Abstract
Objective
To explore the effects of oral semaglutide on glycaemic parameters, body weight, and satisfaction in the first recipient patients with type 2 diabetes mellitus in Slovenia, in a real-world clinical practice setting.
Methods
The first consecutive adult patients with type 2 diabetes who were eligible for oral semaglutide treatment were included in this prospective, open-label interventional study. Patients received increasing doses of oral semaglutide and were evaluated at inclusion, at 1 month, then 3–5 months after starting treatment. Fasting blood glucose, glycosylated haemoglobin (HbA1c), body weight, patient satisfaction with oral semaglutide treatment (using the validated Treatment Satisfaction Questionnaire for Medication), and adverse effects, were analysed. Statistical analyses were performed using one-way analysis of variance, and, when significant interactions were found, Bonferroni post-hoc test. A P-value <0.05 was considered statistically significant.
Results
Twenty patients (11 male: 9 female; mean age, 59.9 ± 1.5 years; mean diabetes duration, 8.5 ± 1.4 years) were included. Oral semaglutide (7 and 14 mg) significantly decreased HbA1c (from 9.4 ± 0.3% to 8.2 ± 0.2% and 7.8 ± 0.2%, respectively) and fasting plasma glucose (from 11.2 ± 0.5 mmol/L to 9.2 ± 0.7 mmol/L and 8.9 ± 0.4 mmol/L, respectively). Oral semaglutide (14 mg) significantly decreased body weight (from 100.9 ± 2.7 kg to 92.7 ± 2.4 kg). Patients reported that treatment was easy to use and expressed high global satisfaction. Mild and transient, mostly gastrointestinal, adverse effects were reported in 10 patients.
Conclusions
Oral semaglutide, the first oral glucagon-like peptide 1 receptor agonist, was effective and safe, and associated with high patient satisfaction, in its first recipients in Slovenia. The results are important for daily clinical practice involving patients with type 2 diabetes, however, due to the small study population, lack of placebo control, and short exposure to oral semaglutide, the effectiveness of oral semaglutide in clinical practice requires further investigation.
Keywords
Introduction
Type 2 diabetes mellitus is a chronic disease that lasts a lifetime. Not only the disease burden itself, which requires adequate antidiabetic therapy, but also its complications, lead to a worse quality of life for the affected individuals.1,2 The pace of disease development is unpredictable, but it ultimately leads to complete pancreatic β-cell failure and the eventual need for insulin treatment. However, treatment options for type 2 diabetes are advancing at a rapid pace. One of the novel antidiabetic treatment options is glucagon-like peptide 1 receptor agonists (GLP-1RA) that, until recently, were only available for administration by subcutaneous injection, 3 which frequently represented a treatment barrier for the patient or healthcare worker. 4 Recently, oral semaglutide, the first GLP-1RA for oral administration, was made available for use in clinical practice worldwide, 5 including in Slovenia. Oral semaglutide offers a good alternative to injection treatment and allows a timely and broad use of this class of drugs, which is also currently recommended as first-line therapy for type 2 diabetes in certain clinical scenarios. 6
Oral semaglutide has been shown to be effective in reducing glycaemia, that is, glycosylated haemoglobin (HbA1c) and body mass, in the series of Peptide InnOvatioN for Early diabEtes tReatment (PIONEER) studies. 7 In these studies, oral semaglutide was administered to individuals with baseline HbA1c levels between 7.0 and 9.5% and different durations of diabetes (3.5–15 years), and was compared with placebo or other antidiabetic treatment (including insulin) for a period of between 26 and 52 weeks. Oral semaglutide, at doses of 7 or 14 mg, reduced HbA1c, with reductions of 0.9–1.7% and 1.0–2.0%, respectively, and was also shown to be effective in reducing body weight, with reductions of 1.0–2.4 kg for 7 mg and 2.2–4.4 kg for 14 mg. 7 Furthermore, it was proved safe, with the most common patient-reported adverse events comprising nausea, vomiting, diarrhoea, constipation, and dyspepsia. All of these adverse events were mild and transient in all PIONEER studies, and a very small proportion of patients discontinued therapy. 7 Oral semaglutide showed a favourable cardiovascular risk profile in the PIONEER 6 study and was shown to be noninferior to placebo in terms of cardiovascular safety. 8 The Semaglutide Anti-Atherosclerotic Mechanisms of Action Study (SAMAS), evaluating the general safety and specific cardiometabolic effects of the latest semaglutide formulation available on the market, is currently ongoing. 9
Data on oral semaglutide in real-world settings remains scarce. 10 Furthermore, patient satisfaction with this new drug, which requires a certain administration regime, has not yet been evaluated. Therefore, the objective of the present study was to: (1) investigate the efficacy of oral semaglutide on glycaemic parameters and body mass, (2) explore the safety of oral semaglutide, and (3) determine patient satisfaction with oral semaglutide in the first recipients of this drug in Slovenia.
Patients and methods
Study population and design
This prospective, open-label, non-randomized interventional study included the first consecutive adult patients with type 2 diabetes who were eligible for oral semaglutide treatment at the outpatient clinic of the University Medical Centre Ljubljana, Ljubljana, Slovenia.
Inclusion criteria for the study population comprised a confirmed diagnosis of type 2 diabetes with HbA1c > 7.0%, age 30–65 years, and body mass index (BMI) > 30 kg/m2, as required by the Health Insurance Institute of Slovenia regarding all GLP-1RA medications at the time of the study. Patients were required to have been treated with at least two other oral antidiabetic agents prior to study inclusion, and prior therapy should have been stable for the previous 3 months. Exclusion criteria were treatment with subcutaneous GLP-1RA or insulin therapy, advanced renal failure with an estimated glomerular filtration rate (eGFR) < 30 ml/min/1.73 m2, proliferative diabetic retinopathy, positive family history of multiple endocrine neoplasia 2, or history of medullary thyroid carcinoma.
All participants entered the study voluntarily and provided written informed consent to study inclusion. The study was performed according to the ethical standards of the Declaration of Helsinki and was approved by the Slovenian National Medical Ethics Committee (approval No. 0120-245/2021/3). The investigation is a substudy of the trial registered at http://clinicaltrials.gov (NCT05147896), and the reporting of this study conforms to CONSORT guidelines. 11
Study protocol
Participants were evaluated at study inclusion (visit 1) and then at 1 month and 3–5 months after the initiation of oral semaglutide treatment (Figure 1). At inclusion, a complete medical history and full medical examination were performed. At all visits, fasting venous blood samples were also collected in the morning, after an overnight fast, for the analysis of blood glucose values and HbA1c values. An automated sphygmomanometer was used to measure blood pressure (mmHg). Body height (m) was measured using a standard wall mounted stadiometer and weight (kg) was measured using a standard body scale. BMI (kg/m2) was then calculated.

Schematic of the study protocol. Symbols represent collection of venous blood, measurement of weight, height and blood pressure, and administration of the Treatment Satisfaction Questionnaire for Medication (TSQM) version 1.4 at visit 4; and 3, 7 and 14 mg represents oral semaglutide dose.
Participants received oral GLP-1RA semaglutide (Rybelsus® 3 mg, 7 mg, 14 mg, Novo Nordisk A/S Bagsvaerd, Denmark) in addition to their previous antidiabetic treatment, and were instructed to discontinue any treatment with dipeptidyl dipeptidase-4 (DPP-4) inhibitor. Patients were instructed to take oral semaglutide once daily, in the morning on an empty stomach after a fasting period of at least 8 h. After taking the oral semaglutide, participants were instructed to wait at least 30 min before ingestion of food, beverages, or other oral medications. The oral semaglutide tablet had to be taken with no more than a half glass of water (equivalent to 120 ml), swallowed whole and could not be split, crushed, or chewed.
To avoid the development of potential side-effects, a dose-escalation regimen was followed, according to standard protocols: the starting dose of oral semaglutide was 3 mg, which was increased to 7 mg after at least 4 weeks, and then to 14 mg after at least a further 4 weeks. Background antidiabetic therapy (metformin and sodium-glucose cotransporter-2 [SGLT-2] inhibitors) was maintained at the same dose throughout the study. The dose of sulphonylurea was modified, if necessary, due to adverse events (e.g., hypoglycaemia).
Blood sample analyses
Blood glucose was analysed according to the principle of enzyme amperometry using a SensoStar GL one analyser (DyaSys Diagnostic Systems GmbH, Holzheim, Germany) and HbA1c was determined by a high-performance liquid chromatography D-100 system (Bio-Rad Laboratories, Inc., Hercules, CA, USA), according to the manufacturers’ instructions.
Side-effects evaluation
The side-effects of oral semaglutide treatment were explored at each visit with active questions. Evaluation consisted of the occurrence of hypoglycaemia, decreased appetite sensation, nausea, bloating, and abdominal pain, as well as vomiting, diarrhoea, constipation, dyspepsia, and signs of nasopharyngitis. The possible answers were yes or no. To detect hypoglycaemia, patients were provided with glucometers and advised to check blood glucose values in the event of symptoms and signs of hypoglycaemia.
Treatment satisfaction evaluation
Patient satisfaction with oral semaglutide treatment was assessed using the Treatment Satisfaction Questionnaire for Medication (TSQM), version 1.4 (IQVIA; Madrid, Spain), which has been validated in the Slovenian language.12,13 The TSQM (version 1.4) consists of 14 questions divided into 4 subscales: effectiveness (questions 1–3), side-effects (questions 4–8), convenience (questions 9–11), and global satisfaction (questions 12–14). Responses were measured on a Likert-type scale, each with a maximum of 5 or 7 points, except for question 4 (side-effects subscale) which is a question about the presence or absence of side-effects (yes/no answer). If the answer to this question was no (no side-effects reported by the participant), other questions in the side-effects subscale (questions 5–8) were not asked. In the present study, mean scores were calculated for each question (in addition to composite subscale scores) and converted to a range of 0 to 100, with higher scores indicating greater treatment satisfaction, better treatment efficacy, fewer side-effects, and better practicality of treatment. Patients were asked to complete the TSQM at the last study visit, which was 5 months after the introduction of oral semaglutide treatment, at a time-point when all participants were expected to be receiving oral semaglutide at a dose of 14 mg daily.
Statistical analyses
Data are presented as mean ± SEM or n (%) prevalence, and were statistically analysed using one-way analysis of variance (ANOVA). When significant interactions were found, Bonferroni post-hoc test was performed. GraphPad Prism, version 5.0 (GraphPad Software, Boston, MA, USA) was used for all statistical analyses and a P value < 0.05 was considered statistically significant.
Results
Patient characteristics
Twenty middle-aged patients with type 2 diabetes, comprising nine female and 11 male patients, were included in the study, with their baseline clinical characteristics presented in Table 1. Most of the patients had one or two risk factors for cardiovascular disease (in addition to diabetes), and a few patients had a history of atherosclerotic cardiovascular disease. Most of the patients had a combination of three oral antidiabetic drugs as prior therapy, and none of the patients received an injectable GLP-1RA or insulin (Table 1).
Demographic and clinical characteristics of 20 patients with type 2 diabetes mellitus at study inclusion (baseline).

Data presented as mean ± SEM or n (%) prevalence.
BMI, body mass index; BP, blood pressure; HbA1c, glycosylated haemoglobin; ALT, alanine transaminase; AST, aspartate aminotransferase; eGFR, estimated glomerular filtration rate; LDL, low-density lipoprotein; γ-GT, gamma-glutamyl transferase; SGLT-2, sodium glucose cotransporter-2; DPP-4, dipeptidyl dipeptidase-4.
At study inclusion, all patients received oral semaglutide, 3 mg daily, and at 1 month after study inclusion, all patients received an increased dose to 7 mg daily. At the 3-month visit, the dose of oral semaglutide was further increased to 14 mg in 14 patients who did not report side effects; in the other six patients who reported mild gastrointestinal side-effects, the dose of oral semaglutide was increased to 14 mg at 4 months after study inclusion. At the last study visit (5 months after inclusion) all patients received oral semaglutide at a dose of 14 mg daily.
Effect of oral semaglutide on glycaemic parameters
Oral semaglutide at doses of 7 mg or 14 mg daily (at 3 and 5 months of study inclusion, respectively), was associated with significantly decreased HbA1c compared with baseline values (from 9.4 ± 0.3% to 8.2 ± 0.2% [–1.2%] at 3 months [P < 0.05] and from 9.4 ± 0.3% to 7.8 ± 0.2% [–1.6%] at 5 months [P < 0.01]; Figure 2a). Oral semaglutide at a dose of 3 mg daily, measured after 1 month of treatment, did not significantly influence HbA1c values. HbA1c ≤7.0% was reached at 3–5 months after initiation of oral semaglutide in six of the included patients (30%).
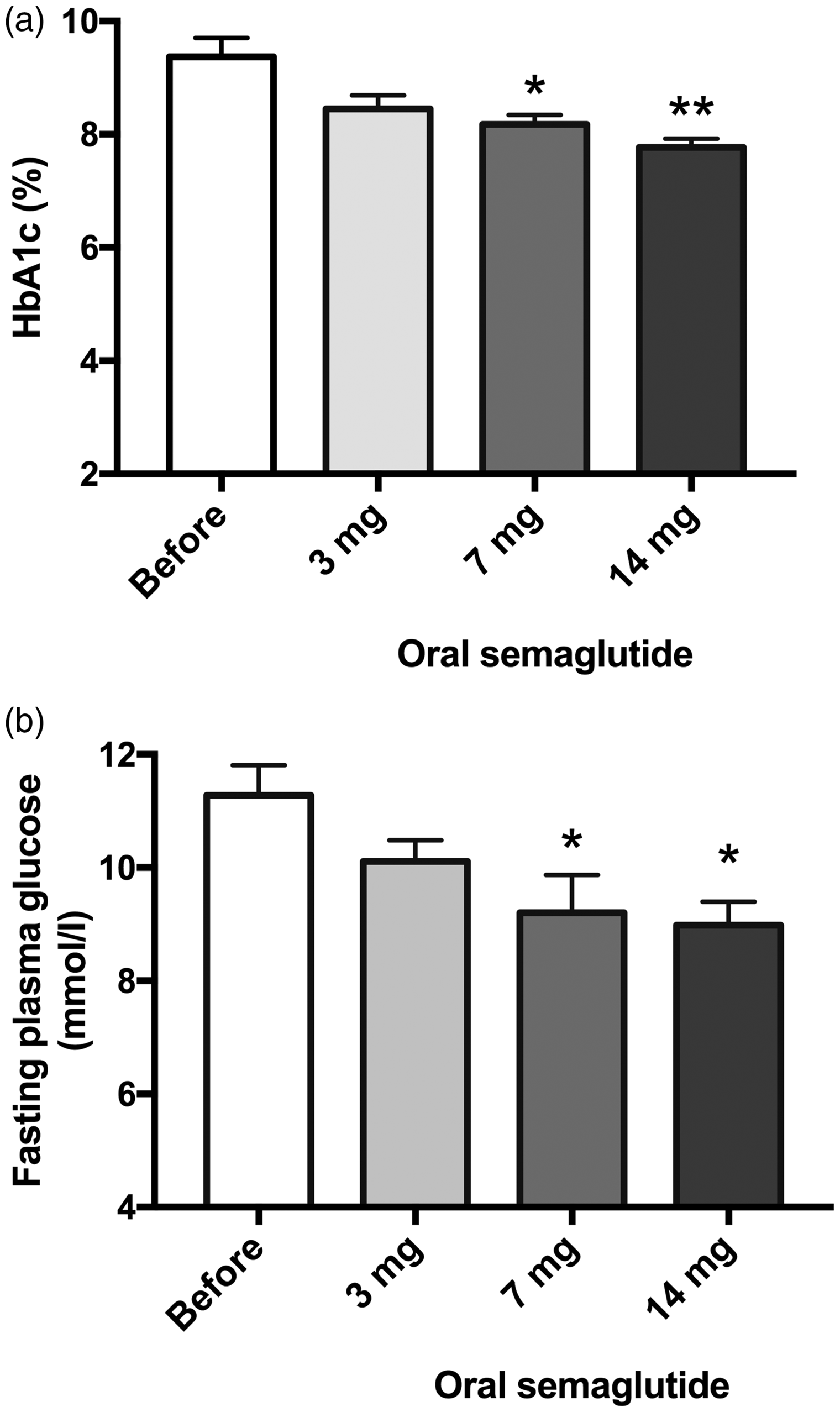
Glycosylated haemoglobin (HbA1c) (a) and fasting plasma glucose (b) at baseline (before starting oral semaglutide) and after treatment with oral semaglutide at doses of 3, 7 or 14 mg daily in 20 patients with type 2 diabetes mellitus. Data presented as mean ± SEM; *P < 0.05 versus baseline value; **P < 0.01 versus baseline value.
Oral semaglutide at doses of 7 mg or 14 mg daily (at 3 and 5 months of study inclusion, respectively), was associated with significantly decreased fasting plasma glucose values compared with baseline (from 11.2 ± 0.5 mmol/L to 9.2 ± 0.7 mmol/L [17.9% decrease] and from 11.2 ± 0.5 mmol/L to 8.9 ± 0.4 mmol/L [20.5% decrease]; both P < 0.05 versus baseline; Figure 2b). One month of treatment with 3 mg oral semaglutide daily did not significantly influence fasting plasma glucose values.
Effect of oral semaglutide on body weight
At 5 months of oral semaglutide treatment (1 or 2 months of receiving 14 mg daily), a significant decrease in body weight was observed compared with baseline values (from 100.9 ± 2.7 kg to 92.7 ± 2.4 kg [8.1% decrease]; P < 0.05 versus baseline; Figure 3). Oral semaglutide at doses of 3 and 7 mg had no statistically significant effect on body weight (Figure 3). Patients included in the present study were obese (mean BMI at inclusion, 34.6 ± 1.4 kg/m2, and at the end of the study, 30.8 ± 1.1 kg/m2).

Body weight at baseline (before starting oral semaglutide) and after treatment with oral semaglutide at doses of 3, 7 or 14 mg daily in 20 patients with type 2 diabetes mellitus. Data presented as mean ± SEM; *P < 0.05 versus baseline value.
Effect of oral semaglutide on blood pressure and lipoproteins
Systolic and diastolic in-office blood pressure values were measured at every visit, but changes during the study were insignificant. Lipoproteins were measured at inclusion and at the end of the study, but these values also did not change significantly.
Adverse events of oral semaglutide
After the initiation of oral semaglutide, patients most frequently reported gastrointestinal adverse effects that consisted of nausea, constipation, upper abdominal pain, and dyspepsia (Table 2). Overall, gastrointestinal adverse events occurred in 10 patients, were mild to moderate in severity, and occurred predominantly during the oral semaglutide dose escalation period. Four included patients reported abdominal pain, and laboratory results were determined, including pancreatic enzymes. No increase in pancreatic enzyme values was observed in these patients, therefore, oral semaglutide treatment was continued. The incidence of hypoglycaemia was low, with two patients reporting mild hypoglycaemia. Both patients received sulfonylurea as background diabetes treatment, and after modification of the sulfonylurea dose, no further hypoglycaemia occurrence was observed. No serious adverse events were observed that led to the discontinuation of oral semaglutide.
Frequency of adverse events during oral semaglutide treatment, and oral semaglutide dose at the time of their occurrence, in a population of 20 patients with type 2 diabetes mellitus.

Data presented as n (%) incidence.
Patient satisfaction with oral semaglutide treatment
All patients completed the TSQM. In questions 1–3, regarding efficacy of oral semaglutide, patients reported being very satisfied with the ability of oral semaglutide to treat diabetes, relieve symptoms of diabetes, and with the time it takes for the oral semaglutide to exert its effect (mean composite subscale score, 84.7 ± 2.2). In the group of questions relating to oral semaglutide side-effects, half of the patients reported side-effects in question No. 4 and subsequently completed questions 5–8. Side-effects were described as mild and without minimal interference with physical function and mental activity. Patients reported that side-effects minimally affected their overall satisfaction with treatment medication. In the group of convenience questions (questions 9–11), patients evaluated oral semaglutide treatment as very easy to use (mean composite subscale score, 83.2 ± 2.7) and planning the time to take the medication was also very easy. They reported a high global satisfaction with oral semaglutide treatment (questions 12–14; mean composite subscale score, 87.6 ± 1.7). Results of the TSQM questionnaire are presented in Figure 4.

Patient satisfaction with oral semaglutide treatment in 20 patients with type 2 diabetes mellitus according to the Treatment Satisfaction Questionnaire for Medication (TSQM) version 1.4. Each score has been converted to a value on a scale of 0–100. Question 4 is not presented, 10 patients reported side-effects and completed questions 5–8. Data presented as mean ± SEM.
Changes in key parameters
Changes in HbA1c, fasting plasma glucose and body weight during treatment with oral semaglutide are summarised in Table 3.
Changes in glycosylated haemoglobin (HbA1c), fasting plasma glucose and body weight during oral semaglutide treatment at doses of 3, 7 or 14 mg daily in 20 patients with type 2 diabetes mellitus.

Data presented as mean absolute change.
*P < 0.05 and **P < 0.01 versus baseline value (before oral semaglutide treatment).
Discussion
In the present study, the efficacy, safety, and satisfaction with oral semaglutide was explored in the first 20 consecutive patients with type 2 diabetes who were eligible for therapy in the outpatient clinic of the University Medical Centre Ljubljana. Oral semaglutide, at therapeutic doses of 7 mg and 14 mg daily, significantly decreased HbA1c and fasting plasma glucose values; at the 14 mg daily dose, it also significantly decreased body weight; and these effects were observed after 3–5 months of starting treatment. Patients reported being very satisfied with the treatment, referring to it as easy to use, and the global or general satisfaction level was high. Half of the patients reported mild transient adverse effects. Thus, oral semaglutide treatment appears to be a very effective and safe antidiabetic option. These results are important, as they represent one of the first experiences of oral semaglutide from a routine (real-world) clinical practice setting.
In summary, treatment with oral semaglutide by routine dose escalation was associated with significantly decreased HbA1c and fasting plasma glucose values after 3 to 5 months of treatment, once patients were receiving therapeutic doses of 7 or 14 mg daily (decrease of 1.2% and 1.6% HbA1c; and 17.9% and 20.5% decrease in fasting plasma glucose, with 7 mg or 14 mg semaglutide, respectively). In addition, the 14 mg oral semaglutide dose significantly decreased body weight by a mean of 8.1%, which is a very beneficial effect in overweight or obese patients with type 2 diabetes. Patients included in the present study were obese (mean BMI at inclusion 34.6 ± 1.4 kg/m2 and at the end of the study 30.8 ± 1.1 kg/m2). In addition to its effect on glycaemic parameters, the patient’s perception of treatment, patient satisfaction, and the occurrence of adverse effects were examined. According to results of the validated TSQM, patients reported being very satisfied with the ability of oral semaglutide to treat diabetes (84.7 ± 2.2); treatment was evaluated as easy to use (83.2 ± 2.7); global satisfaction with oral semaglutide treatment was high (87.6 ± 1.7). As TSQM was evaluated at the last study time-point, when all patients were receiving oral semaglutide at a dose of 14 mg, differences in treatment satisfaction between different oral semaglutide doses could not be evaluated. However, it should be noted that evaluation of patient satisfaction is most meaningful at the maximal therapeutic dose, as this is the targeted maintenance therapeutic dose. Therefore, patient satisfaction information at the evaluated time-point in the present study is the most valuable for daily clinical practice. Use of the TSQM allowed exploration of the most important dimensions of patient experiences with oral semaglutide treatment. The main advantages of the questionnaire are that it is easy to self-administer, it is standardized, it includes validated measures of patient satisfaction with treatment, and offers good metric properties of reliability and validity.12,14 One of the possible disadvantages of the TSQM used in the present study is that it is relatively complex and long, and therefore patients must have enough time to complete it satisfactorily. Furthermore, as with all questionnaires, patients may not have been entirely honest when completing the TSQM, as they may be biased by active questions related to the treatment offered to them. For several reasons, e.g., fear of injections or fear of needles, most patients included in the present study had rejected an initial proposal of once weekly semagluitde injection treatment prior to study inclusion. Therefore, patients may have falsely evaluated oral semaglutide treatment more positively, after primarily rejecting subcutaneous semaglutide treatment. Half of the present patients reported mild and transient adverse effects, which was lower that the incidence of adverse events reported in the PIONEER trials (57–85% of patients who received oral semaglutide). 7 The most frequent adverse events in the present study were gastrointestinal, such as nausea (reported by 30% of patients). Gastrointestinal side-effects were expected, due to the results of previous studies,15–17 and patients were warned of the possibility of their appearance. Importantly, these side-effects did not cause discontinuation of treatment.
The effectiveness of oral semaglutide on glycaemic parameters and body weight has been explored in previously published randomized controlled trials (RCTs), whereby the effects were compared with placebo or active comparators, such as injectable GLP-1RAs, SGLT-2 inhibitors, and other antidiabetic drugs. In the PIONEER studies, oral semaglutide significantly improved glycaemic control: the HbA1c values decreased by 1.1% with a 3 mg daily dose, 1.7% with a 7 mg daily dose and 2.0% with a 14 mg daily dose of oral semaglutide. 7 These results are in line with the present study, conducted in a routine clinical practice setting. In the PIONEER studies, body mass decreased by 2.4 kg with a 7 mg daily dose and 4.4 kg with a 14 mg daily dose of oral semaglutide. 7 As mentioned above, the previous studies were RCTs, which are conducted under strict study conditions that usually lead to better results compared with daily clinical practice. However, a comparable decrease was observed in the present study, and the observed effects appeared to be dose-dependent and more pronounced at higher doses of oral semaglutide. Therefore, the effectiveness of oral semaglutide appears to be comparable between the real-world setting and RCT conditions. Of note, oral semaglutide is shown to be effective as the third or fourth antidiabetic drug, and comparative antidiabetic efficacy has been observed regardless of the duration of diabetes. 7
A previously published clinical practice study that was retrospective in design, using data from a health record database, reported a 0.9% decrease in HbA1c at 6 months after starting oral semaglutide treatment. 10 Although this was the first study of oral semaglutide effectiveness in routine clinical practice, it differed from the present study in several ways, in that it was registry-based, performed in the USA, oral semaglutide dose escalation was very slow, and patient satisfaction was not evaluated. 10 Other real-world studies have mostly explored the effectiveness of once-weekly subcutaneous semaglutide. The use of once weekly semaglutide was shown to be associated with a statistically significant decrease in HbA1c (by 0.91%) after 24 months of follow-up, which was comparable to the present study. However, a higher reduction in body weight was observed compared with the present study (up to 12.4 kg), as expected. 18 In another study, persistence and adherence to injectable GLP-1RAs were explored. After 360 days, the highest persistence and highest adherence was observed in the semaglutide once weekly group compared with other GLP-1RAs. 19 However, to the best of our knowledge, there are no studies exploring patient satisfaction with oral semaglutide treatment in the clinical practice or real-world setting. Therefore, the present results could not be compared with previously published data.
No significant effect on blood pressure values was observed during oral semaglutide treatment in the present study, although initial values were in the range of hypertension; this contrasts with the results of some other studies, where significant decreases in systolic blood pressure values were observed.20,21 In the analysis of all PIONEER studies, blood pressure remained similar, or was slightly reduced over time, in patients treated with oral semaglutide compared with active comparators. 7 In addition, the present study did not reveal significant effects of oral semaglutide on lipid parameters. In the PIONEER 6 trial, low-density lipoprotein (LDL)-cholesterol and triglyceride levels were modestly lower in the oral semaglutide group, 21 while in other studies, a significant decrease in LDL-cholesterol and non-high-density lipoprotein-cholesterol was observed during oral semaglutide treatment. 20 In the study by Dahl et al., 22 a significant decrease in very low-density lipoproteins, apolipoprotein B48, and triglycerides was observed after 12 weeks of treatment with oral semaglutide at a dose of 14 mg daily.
The main strength of the present study is that the effectiveness of oral semaglutide, patient satisfaction with treatment, and adverse events were systematically explored in everyday clinical practice. Furthermore, satisfaction with oral semaglutide treatment and the occurrence of adverse events were also observed, which is very important to the patient’s perception of oral semaglutide treatment and influences adherence to treatment. Patients may have been very satisfied with oral semaglutide treatment because they avoided injectable treatment, which was among their greatest fears, and considering the analogy that injectable treatment is insulin, they perceived oral semaglutide as an escape option with very satisfactory results in terms of glycaemia control and body weight. Thus, it seems that, from the perspective of the healthcare worker, a relatively complex oral semaglutide administration regime did not represent an evident obstacle for patients, or their consideration of patient satisfaction. The study also has several limitations: it was not randomized or placebo controlled, and a low number of patients were included, due to restrictions in oral semaglutide availability during the study. No sample-size calculation was performed, and thus, the limited number of participants may have affected the statistical significance of the results. Another limitation was the relatively short duration of exposure to 14 mg semaglutide, which may lead to high variability in the results. However, this did not appear to influence the present results that supported the previously established high efficacy. Additional limitations of the study were that possible changes in the non-pharmacological treatment of diabetes (e.g., changes in lifestyle and physical activity), that may potentially influence the study results, were not monitored.
Conclusions
Therapeutic doses of oral semaglutide, after 3 to 5 months of initiating treatment, significantly improved glycaemic control and decreased body weight; patients were very satisfied with the treatment; and adverse effects were mild and transient. The effectiveness of oral semaglutide in everyday clinical practice was comparable to the results of previous RCTs. More studies are needed to explore in detail the effectiveness of oral semaglutide in clinical practice.
Footnotes
Author contributions
Conceptualization, MJ, ML and AJ; methodology, ML; formal analysis, ML; investigation, MJ; writing–original draft preparation, MJ, MJ, and ML; writing–review and editing, AJ; supervision, AJ; funding acquisition, AJ. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The datasets generated and/or analysed during the study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was funded by the University Medical Centre Ljubljana, Ljubljana, Slovenia: grant No. 20210043.
