT at discharge.

Abbreviations: KDIGO, Kidney Disease Improving Global Guidelines; KRT, kidney replacement therapy ns, not statistically significant.
See Figure 1 for implementation rates in the total population.
Discussion
Surprisingly, our study in 200 patients from one centre, showed frequent implementation of several of the KDIGO recommended measures in de novo-AKI. Notably, hyperglycemia control was implemented in all patients diagnosed with elevated blood glucose. Volume therapy was applied in 82% of cases and fluid balance control in 65% of patients. However, substantial gaps were identified with regard to the discontinuation of nephrotoxic drugs (yes, 51%) and use of iodinated contrast media (yes, 35%). These findings are alarming considering the known risk of contrast-media associated AKI. 2 Subclavian access for central venous lines if indicated, had the lowest implementation rate (yes, 5%).
Subgroup analysis showed increased measure implementation in patients with a high risk of a severe disease course. For instance, volume therapy was applied significantly more often in AKI stage 3 as opposed to stage 1, in individuals with the need for ICU therapy or, with sepsis. Overall, patients in ICU had the greatest total implementation rate compared with all risk subgroups; for ICU patients five out of the seven measures were established. Dosage adaptation of medication and discontinuation of nephrotoxic drugs were the two measures that did not show a difference in implementation.
AKI dramatically worsens the prognosis of hospitalized and non-hospitalized patients in the short- and long-term. 13 It has been estimated that globally, AKI accounts for a higher death rate than diabetes mellitus, heart failure, breast and prostate cancer combined. 13 Therefore, strategies for improving early recognition and therapeutic intervention are urgently required. Some, progress has been made since the 2012 KDIGO recommendations. For example, numerous potential AKI biomarker molecules have been investigated over the past 10–15 years.14,15 From the 2012 diagnosis of AKI based exclusively on the basis of dynamic changes in serum creatinine and urine output, 5 revised diagnostic criteria will most likely include certain biomarkers.6,15 In addition, so-called ‘AKI alert systems’ have been tested under various clinical conditions.16,17 In principle, these systems critically depend on dynamic changes in serum creatinine. If a pre-defined threshold has been reached, automatic messages are delivered to the nephrologist in charge. Such messages usually contain patient-related information in an anonymized manner. Furthermore, AKI care bundles have been defined to ensure a more standardized approach to the management of patients with AKI. 18 Care bundles consist of sets of practices or measures that fulfill criteria of reliable evidence. 18
Our study had some limitations. For instance, it was from a single centre and had a retrospective design. Nevertheless, when evaluating the quality of care at a specific point in time or before implementation of local guidelines, a retrospective approach is often necessary. Additionally, our patient sample included individuals from several medical disciplines such as internal medicine, trauma/orthopedics, and obstetrics. It is possible that AKI-related guidelines may be less well-known in some surgical disciplines. This possibility emphasizes the need for additional AKI-related education among physicians from all disciplines.
In summary, our study highlighted the need for improvement in the quality of care for patients with AKI. While some 2012 KDIGO recommended measures were implemented for a substantial number of affected individuals, others were not. It is important to provide further AKI-related education to physicians to address this gap. Researchers in a previous study, implemented a multifaceted educational program, including lectures, case-based teaching in small groups, and interactive learning components to increase the number of physicians capable of initiating appropriate therapy upon the diagnosis of AKI. 11 The ultimate goal is to reduce the considerable morbidity and mortality associated with AKI in both the short and long term.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231221011 - Supplemental material for Adherence to guidelines for management of acute kidney injury
Supplemental material, sj-pdf-1-imr-10.1177_03000605231221011 for Adherence to guidelines for management of acute kidney injury by Sarah Hauptmann, Igor Matyukhin, Susann Patschan, Oliver Ritter and Daniel Patschan in Journal of International Medical Research
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research was supported by Brandenburg Medical School Theodor Fontane (Medizinische Hochschule Brandenburg Theodor Fontane, MHB) and publication fund supported by the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
