Abstract
Objective
Multiple organ failure can occur as a result of postoperative complications. Research has indicated that the underlying mechanism of organ dysfunction is a microcirculation disorder. Because of its antioxidant and anti-inflammatory properties, lidocaine has the potential to improve microvascular blood flow. This study was performed to assess the effect of intraoperative intravenous lidocaine infusion on the microcirculation and determine the incidence of postoperative complications.
Methods
In this prospective randomized double-blind pilot study, 12 patients scheduled for abdominal surgery were randomly allocated to receive an intraoperative infusion of either 1% lidocaine or the same volume of 0.9% sodium chloride solution. The microcirculation was monitored using sidestream dark-field imaging and the vascular occlusion test combined with near-infrared spectroscopy.
Results
Lidocaine significantly increased the total vascular density and small vessel density after 2 hours of infusion, with preservation of 99% to 100% of the capillary perfusion in both groups. No patients developed organ failure.
Conclusions
An increase in vessel density may be beneficial in major abdominal surgeries because it is associated with better tissue perfusion and oxygen delivery. However, this finding requires further investigation in patients with increased surgical risk. Overall, this study indicates that lidocaine has potential to improve microvascular perfusion.
Keywords
Introduction
General anesthesia and surgery have a significant impact on the circulatory system and may lead to hemodynamic instability. Many factors contribute to intraoperative circulatory derangement, including anesthetic drugs, tissue injury, blood loss, and inflammatory responses. A wide variety of systemic circulation monitoring devices are used. They allow early detection and correction of hemodynamic abnormalities, and their use is crucial for optimizing blood flow to vital organs. However, the systemic circulation is not the only factor that plays an important role in tissue perfusion. The microcirculation, together with its autoregulatory processes, controls the supply of oxygen and nutrients as well as the removal of metabolic products. Microcirculatory flow disorders are not always associated with hemodynamic alterations. Changes in capillary perfusion may precede systemic circulatory disturbances and cause organ dysfunction during the postoperative period.1,2 Studies have shown that the microcirculation is not fully correlated with the systemic circulation; therefore, hemodynamic stabilization is not always followed by capillary recruitment and flow normalization.3–5
Therefore, microcirculation monitoring appears to be a promising tool for optimizing capillary perfusion and maintaining proper organ function. Particular attention has been paid to drugs with anti-inflammatory properties that can stabilize the microcirculation. Lidocaine is a promising drug because it is widely used intravenously in the perioperative period to reduce pain and minimize the need for opioids. It is also an important component of multimodal analgesia. 6
Administration of lidocaine during surgery reduces the use of opioids and anesthetics, leads to an earlier return of bowel movements in the postoperative period, and shortens the length of hospital stay.7–10 In addition, lidocaine has antioxidant and anti-inflammatory properties that interfere with many stages of the inflammatory cascade.11–14 It affects the production of inflammatory mediators and nitric oxide, the activation and migration of lymphocytes, and the permeability of capillary membranes. Therefore, lidocaine is expected to have an effect on the microcirculation. In major surgeries, there is a correlation between microvascular perfusion and organ dysfunction as a postoperative complication. 2 For this reason, lidocaine infusion may be beneficial for patients not only in terms of pain relief but also in terms of the postoperative course, incidence of complications, and length of hospital stay.
The primary outcome of this study was to assess the impact of intraoperative intravenous lidocaine infusion on the microcirculation. To achieve this, two distinct methods of microvessel assessment were employed: direct observation using a microcamera and indirect evaluation using the vascular occlusion test (VOT). Our secondary aim was to evaluate the potential influence of lidocaine on the incidence of postoperative complications, which can arise as a consequence of alterations in microcirculatory dynamics.
Materials and Methods
In this prospective, single-center, randomized, double-blind pilot study, we focused on the microcirculation in patients undergoing elective abdominal surgery (laparoscopic prostatectomy, bowel resection, and cystectomy) and receiving intravenous lidocaine. This study was conducted in accordance with the principles of the Declaration of Helsinki on Human Research of 1975 as revised in 2013. It was approved by the Ethics Committee of Wroclaw Medical University (Judgment Date: 23 August 2018, Judgment Number: 476/2018). In addition, the reporting of this study conforms to the CONSORT statement. 15
The pilot study group initially comprised 13 patients who were hospitalized at the University Clinical Hospital in Wroclaw (Poland) from October 2018 to May 2019 and scheduled for elective abdominal surgery. The day before surgery and after the medical assessment, written consent was obtained from each patient to participate in the study. The exclusion criteria were an age of <18 years, pregnancy, epilepsy or a history of documented arrhythmia, an American Society of Anesthesiologists (ASA) score of >3 (including severe heart, lung, kidney, and/or liver disease), body weight of <55 or >140 kg, severe peripheral arteriosclerosis, unilateral vascular disease of the upper limb (stroke or trauma), pathology of the sublingual mucosa, and allergy to general or local anesthetics.
On the day of surgery, the patients were randomly allocated to one of two groups based on the solution administered during anesthesia (parallel assignment): the lidocaine group or the placebo group. Randomization was always conducted by the same nurse with application of www.random.org. The results of randomization and group assignment were stored in closed envelopes by the nurse responsible for randomization and were revealed after the study had ended.
The solutions for both groups were prepared by the same nurse, who was not involved in the administration of anesthesia. The content of the study solution (labeled “SS”) was not known to the patient or team involved in the procedure. However, the nurse preparing the study solution was present in the operating department during anesthesia and patient recovery. In case of any adverse events, the anesthesiologist could contact the nurse, who was obliged to then reveal the contents of the study solution.
During the operation, the lidocaine group received 1% lidocaine as an intravenous infusion along with standard analgesics (systemic opioids, metamizole, and paracetamol). The placebo group received the same drugs except that lidocaine was replaced with normal saline.
We used two methods to monitor the microcirculation. The first method was based on direct imaging of the microcirculatory flow using a MicroScan® camera (MicroVision Medical, Amsterdam, the Netherlands). In the second method, the indirect VOT was combined with near-infrared spectroscopy (NIRS).
The MicroScan® is a handheld camera that uses the sidestream dark-field (SDF) technique to capture erythrocyte movement in the smallest vessels (terminal arterioles, capillaries, and venules). It emits green light with a wavelength of 530 nm from light-emitting diodes located around the tip of the camera. The light source is isolated from the central detecting and image-transmitting part. The emitted light passes through the mucous membrane, reaches a maximum depth of 3 mm below its surface, and illuminates structures located at shallow levels. Light is absorbed by the hemoglobin of erythrocytes; thus, vessels filled with erythrocytes become visible.
Using this device, we recorded three different sequences of microcirculatory flow in the sublingual area of the oral cavity. The films were further analyzed using AVA software Version 3.0 (MicroVision Medical). The total vessel density (TVD) and small vessel density (sVD) were calculated using software, whereas the percentage of perfused vessels (PPV) and microvascular flow index (MFI) were assessed by the investigator, as described by Boerma et al. 16 During imaging, efforts were made to remove all saliva from the mucosal surface. We also reduced the pressure exerted by the camera on the observation area to avoid vascular compression.
The VOT was used for indirect assessment of the microcirculation. In this test, a sphygmomanometer on the patient’s right arm was inflated to a pressure 30 mmHg above the systolic pressure, inducing limb ischemia and maintaining it for 3 minutes. Tissue oximetry was simultaneously monitored over the patient's right forearm every 15 s using an NIRS device connected to a tissue oximetry sensor (FORE-SIGHT; CASMED, Branford, CT, USA). After cuff deflation, oximetry measurements were performed every 5 s for the first minute and every 15 s for the next 3 minutes.
This method allows for the assessment of dynamic changes in tissue blood supply under conditions of artificially created ischemia. Thus, it indirectly indicates the state of the microcirculation. Of particular importance are the rate of the decrease in saturation during ischemia (the occlusion downslope), the minimum value of saturation, and the rate of the increase in saturation after releasing the pressure of the sphygmomanometer (the reperfusion slope). It is a relatively new method but has proven application in critical conditions, such as sepsis and trauma.17,18
The patients fasted for 6 hours before surgery. They were allowed to drink water for up to 2 hours before surgery. The patients were premedicated with oral midazolam (7.5 mg). All study participants were monitored in a standard manner upon arrival in the operating room. Prior to induction, the patients received 1 g of paracetamol intravenously and 1 g of metamizole intravenously. General anesthesia was induced with remifentanil (0.25–1 µg/kg/minute), propofol (1.5–2.5 mg/kg), and rocuronium (0.6 mg/kg). All patients were intubated and ventilated using a Datex-Ohmeda Aespire anesthesia machine (GE Healthcare, Chicago, IL, USA) with a tidal volume of 6 to 8 mL/kg. The ventilator settings were further corrected based on the blood oxygen saturation, capnography, and blood gas analysis.
Before induction, the patients were given a 0.15-mL/kg bolus of the study solution (1% lidocaine in the lidocaine group and 0.9% sodium chloride solution in the placebo group) over 10 minutes, and infusion was then started at 0.2 mL/kg/hour. The lidocaine doses of 1.5 mg/kg for the bolus and 2.0 mL/kg/hour for the infusion were based on guidelines and research described elsewhere.6,19
Further anesthesia was maintained with sevoflurane at a concentration of 1.5 to 2.5 vol%, continuous infusion of remifentanil (0.05–2 µg/kg/minute), and boluses of rocuronium (5–10 mg based on neuromuscular block monitor recordings).
All patients were hemodynamically monitored during anesthesia. After anesthetic induction, an arterial line was placed in the left radial artery and connected to a FloTrac system and EV1000 monitor (Edwards Lifesciences, Irvine, CA, USA). We recorded the stroke volume, indexed stroke volume, stroke volume variation (SVV), cardiac output, and cardiac index.
Throughout the intraoperative period, the patients received a crystalloid solution at an infusion rate of 8 mL/kg/hour. Fluid management was based on a protocol described elsewhere. 20 If the SVV was >12%, a 250-mL crystalloid bolus was administered. If the SVV was <8%, the patients were treated with norepinephrine and/or dobutamine based on the mean arterial pressure and cardiac index.
In both groups, SDF imaging, occlusion tests, and hemodynamic measurements were performed at hourly intervals starting from anesthetic induction. Simultaneously, arterial blood was collected and analyzed to determine the lactate concentration, pH, partial pressures of oxygen and carbon dioxide, hematocrit, and hemoglobin concentration.
After the operation, the patients were transferred to the recovery room and then to the original wards of the University Clinical Hospital in Wroclaw. Complications and length of hospital stay were recorded in the following days. Complete blood counts and creatinine levels were routinely tested on the first postoperative day.
Statistical analysis
The results were tested for normality using the Shapiro–Wilk test. Depending on the result of the normality test, we used either Student’s t-test or its non-parametric equivalent, the Mann–Whitney U test. The significance of changes in the given parameters over time was determined using Friedman’s analysis of variance followed by a post hoc test (Dunn’s test). Statistical significance was set at p < 0.05. However, we did not perform a sample size calculation, and the limited sample size may have affected the statistical significance of our results.
Results
Trial recruitment
Of the 29 patients assessed for eligibility, 16 were excluded (Figure 1). Six patients met more than one exclusion criterion, eight refused to participate, and two were excluded for technical reasons (discharged MicroScan® battery and limited access to the NIRS sensor). The remaining 13 patients were randomly assigned to the two study groups. Finally, one patient was excluded from the analysis because of the duration of the operation (<2 hours).

CONsolidated Standards of Reporting Trials (CONSORT) flow diagram presenting patients’ recruitment and allocation.
Baseline characteristics
The patients’ baseline characteristics are shown in Table 1. There were no significant differences between the two groups. In both groups, comorbidities were mild and well-controlled, and all patients had an ASA score of 1 or 2.
Baseline characteristics of study groups.
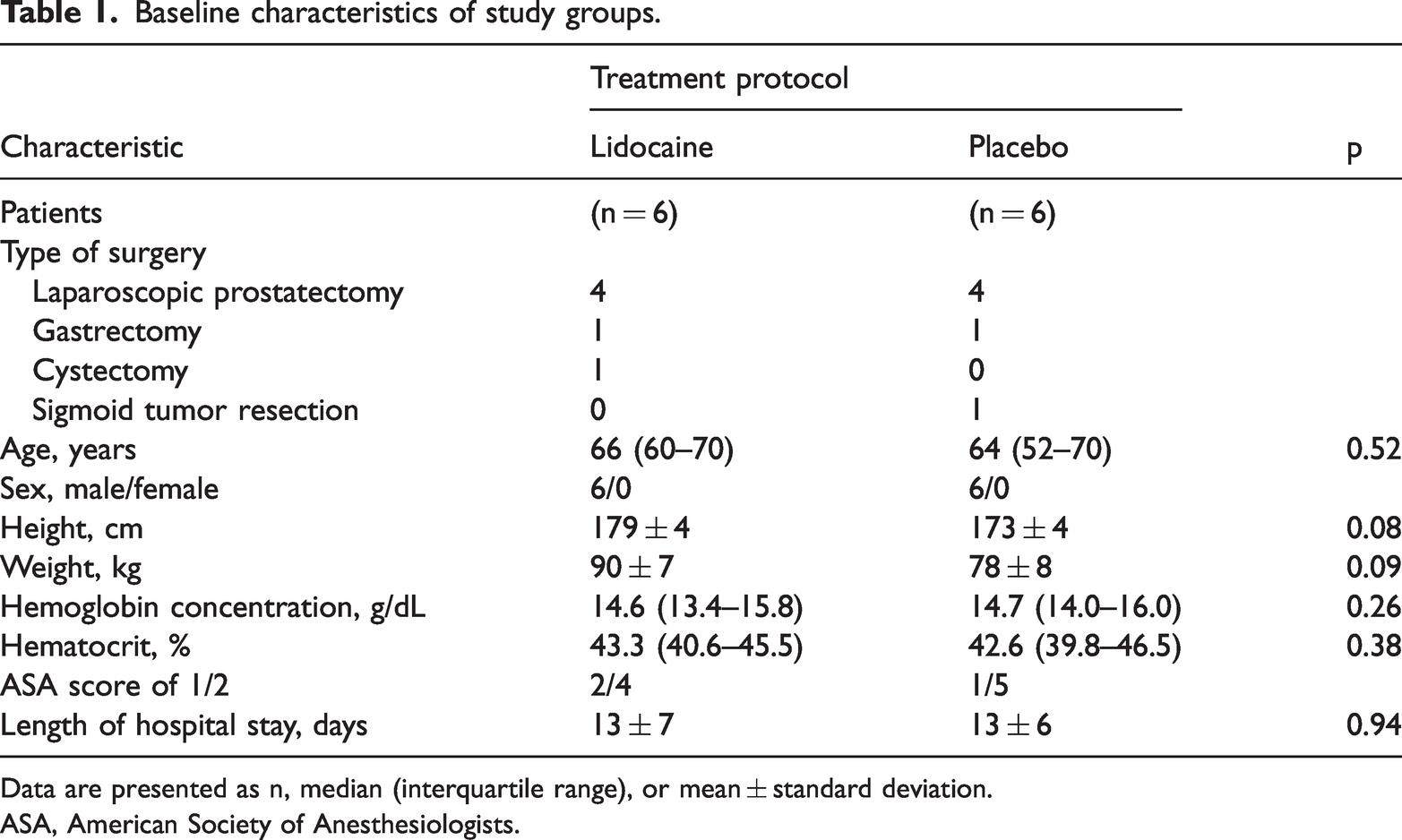
Data are presented as n, median (interquartile range), or mean ± standard deviation.
ASA, American Society of Anesthesiologists.
All patients were male except one, who was not included in the final analysis because of the duration of the operation. The high number of male patients can be explained by the fact that most of the enrolled patients were scheduled for prostatectomy.
Microcirculation in SDF imaging
During the procedure, the microcirculation was well preserved in both groups. Within 2 hours of observation, almost all microvessels were perfused with blood, with the PPV reaching 99% to 100% and the mean MFI equal to 3.0 in both study groups (Table 2). The TVD and sVD in the lidocaine group significantly increased after 2 hours (ΔTVD = 1.3 mm/mm2 and ΔsVD =1.3 mm/mm2, p < 0.05 for both variables) (Figure 2). No significant changes in these variables were observed in the placebo group. The final difference in the TVD and sVD between the groups was statistically significant (ΔTVD = 1.5 mm/mm2 and ΔsVD = 1.1 mm/mm2, p < 0.05 for both variables).
Microcirculation variables recorded with sidestream dark-field imaging in the study groups.

*p < 0.05.
Data are presented as median (interquartile range).
TVD, total vessel density; pVD, perfused vessel density; sVD, small vessel density; psVD, density of perfused small vessels; sMFI, microvascular flow index of small vessels (<20 μm); HI, heterogeneity index.

Evolution of (a) total vessel density (TVD) and (b) small vessel density (sVD) during anesthesia and surgery in groups receiving lidocaine and placebo infusions. *p < 0.05 between the two study groups (inter-group analysis); #p < 0.05 within the lidocaine group compared with basal values (intra-group analysis).
Microcirculation in VOT
The results of the dynamic assessment of the microcirculation are presented in Table 3. The initial tissue oxygen saturation, its fluctuation in consecutive phases of the VOT, and the steepness of the occlusion and reperfusion slopes did not differ significantly between the groups. All VOT-derived parameters were stable at all time points.
VOT-derived variables in the study groups.

Data are presented as median (interquartile range).
No statistically significant differences were observed between the VOT parameters in the study groups (p > 0.05).
VOT, vascular occlusion test; StO2, tissue oxygen saturation.
Hemodynamic variables
Implementation of the Cannesson fluid protocol 20 resulted in a very stable anesthesia course, with no significant intergroup differences in hemodynamic variables. The hemoglobin, hematocrit, and lactate levels in the lidocaine group were comparable to those in the placebo group. None of the patients required a red blood cell transfusion during the 2-hour observation period.
Postoperative assessment
The patients in both groups presented a stable postoperative course, with only one complication in the lidocaine group (anastomotic leakage) (Table 4). The mean length of hospitalization was the same in both groups.
Hemoglobin concentration, platelet count, creatinine on the first postoperative day, length of hospital stay, and complications in the study groups.

Data are presented as median (interquartile range), mean ± standard deviation, or n.
POD 1, postoperative day 1.
Discussion
The main finding of this study is that in the group of patients receiving lidocaine infusion during abdominal surgery, the TVD and sVD significantly increased in the sublingual microcirculation with the duration of anesthesia. However, no such association was observed in the placebo group. Moreover, after a 2-hour drug infusion preceded by a bolus, both variables were also significantly higher in the lidocaine group than in the placebo group. Combined with the high PPV of 99% to 100%, the above condition indicates increased perfusion of the sublingual area. This could have both positive and negative effects.
A benefit of densification of the capillary network is that oxygen must travel a shorter distance through the interstitial space to reach its target cells. Consequently, the risk of tissue hypoxia and anaerobic processes is reduced. This phenomenon has been well-described under conditions of sepsis. Heterogeneity of microvascular perfusion and local reduction in microvessel density create unfavorable conditions for oxygen extraction and promote anaerobic metabolism, ultimately leading to organ failure.21,22 Therefore, resuscitation of the microcirculation in vital organs seems to be a new goal in the prevention of organ failure, not only in sepsis. In addition, several studies have shown that poor perfusion of the gastrointestinal mucosa increases the risk of anastomotic dehiscence after surgery.23,24 These data suggest that an increase in the TVD and sVD may be beneficial in patients undergoing major abdominal surgeries because such increases are associated with better tissue perfusion and oxygen delivery. Increased perfusion of organs not only guarantees better oxygenation but also creates better conditions for wound healing. This phenomenon is associated with the presence of collateral microvessels that are ready to supply the site of injury. 25
It should be noted that a homogeneous increase in the density of perfused vessels can lead to even distribution of blood to all tissues and hemodynamic instability with poor perfusion of vital organs. Dilatation of all microvessels would introduce an incredible challenge for the heart as a pump. This implies the need to increase the output to 50 L/minute. 26 Previous studies have shown that lidocaine causes hemodynamic depression.27,28 In our study, hemodynamic stabilization in both groups was achieved with goal-directed fluid therapy, which resulted in statistically insignificant hemodynamic differences between the groups. However, the consequences of this in patients with septic shock are unknown. Centralization of the circulation is a defense mechanism. Its purpose is to protect vital organs by ensuring adequate and minimal blood perfusion through the key organs. Further research is needed to determine whether lidocaine would cause a similar effect in patients with severely disturbed microcirculation (e.g., septic shock), whether it would be homogeneous within the entire microcirculation, or what the consequences would be.
In the present study, we did not observe increased bleeding in the lidocaine group despite an increase in capillary density. None of the patients required packed red blood cell transfusions, and the hemoglobin concentrations remained stable during and after surgery. Assuming that the coagulation cascade is initiated immediately after injury, it can be presumed that increased microcirculation perfusion should not be a significant problem in the context of perioperative bleeding. 25
During the course of the study, no patients developed postoperative organ failure, and no other serious life-threatening complications (e.g., infection or hemorrhage) were noted. We recorded one case of anastomotic dehiscence. These findings can be explained by the fact that we enrolled relatively healthy patients (ASA scores of 1 and 2) in this pilot study, and most of them underwent laparoscopic surgery associated with limited tissue damage. Therefore, further studies involving high-risk patients are needed to determine whether increasing the vascular density by lidocaine infusion affects the frequency of complications after major surgical procedures. Pustetto et al. 29 raised a similar issue after they failed to prove the protective and anti-inflammatory effects of lidocaine on the endothelium during major abdominal surgeries. The authors concluded that in major abdominal surgeries, endothelial damage was overestimated because the concentration of Syndecan-1 (an integral membrane protein and a marker of endothelial glycocalyx damage) was only moderately increased. 29 Bansch et al. 30 also concluded that microcirculation disorders detected by SDF imaging seem to be minor and that there is no correlation between complications of major abdominal surgeries and sublingual capillary perfusion. By contrast, Jhanji et al. 2 showed that impaired microcirculatory flow is associated with complications after major abdominal surgery. Unexpectedly, capillary flow abnormalities were observed not only during but also before surgery, and they normalized within 8 hours after surgery. This finding leads to the hypothesis that surgery itself may not cause microcirculatory flow impairment and further organ complications, but certain preoperative factors (e.g., hypovolemia) that affect capillary flow before surgery may lead to organ failure in the postoperative period. 31 This could also explain the development of microcirculation disorders in patients with trauma.3,32 Research has shown that major surgical procedures can activate the inflammatory response and cause damage to the endothelial glycocalyx.33,34 However, what determines this reaction and how it affects the microcirculation remains unclear.
Similar to the findings reported by Pustetto et al., 29 we found no differences in the dynamic properties of the microcirculation expressed by the variables of the VOT. In many studies, the VOT in combination with NIRS has proven valuable for functional assessment of the microcirculation. The VOT provides information on the static properties of the microcirculation, expressed as tissue oxygen saturation, as well as the dynamic properties of the capillary bed. The rate of decrease in tissue saturation during ischemia (occlusion slope) and its minimum value reflect the adequacy of the oxygen supply for the organs’ metabolic demand. The rate at which blood flow is restored in the area affected by ischemia (reperfusion slope) proves the size of its reserves and the recruitment potential of the microcirculation. 35 Unfortunately, use of the VOT and tissue saturation monitoring by NIRS, although widely used for dynamic assessment of the microcirculation, have not yet been standardized. The obtained values may vary depending on the ischemic protocol (cuff deflation threshold), location of the probe, and the size of the NIRS probe used. 36
The present study had some limitations. The microcirculation was only observed in the sublingual area. All conclusions are true only if they can be extrapolated to other tissues. Unfortunately, microcirculation autoregulation and capillary permeability vary depending on the organ and are crucial for proper blood supply and functioning of the brain, liver, kidneys, heart, and muscles. 24 By contrast, the sublingual and gastrointestinal mucosae have the same embryological origins. Creteur et al. 37 showed that microcirculatory flow represented by the partial pressure of carbon dioxide in the sublingual area is correlated with changes in capnometry measured in the gastric mucosa. This finding contrasts with later studies showing that capillary perfusion in the sublingual and intestinal microcirculation does not always correlate in patients with sepsis. 38
Additionally, this study did not clarify the mechanism of the capillary bed opening. Like other aminoamide local anesthetics, lidocaine has a biphasic effect on vascular smooth muscles. 38 Low concentrations of lidocaine cause peripheral vasoconstriction. Peripheral arterial dilation occurs at higher drug concentrations. Satoh et al. 39 showed that a lidocaine concentration of 1 μg/mL caused lingual and pulmonary artery contractions in pigs. Higher concentrations (10–100 μg/mL) have a vasodilatory effect. In our experiment, we did not measure the concentration of lidocaine in the serum; based on previous studies, however, we can conclude that it exceeded 1 μg/mL. 40 Earlier studies on the vasoconstricting and vasodilating properties of lidocaine focused on arteries, not the microcirculation. The regulation of the capillary bed differs significantly from that of the macrocirculation. Hemodynamic changes do not strictly reflect the state of the microcirculation.41,42 This can be explained by the fact that the microcirculation is regulated at multiple levels and by numerous factors. Systemic hemodynamics have a significant impact on the microcirculation because of the mechanical autoregulation of smooth muscle tension of precapillary resistance vessels in response to changes in blood pressure (Bayliss effect). 43 Moreover, arterioles and venules receive autonomic adrenergic innervation (except for the gastrointestinal system and genitals, where microvessels are also supplied by cholinergic nerves). 26 These two mechanisms may be directly related to the macrocirculation. A similar hemodynamic profile was observed in both groups, and adrenergic stimulation was blunted by anesthesia. However, other factors such as humoral and tissue-generated agents (e.g., bradykinin, vasopressin, angiotensin II, free catecholamines, and bradykinin), local metabolic products (lactate, pH, and partial pressures of oxygen and carbon dioxide), autacoids (nitric oxide and prostaglandin), and shear stress are more closely related to the local tissue status and may differ from organ to organ. The question remains as to which of these processes are influenced by lidocaine. Perhaps the analysis of local humoral and tissue factors, as well as metabolic products, will bring us closer to answering this question.
Finally, our pilot study included a limited number of patients and required follow-up with a larger group of patients to confirm the original conclusions. Because of the lack of observed microcirculation disorders, more severely ill patients undergoing more invasive procedures should be included in future studies.
Conclusions
In patients receiving continuous intravenous lidocaine infusion during the course of abdominal surgery, the TVD and sVD assessed in the sublingual circulation significantly increased with the duration of anesthesia. However, this relationship was not observed in the placebo group. The clinical significance of this phenomenon requires further investigation in studies that include patients with increased surgical risk, an extended follow-up time, and assessment of other humoral and tissue damage markers.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231209820 - Supplemental material for Intraoperative intravenous infusion of lidocaine increases total and small vessel densities of sublingual microcirculation: a randomized prospective pilot study
Supplemental material, sj-pdf-1-imr-10.1177_03000605231209820 for Intraoperative intravenous infusion of lidocaine increases total and small vessel densities of sublingual microcirculation: a randomized prospective pilot study by Małgorzata Grotowska and Waldemar Gozdzik in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231209820 - Supplemental material for Intraoperative intravenous infusion of lidocaine increases total and small vessel densities of sublingual microcirculation: a randomized prospective pilot study
Supplemental material, sj-pdf-2-imr-10.1177_03000605231209820 for Intraoperative intravenous infusion of lidocaine increases total and small vessel densities of sublingual microcirculation: a randomized prospective pilot study by Małgorzata Grotowska and Waldemar Gozdzik in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605231209820 - Supplemental material for Intraoperative intravenous infusion of lidocaine increases total and small vessel densities of sublingual microcirculation: a randomized prospective pilot study
Supplemental material, sj-pdf-3-imr-10.1177_03000605231209820 for Intraoperative intravenous infusion of lidocaine increases total and small vessel densities of sublingual microcirculation: a randomized prospective pilot study by Małgorzata Grotowska and Waldemar Gozdzik in Journal of International Medical Research
Footnotes
Author contributions
Conceptualization, W.G. and M.G.; methodology, W.G. and M.G.; formal analysis, W.G. and M.G.; investigation, M.G.; data curation, M.G.; writing—original draft preparation, M.G.; writing—review and editing, W.G.; supervision, W.G.; project administration, W.G. and M.G. Both authors have read and agreed to the published version of the manuscript.
Data availability statement
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
