Abstract
Cerebral cavernous malformations constitute a subtype of cerebral vascular malformation typically located in the cerebral cortex. However, their occurrence in the suprasellar optic pathway is relatively rare. There is some uncertainty surrounding the clinical diagnostic methods and optimal treatment strategies specific to suprasellar optic pathway cavernous malformations. In this narrative review, we retrospectively analyzed relevant literature related to suprasellar visual pathway cavernous malformations. We conducted a study involving 90 patients who were postoperatively diagnosed with cavernous malformations, including the 16-year-old male patient mentioned in this article. We have summarized crucial clinical data, including the patient age distribution, sex ratio, lesion locations, primary symptoms, and surgical approaches. The comprehensive analysis of this clinical information underscores the critical importance of timely intervention in relieving symptoms and improving neurological deficits in affected patients. These findings provide valuable guidance and insight for clinical practitioners and researchers dealing with this specific medical condition.
Introduction
Cerebral cavernous malformations (CMs) are a type of cerebral vascular malformation (CVM) composed of dilated capillaries without smooth muscle or elastic tissue. The vessel walls are thin, and there is an absence of brain parenchyma between the vessels. The lesion exhibits slow blood flow and contains blood and various stages of thrombosis. 1 The incidence of this condition ranges from 0.16% to 0.50%. 2 Within the nervous system, CMs primarily occur in the cerebral hemispheres, accounting for approximately 75% of cases, followed by the infratentorial region (15%) and the spinal cord (5%). 3 In some cases, lesions may also develop in the intracranial cavernous sinus area. 4 Although CMs rarely affect cranial nerves, some reports have described instances in which CMs involved various cranial nerves such as the optic nerve, oculomotor nerve, facial nerve, and auditory nerve. 5 When CMs are situated in the suprasellar optic pathway, they have the potential to compress the optic nerve, resulting in symptoms such as visual impairment, visual field defects, and headaches.
Is should be noted that because of the rarity of such patients in clinical practice, there are currently no established standard diagnostic or treatment protocols. Therefore, we compiled the clinical data from 90 relevant cases, including a recent case reported by our institution. We conducted a comprehensive analysis of these cases, including the age distribution, sex ratio, choice of surgical approach, postoperative treatment outcomes, and relationship between treatment timing and prognosis during follow-up. We then summarized the latest research developments in nonsurgical treatments for cerebral CMs. The aim of this narrative review is to provide neurosurgeons with valuable insight and experience that will help facilitate timely diagnosis and selection of appropriate treatment strategies for CMs occurring in the suprasellar visual pathway.
Case presentation
A 16-year-old male patient presented with a 1-week history of intermittent blurred vision and unexplained headaches. Bilateral temporal hemianopsia was detected during the physical examination. The patient was subsequently recommended to undergo a sellar magnetic resonance imaging (MRI) scan. The MRI results revealed an irregular mixed T2 signal nodule within the sella turcica surrounded by a ring-like low-signal band. After contrast enhancement, mild heterogeneous enhancement was observed along with compression of the optic chiasm (Figure 1). Head computed tomography angiography indicated the presence of a space-occupying lesion in the sella turcica, adjacent to the right cervical internal carotid artery and the right anterior cerebral artery A1 segment. Based on the patient’s radiological presentation, a preliminary diagnosis of a suprasellar mass was made; however, the nature of the lesion remained unclear.

An irregular and heterogeneous nodule, as indicated by the white arrows, is observed within the saddle-shaped pool on T1-weighted imaging (T1WI) and T2-weighted imaging (T2WI). Mild inhomogeneous enhancement is visible on postcontrast gadolinium-enhanced T1WI (CE-T1WI): sagittal (a) T1WI, (b) T2WI, and (c) CE-T1WI and coronal (d) T1WI, (e) T2WI, and (f) CE-T1WI.
To determine the nature of the lesion and alleviate compression, we decided to proceed with surgical treatment following extensive discussions with the patient and his family. Given the lesion’s proximity to critical structures such as the pituitary, optic chiasm, and hypothalamus, a comprehensive preoperative assessment of the patient’s hormone levels and visual field was conducted. Consultation with an ophthalmologist revealed normal preoperative visual acuity but bilateral temporal hemianopsia (Figure 2). All preoperative hormone levels were within the normal range.

Preoperative visual field test report.
The patient underwent general anesthesia, and a pterional approach was used for lesion resection. Intraoperatively, the mass was identified at the site of the optic chiasm and appeared dark yellow in color. The lesion was punctured and revealed to be a hematoma after electrocoagulation. Microscopic examination allowed for complete removal of the lesion, given its relatively soft consistency and moderate vascularity (Figure 3). Postoperative pathologic examination confirmed a CM with hemorrhage (Figure 4), ultimately establishing a definitive clinical diagnosis of a cavernous hemangioma of the optic chiasm.
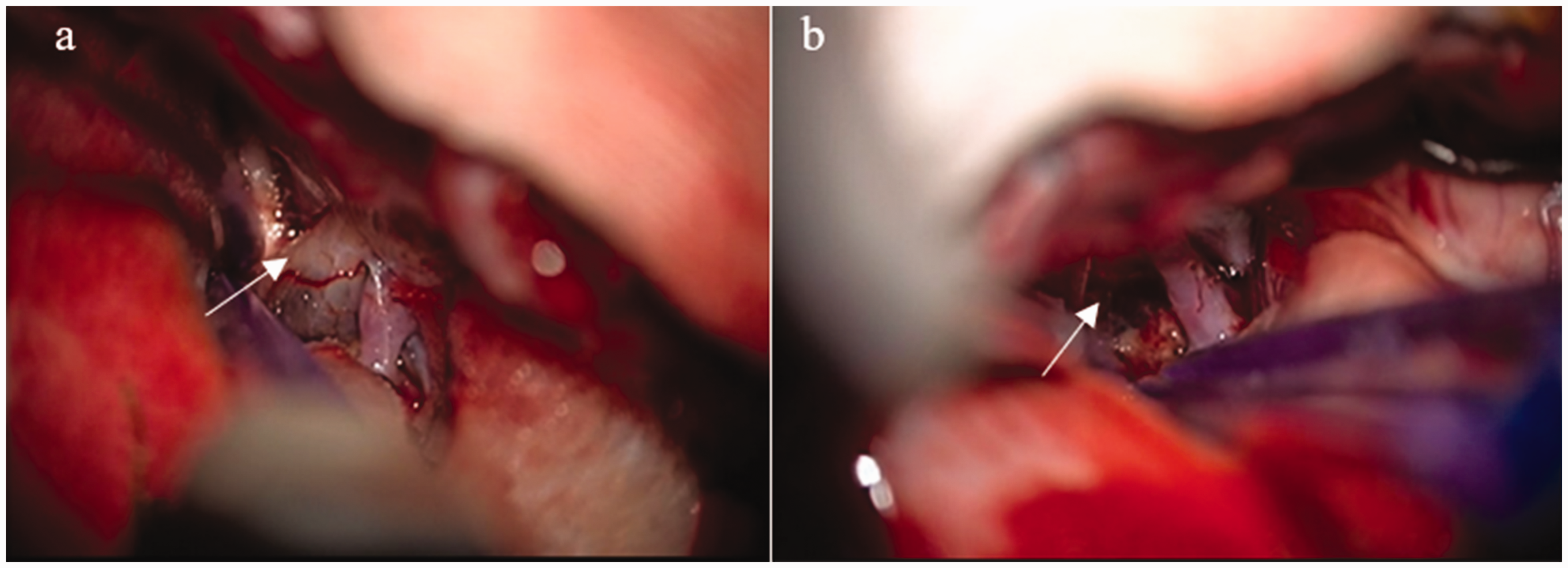
Intraoperative mass in the patient’s optic chiasma. (a) Pre-excision image (white arrows). (b) Image of resected mass after complete excision (white arrows).

The patient’s pathologic examination revealed dilated capillaries with bleeding.
One week postoperatively, a follow-up examination revealed normal vision and a normal fundus examination with a reduction in the range of bilateral temporal hemianopsia. All pituitary-related hormones remained within the normal range postoperatively. Three months after surgery, a follow-up MRI scan indicated that the tumor had been completely removed with no signs of recurrence (Figure 5).

The patient underwent follow-up head magnetic resonance imaging 3 months postoperatively, which revealed complete tumor resection with no signs of recurrence. (a) T1-weighted imaging, (b) T2-weighted imaging, (c) contrast-enhanced T1-weighted imaging.
Review
Methods
Ethics
All patients described in this article provided verbal informed consent, and the clinical information of the other patients was derived from publicly available literature. This study involved no drug trials or clinical research; therefore, approval by an ethics committee was not required.
Search methodology and inclusion criteria
The PubMed database was searched for articles published from 1970 to 2024. Medical Subject Headings (MeSH) terms, combining “optic nerve,” “optic chiasm,” “optic tract,” and “cavernous malformation,” were used for the PubMed search. All published articles that focused on CMs confirmed through postoperative pathological examination and located in the suprasellar optic pathway were included in the review.
Extraction of clinical data
Patients’ clinical characteristics were categorized and summarized, including age, sex, site of occurrence, brief medical history, time from symptom onset to first surgery, extent of resection in the first surgery, surgical approach in the first surgery, and postoperative outcomes of the first surgery. If patients underwent a second surgery, data on the time from the first surgery to the second surgery, surgical approach in the second surgery, extent of resection in the second surgery, and postoperative outcomes of the second surgery were recorded. Because this condition primarily affects visual acuity and the visual field, the patients’ postoperative status was classified into the following four levels by comparing the preoperative status and postoperative improvement in these two parameters: improved (improvement in both visual acuity and the visual field compared with the preoperative status), partially improved (improvement in either visual acuity or the visual field), unchanged (no change in visual acuity or the visual field compared with the preoperative status), and deteriorated (worsening of visual acuity or the visual field compared with the preoperative status).
Statistical analysis
Statistical analysis of the collected data was performed using SPSS 26.0 (IBM Corp., Armonk, NY, USA). Count data are presented as n (%), and Spearman’s rank correlation analysis was used to examine the relationship between the time from symptom onset to the first surgery and the postoperative improvements in visual acuity and the visual field. A significance level of p < 0.05 was used to indicate statistical significance.
Results
The cases identified in this review are summarized in Table 1. Among a total of 90 patients, data on sex were available for 87 patients. These 87 patients comprised 41 (47.1%) male patients and 46 (52.9%) female patients. Their average age was 34.9 years, with a standard deviation of 13.9 years and range of 4 to 78 years. The optic chiasm was the most commonly affected site in the visual pathway, accounting for 96.6% of cases. Adjacent structures were also involved, including involvement of the hypothalamus in two cases6,7 and involvement of the pituitary gland in one case. 8 Of the 89 patients who exhibited symptoms, 90.0% experienced vision loss, 84.3% developed visual field defects, and 60.0% reported headaches as the three main symptoms. Some patients had pupillary reflex abnormalities, with 10% showing relative afferent pupillary defects and one patient presenting with an absent pupillary light reflex. Among the four patients with involvement of the hypothalamus, pituitary gland, or floor of the third ventricle, hormone disorders were observed, manifesting as diabetes insipidus, sexual dysfunction, adrenal insufficiency, and hypopituitarism. One patient exhibited ocular motor dysfunction due to restricted abduction, resulting in diplopia. Some severely affected patients presented with impaired consciousness, including two cases of delirium, one case of somnolence, and, in the most severe case, coma resulting in eventual death. Two patients showed radiological evidence of subarachnoid hemorrhage. The surgical procedures were clearly documented in 61 patients. The surgical approaches were broadly categorized as lateral approaches and midline approaches. The lateral approaches were the most commonly chosen, including the pterional approach (30 cases), frontotemporal approach (9 cases), orbitozygomatic approach (5 cases), supraorbital or lateral supraorbital approach (6 cases), and subfrontal approach (4 cases). The midline approaches included two main approaches: the interhemispheric approach (4 cases) and transsphenoidal approach (3 cases). A total of 67 patients had recorded data on the extent of resection and postoperative follow-up. Complete resection was achieved in 43 patients. A total of 18% of patients had an unchanged visual acuity and visual field compared with the preoperative status, while 82% of patients showed varying degrees of improvement in visual acuity and the visual field. Only two patients experienced worsening of their visual field defects, one of whom underwent the transsphenoidal approach. Six patients underwent subtotal resection, which carried a risk of recurrence. Among the statistically analyzed patients, three redeveloped abnormalities in their visual acuity and visual field, and MRI showed a correlation of these changes with residual or recurrent tumors. Patients who only underwent biopsy or decompression experienced minimal clinical benefit. Among the 13 patients who underwent biopsy, 5 experienced postoperative deterioration in their visual acuity and visual field. Three of them underwent subsequent surgical resection, resulting in improvements of their visual acuity and visual field despite the delayed surgery.
Summary of patient information.
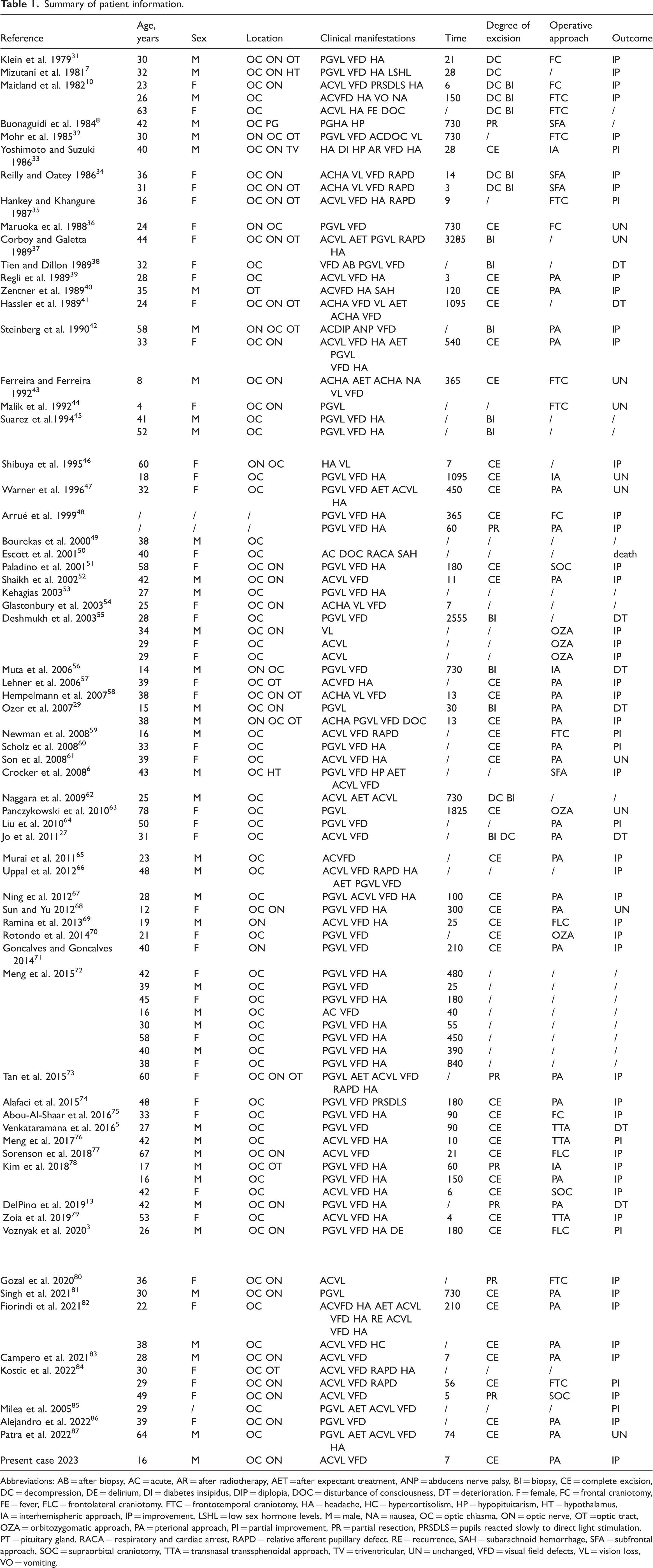
Abbreviations: AB = after biopsy, AC = acute, AR = after radiotherapy, AET = after expectant treatment, ANP = abducens nerve palsy, BI = biopsy, CE = complete excision, DC = decompression, DE = delirium, DI = diabetes insipidus, DIP = diplopia, DOC = disturbance of consciousness, DT = deterioration, F = female, FC = frontal craniotomy, FE = fever, FLC = frontolateral craniotomy, FTC = frontotemporal craniotomy, HA = headache, HC = hypercortisolism, HP = hypopituitarism, HT = hypothalamus, IA = interhemispheric approach, IP = improvement, LSHL = low sex hormone levels, M = male, NA = nausea, OC = optic chiasma, ON = optic nerve, OT = optic tract, OZA = orbitozygomatic approach, PA = pterional approach, PI = partial improvement, PR = partial resection, PRSDLS = pupils reacted slowly to direct light stimulation, PT = pituitary gland, RACA = respiratory and cardiac arrest, RAPD = relative afferent pupillary defect, RE = recurrence, SAH = subarachnoid hemorrhage, SFA = subfrontal approach, SOC = supraorbital craniotomy, TTA = transnasal transsphenoidal approach, TV = triventricular, UN = unchanged, VFD = visual field defects, VL = vision loss, VO = vomiting.
Forty-four patients underwent complete tumor resection during surgery, and 82% of the patients showed varying degrees of recovery of their visual acuity and visual field. We categorized the patients based on the duration of time from symptom onset or diagnosis to the first surgery. We used a four-grade scale to classify the improvement in visual acuity and the visual field after surgery: improvement, partial improvement, unchanged, and deterioration. Spearman rank correlation analysis was performed to explore the relationship between the occurrence of symptoms or the time to first surgery and the degree of postoperative recovery in visual acuity and the visual field. The results showed a Spearman correlation coefficient of −0.421 and a p-value of <0.05 between the occurrence of symptoms or the time to first surgery and the degree of postoperative recovery. These data suggest that timely and complete tumor resection tends to be associated with positive visual and visual field recovery outcomes for many patients.
Discussion
Epidemiology
CM is one type of CVM, which also includes arteriovenous malformation, venous angioma, and capillary telangiectasia. The prevalence of CVM in the general population is estimated to range from 0.4% to 0.8%. CMs account for approximately 10% to 25% of all CVMs. 9 Lesions in the central nervous system primarily occur above the tentorium. The occurrence of CMs in the optic nerve is extremely rare, with only a few cases having been reported to date.
Clinical presentation
CMs in the region of the optic chiasm can impact the optic nerve because of acute hemorrhage or tumor compression, resulting in visual impairment and visual field defects. Acute hemorrhage can trigger sudden onset of headaches, visual decline, and hemianopsia, collectively known as “optic chiasm syndrome.” This syndrome was first described by Maitland et al. 10 in 1982.
In our cohort, 52 (59%) patients exhibited acute optic nerve dysfunction. Acute hemorrhage is a significant contributor to severe complications in patients with CMs. According to the literature, CMs that occur within the brain parenchyma have a hemorrhage rate of 25%. 2 Because of their proximity to the optic nerve, CMs located in the suprasellar optic chiasm region can cause symptoms even with minor bleeding. This is one potential factor leading to a higher rate of patient visits for acute bleeding-induced visual and visual field disturbances.
By contrast, chronic hemorrhage primarily presents radiologically as equal or low signal intensity on T1-weighted imaging (T1WI) and T2-weighted imaging (T2WI), along with a hemosiderin rim at the periphery of the lesion. 11 Repeated chronic bleeding is considered a cause of seizures associated with CMs within the brain parenchyma, 2 although there is currently no specific report regarding its impact on the optic chiasm region.
When CMs extend, they can affect structures adjacent to the optic chiasm, leading to corresponding symptoms. When the lesion involves the hypothalamus or pituitary, it can result in hormonal imbalances such as diabetes insipidus, sexual dysfunction, and adrenal insufficiency. Severely affected patients may also exhibit varying degrees of consciousness disturbances, including excessive drowsiness or even coma.
Diagnostic tests
MRI findings of CMs
MRI is currently recommended for the diagnosis and follow-up of CMs. 2 Considering the patient’s symptoms and site of onset, differential diagnoses should include diseases such as craniopharyngioma, optic neuritis, pituitary adenoma, germ cell tumors, optic nerve glioma, aneurysm, and sarcoidosis (chiasmal involvement due to basal leptomeningitis). 12 On MRI, lesions affecting the optic nerve or tract typically manifest as thickened nerves with an increased diameter, whereas lesions involving the chiasm or hypothalamus are characterized by focal and round suprasellar masses.
CMs can exhibit various signal characteristics on MRI based on the presence of different stages of hemorrhage, thrombosis, calcification, and fibrosis within the lesions. CMs can be classified into four types based on their imaging features. Type I shows high signal intensity on T1WI, representing subacute hemorrhage with high iron content. Type II shows mixed signal intensity on both T1WI and T2WI, with surrounding low signal intensity on T2WI indicating hemosiderin deposition and the presence of thrombi and hemorrhage at different stages within the lesion. Type III appears as isointensity or hypointensity on both T1WI and T2WI, representing chronic hemorrhage. Type IV is characterized by small, multiple, punctate lesions with low signal intensity on both T1WI and T2WI, indicating the presence of multiple microhemorrhages within the lesion. 11 The typical presentation of a CM involving the optic chiasm is a sellar mass with low signal intensity on T1WI and mixed signal intensity on T2WI. It may exhibit a characteristic popcorn-like appearance with peripheral low signal intensity representing the hemosiderin ring. Usually, no significant enhancement is observed on contrast-enhanced scans.13,14
Susceptibility-weighted imaging is highly sensitive to the detection of hemosiderin and calcifications, enabling more precise depiction of multiple lesions and multifocal abnormalities. As a result, susceptibility-weighted imaging can effectively determine the presence of multiple CM lesions. Notably, cerebral angiography is not useful for visualizing lesions within CMs because of thrombus formation and low blood flow. The primary role of cerebral angiography is to identify associated CVMs and differentiate them from aneurysms. 9
Pathological features of CMs
The confirmation of an intracranial CM primarily relies on postoperative histopathology. Grossly, the tumor specimen appears as a raspberry-like structure with a purplish-red color. The tumor has well-defined margins and is stained with hemosiderin because of recurrent hemorrhage. Microscopically, it presents as blood-filled sinusoids or cavities lined with endothelial cells. These cavities are typically filled with red blood cells, and a dense connective tissue matrix often exists between them. Under electron microscopy, gaps are observed between closely connected endothelial cells, and astrocytic foot processes are absent. This disruption may confirm breakdown of the blood–brain barrier, which may be the cause of bleeding. 15
Genetic counseling
CMs can manifest in two forms: sporadic and familial. The sporadic form is typically characterized by a single lesion, either with or without an associated venous malformation. By contrast, familial CM presents as multiple lesions and accounts for approximately 20% of cases. 16 In the Spanish-speaking population, there is a higher incidence of familial CMs, which constitute 50% of CMs among Spanish-speaking American patients. 17 Familial CMs and more than half of multiple sporadic CMs are believed to be caused by autosomal dominant mutations in KRIT1 (CCM1), CCM2 (MGC4607), and PDCD10 (CCM3), which are located on chromosomes 7q, 7p, and 3q, respectively. Experimental studies in animal models have demonstrated that these gene mutations result in increased capillary proliferation and enhanced permeability. 18 Patients with CCM3 mutations often exhibit more severe clinical manifestations, and they may develop intracranial hemorrhage at a younger age. 16
The formation of CMs is believed to adhere to the Knudson two-hit mutation hypothesis. This hypothesis posits that complete loss of both copies of one of the CCM genes is necessary to trigger the formation of CMs. 19 In the case of familial cerebral CMs, the first “hit” involves a hereditary genetic mutation occurring in germline cells, and the second “hit” typically involves a somatic mutation affecting the same gene. This second “hit” is a crucial event in the disease development. This concept also clarifies why familial cerebral CM exhibits an autosomal dominant inheritance pattern despite requiring two separate “hits” for disease manifestation. 20
CMs are inherited as an autosomal dominant disorder, making genetic testing crucial for prenatal genetic counseling and timely identification of high-risk individuals, thus allowing necessary interventions. Genetic testing is recommended for individuals at high risk, such as those with multiple intracranial lesions as indicated by MRI or a positive family history. 21
Treatment of CMs
The treatment of CMs is dependent on several factors, including the patient’s age, clinical symptoms, lesion characteristics, and lesion location. Asymptomatic lesions typically do not necessitate intervention and can be managed conservatively through regular monitoring. However, symptomatic or high-risk lesions, particularly those associated with recurrent bleeding, may necessitate surgical removal. The primary treatment modalities for CMs include surgical intervention, stereotactic radiosurgery, and conservative observation.
Conservative management and drug therapy
Conservative treatment may be recommended for asymptomatic patients with incidentally discovered CMs. 2 In the present study, 89 patients presented with relevant symptoms, and 12 (13.5%) exhibited a poor response to conservative treatment with varying degrees of symptom exacerbation. Therefore, close follow-up and monitoring of the lesion size, symptoms, and occurrence of rebleeding are necessary for patients undergoing conservative treatment.
With advancements in the understanding of the pathogenesis of CMs, certain drugs with potential therapeutic value have been identified. For example, Rho kinase inhibitors such as atorvastatin have been found to delay disease progression and reduce the risk of hemorrhage. Superoxide dismutase mimetics such as tempol can help reduce microvascular leakage. β-catenin transcriptional attenuators, such as sulindac sulfone, have shown potential in reducing the volume and number of CMs. β-blockers, such as propranolol, have been found to lower the risk of hemorrhage.22,23 A large clinical controlled study demonstrated that propranolol can reduce the risk of rebleeding in patients with familial CMs. 24
Stereotactic radiotherapy
When the lesion is located in the deep brain or in functional areas, stereotactic radiosurgery can be considered to reduce the risk of bleeding. 25 Karaaslan et al. 26 found that stereotactic radiosurgery can significantly reduce the risk of bleeding in CMs located in critical areas, such as the cerebral hemisphere or brainstem. However, the effectiveness of stereotactic radiosurgery for lesions in the optic chiasm region remains uncertain. Jo et al. 27 reported a case in which the patient developed recurrent bleeding and progression despite undergoing postoperative radiation therapy. To avoid the risk of subsequent bleeding and damage to the visual pathway, some experts suggest avoiding reflex treatment in that region. 13 Additionally, stereotactic radiosurgery is not recommended for patients with a family history of the disease because it may accelerate disease progression. 2
Laser interstitial thermal therapy
The use of laser interstitial thermal therapy, which is guided by MRI, delivers precise laser energy to the lesion site to induce cellular damage. This method has been explored as an alternative treatment for CMs in high-risk surgical areas such as deep brain tissue and the brainstem. It is intended to alleviate symptoms and to prevent and reduce the risk of bleeding. However, complications such as postoperative bleeding and focal functional deficits may occur. No reports to date have described the use of this technique to treat CMs in the optic chiasm region. Further research and clinical evidence are required to determine the efficacy and safety of laser interstitial thermal therapy for treating CMs in this specific location. 28
Surgical resection
Surgical treatment is recommended for patients with clinical symptoms, focal neurological dysfunction, and symptomatic bleeding. 2 The purpose of surgery is to prevent lesion hemorrhage and growth and provide decompression of the optic nerve and optic chiasm. Complete resection of the lesion during surgery is desirable to prevent recurrence. Biopsy is not recommended because it has been reported to cause rapid tumor enlargement in the short term in cases reported by Ozer et al. 29 Furthermore, in the present study, patients who underwent complete resection in a timely manner demonstrated better postoperative visual acuity and visual field recovery. The surgical approach can be selected according to the tumor’s location, with options including a lateral or midline approach. As advancements in neurosurgical techniques and concepts continue, surgical approaches are becoming less invasive. An example of this is the supraorbital keyhole approach. This approach offers several benefits, including smaller surgical incisions, reduced brain tissue retraction and postoperative pain, a shorter surgical duration and hospital stay, and lower infection rates. 30
Conclusion
Suprasellar visual pathway CMs, often caused by tumor hemorrhage, lead to impaired vision and visual field deficits. The primary clinical diagnostic method for this condition before surgery is MRI, with definitive diagnosis primarily reliant on postoperative pathology. In the selection of treatment strategies, timely surgical intervention holds significant importance in improving patients’ visual acuity and field of vision.
With the advancements in microscopic and endoscopic techniques along with the progress in neurosurgical principles, the future of surgery is inevitably moving toward more minimally invasive and precise approaches. An increasing array of relevant pharmacological agents will be applied in clinical treatment to reduce the risk of hemorrhage, reduce the tumor volume, and prevent recurrence. Radiation therapy or other novel treatment modalities also await further exploration. This evolving landscape promises a more optimistic outlook for the diagnosis and management of suprasellar visual pathway CMs.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Dr. Yuxiong Wang for his invaluable assistance in reviewing and editing this manuscript. His expert insights and guidance were instrumental in improving this research.
Author contributions
Liu Yang: Conceptualization, literature review, and manuscript writing.
Songbai Xu: Review of clinical case studies, data analysis, and manuscript editing.
Both authors have reviewed and approved the final version of the manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study did not receive any specific grant from funding agencies in the public, commercial or non-profit sectors.
