Abstract
The differential diagnosis of isolated ophthalmoplegia includes a range of pathologies. In this case, a 26-year-old man of Han nationality presented with ophthalmoplegia. Neuroimaging revealed an atypical focal lesion in the interpeduncular fossa. Initial systemic workup indicated intracranial Mycobacterium tuberculosis infection, but there was no evidence to support a diagnosis of other autoimmune diseases (e.g., myasthenia gravis or multiple sclerosis). Neuroimaging follow-up over the next 3 years revealed progression from atypical solitary lesions to multifocal lesions in the white matter of the brain. Key immunological markers were observed in cerebrospinal fluid during follow-up, suggesting the evolution of multiple sclerosis. Ophthalmoplegia with a focal lesion in the interpeduncular fossa was an unusual set of symptoms indicating multiple sclerosis onset. The findings in this case suggest that M. tuberculosis infection is an important but overlooked factor involved in the pathogenesis of multiple sclerosis. Upon initial detection, atypical lesions should receive sufficient attention and patients should undergo systematic screening to identify M. tuberculosis infection and its associated immunological abnormalities.
Keywords
Introduction
The differential diagnosis of isolated ophthalmoplegia ranges from benign to potentially lethal pathologies, including trauma, aneurysm, infections, autoimmune disorders, tumor, and ischemia.1,2 Lesions causing ophthalmoplegia are often located in the brainstem or cranial nerves. Here, we describe a 26-year-old man of Han nationality who presented with ophthalmoplegia; neuroimaging revealed an atypical focal lesion in the interpeduncular fossa. Initial systemic workup indicated intracranial Mycobacterium tuberculosis (TB) infection, but there was no evidence to support a diagnosis of other autoimmune diseases (e.g., myasthenia gravis or multiple sclerosis [MS]). Although the initial findings did not suggest an immunological etiology, subsequent clinical monitoring led to a diagnosis of ophthalmoplegia with suspected local inflammation or TB-related immunological abnormalities in the interpeduncular fossa, despite the absence of pathological data. The patient exhibited adequate recovery after receiving antitubercular medication and corticosteroids. Magnetic resonance imaging (MRI) follow-up over the next 3 years revealed a gradual increase in white matter lesions, although there were no other symptoms or signs of TB recurrence. On readmission 16 months after the initial symptoms, oligoclonal bands (OBs) were detected in cerebrospinal fluid (CSF) but not in serum. Thus, the patient was diagnosed with MS on the basis of clinical and laboratory findings. This report highlights the atypical nature of ophthalmoplegia with a focal lesion in the interpeduncular fossa as symptoms indicating MS onset. On initial admission, the presence of TB in CSF hindered a clear diagnosis, but the findings during 3 years of follow-up led to a definitive diagnosis.
Case report
A 26-year-old man of Han nationality presented to our hospital with a 10-day history of diplopia and bilateral ptosis. He reported bilateral diplopia and slight dizziness. He had no prodromal symptoms, such as headache, nausea, and fever. He denied any medical history or potentially relevant history of infection, including TB. Physical examination revealed normal temperature and blood pressure. The patient’s systemic findings were generally unremarkable. His visual acuity was normal. He exhibited incomplete bilateral ptosis, which was not related to fatigue. Examination of extraocular movements revealed slightly medial rectus palsy in the right eye; during leftward gaze, the patient displayed bilateral, symmetrical, low-amplitude, and low-frequency horizontal nystagmus. Pupillary sizes, shapes, and light reflexes were normal. The patient had normal muscle strength in the upper and lower extremities, without pyramidal signs or sensory disturbances. His reflexes and coordination were normal, and no other neurological abnormalities were noted. The neostigmine test results were negative.
In terms of laboratory findings, complete blood count revealed generally normal findings (red blood cells, 5.23 × 1012/L; white blood cells, 6.22 × 109/L; neutrophilic granulocytes, 47.5%; lymphocytes, 41.2%; and hemoglobin, 163.6 g/L). Blood glucose, renal function, and liver function results were normal. The erythrocyte sedimentation rate was slightly elevated (20 mm/hour). Analyses of the following serum markers for autoimmune and connective tissue diseases revealed negative results: antinuclear antibody (ANA), anti-dsDNA, anti-nucleosome, anti-phospholipid antibody (APLA), anti-histone, anti-Sm, anti-SS-A, anti-Ro, anti-Scl-70, anti Rib-P-protein, anti-JO, and anti-SS-B. Immunoglobulins IgG, IgA, and IgM were normal; complement C3 and C4 were also normal. Serology findings regarding hepatitis B virus, hepatitis C virus, and human immunodeficiency virus were unremarkable. The patient’s intracranial pressure was normal (125 mmH2O).
CSF examinations revealed normal findings (protein, 0.32 g/L; white blood cells, 0; glucose, 4.4 mmol/L; and chloride, 123.3 mmol/L). Antibodies to the following neuromuscular receptors were absent from serum and CSF: acetylcholine receptor (AChR), Musk, Titin, low-density lipoprotein receptor-related protein 4 (LRP4), ryanodine receptor (RyR), and voltage-gated calcium channels (VGCCs). Moreover, antibodies to aquaporin-4, myelin oligodendrocyte glycoprotein (MOG), and myelin basic protein (MBP) were absent from serum and CSF; no OBs were detected. There were no autoimmune encephalitis-associated antibodies, including antibodies to N-methyl-D-aspartate receptor (NMDA-R), contactin-associated protein-like 2 (CASPR2), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPA-R), γ-aminobutyric acid B receptor (GABAB-R), and glutamic acid decarboxylase 65-kilodalton isoform (GAD65). There were no anti-ganglioside antibodies, such as antibodies to the gangliosides GM1, GQ1B, and GB1B. The findings for thyroid-associated antibodies (e.g., thyroglobulin antibody [TGAb], thyroid peroxidase antibody [TPOAb], and thyroid-stimulating hormone receptor antibody [TRAb]) were normal.
Blood and CSF culture results were negative. Serologic tests for tubercular bacillus, rubella, Epstein–Barr virus, Lyme disease, and Brucella revealed negative results. The skin tuberculin sensitivity test result was positive. Polymerase chain reaction (PCR) analysis of three consecutive sputum samples revealed positive TB findings. PCR revealed TB in CSF, but it did not show evidence of herpes simplex virus. Acid-fast bacillus staining and a Brucella agglutination test using CSF samples both showed negative results.
Brain MRI performed 7 days after disease onset revealed a bright lesion in the interpeduncular fossa on T2-weighted-fluid-attenuated inversion recovery (T2-FLAIR) and diffusion-weighted imaging (DWI) (Figure 1). Magnetic resonance angiography showed no clinically significant abnormalities in the intracranial blood vessels. Ultrasound examination of the thyroid demonstrated normal findings. Pulmonary computed tomography also revealed normal findings.

Magnetic resonance imaging performed 7 days after disease onset revealed a bright lesion in the interpeduncular fossa (arrow) on T2-weighted-fluid-attenuated inversion recovery (T2-FLAIR) (c) and diffusion-weighted imaging (DWI) (d). T1-weighted (a) and T2-weighted (b) images at the same level do not clearly show the lesion. Images of layers above the lesion, taken for comparison with follow-up examinations, reveal normal brain tissue (e, T1-weighted; f, T2-weighted; g, T2-FLAIR; h, DWI).
Physical examination revealed bilateral ptosis and right medial rectus paralysis, which indicated bilateral incomplete extraocular muscle palsy. The presence of the lesion in the ventromedial region of the midbrain explained the clinical manifestations of third cranial nerve palsy. Considering the site of the lesion, pathological examination could not be performed. Systemic workup indicated intracranial TB infection, but there was no evidence to support a diagnosis of other autoimmune diseases (e.g., myasthenia gravis or MS). Laboratory investigations revealed intracranial TB infection, although the patient had no symptoms of pulmonary TB or tuberculous poisoning. Thus, the patient was diagnosed with ophthalmoplegia, intracranial TB infection, and a focal lesion in the interpeduncular fossa (suspected to result from local inflammation or immunological abnormalities). After obtaining informed consent from the patient, we cautiously administered dexamethasone at a dose of 10 mg/day for 7 days, along with oral antitubercular medication. The patient exhibited adequate recovery after the initial hospitalization.
Over the next 3 years, the patient consistently attended follow-up visits. He did not have any symptoms or neurological signs at the 16-month follow-up. MRI performed at the 16-month follow-up revealed lesion enhancement in the interpeduncular fossa on T2-FLAIR, as well as new lesions (without significant enhancement) around the anterior and posterior horns of the lateral ventricles (Figure 2). The patient was re-admitted and underwent repeat lumbar puncture to allow screening for infection-related and immunological etiologies. Serum and CSF examinations showed no indication of antibodies to aquaporin-4 or MOG. Similar to the findings on initial admission, PCR revealed TB in CSF. Furthermore, OBs were detected in CSF (16 months after initial symptom onset). Considering these findings, including the presence of new lesions on MRI, the patient was diagnosed with MS based on the 2017 revised McDonald criteria.3,4 However, the patient refused long-term disease-modifying therapy for various reasons, such as his previous diagnosis of intracranial TB, as well as his socioeconomic situation. At the most recent follow-up (3 years after initial onset), the patient’s overall status was good. However, MRI revealed new lesions (without significant enhancement) in the white matter of the left temporal lobe (Figure 3), further supporting the diagnosis of MS. The patient has since accepted disease-modifying treatment with siponimod (Novartis); the effects will be closely monitored during subsequent follow-up.

Magnetic resonance imaging performed at the 16-month follow-up revealed lesion enhancement in the interpeduncular fossa on T2-weighted-fluid-attenuated inversion recovery (T2-FLAIR) (c), as well as new lesions (without significant enhancement; d, h) around the anterior and posterior horns of the lateral ventricles (arrows, e–g). T1-weighted (a) and T2-weighted (b) images at the same level do not clearly show the lesion.
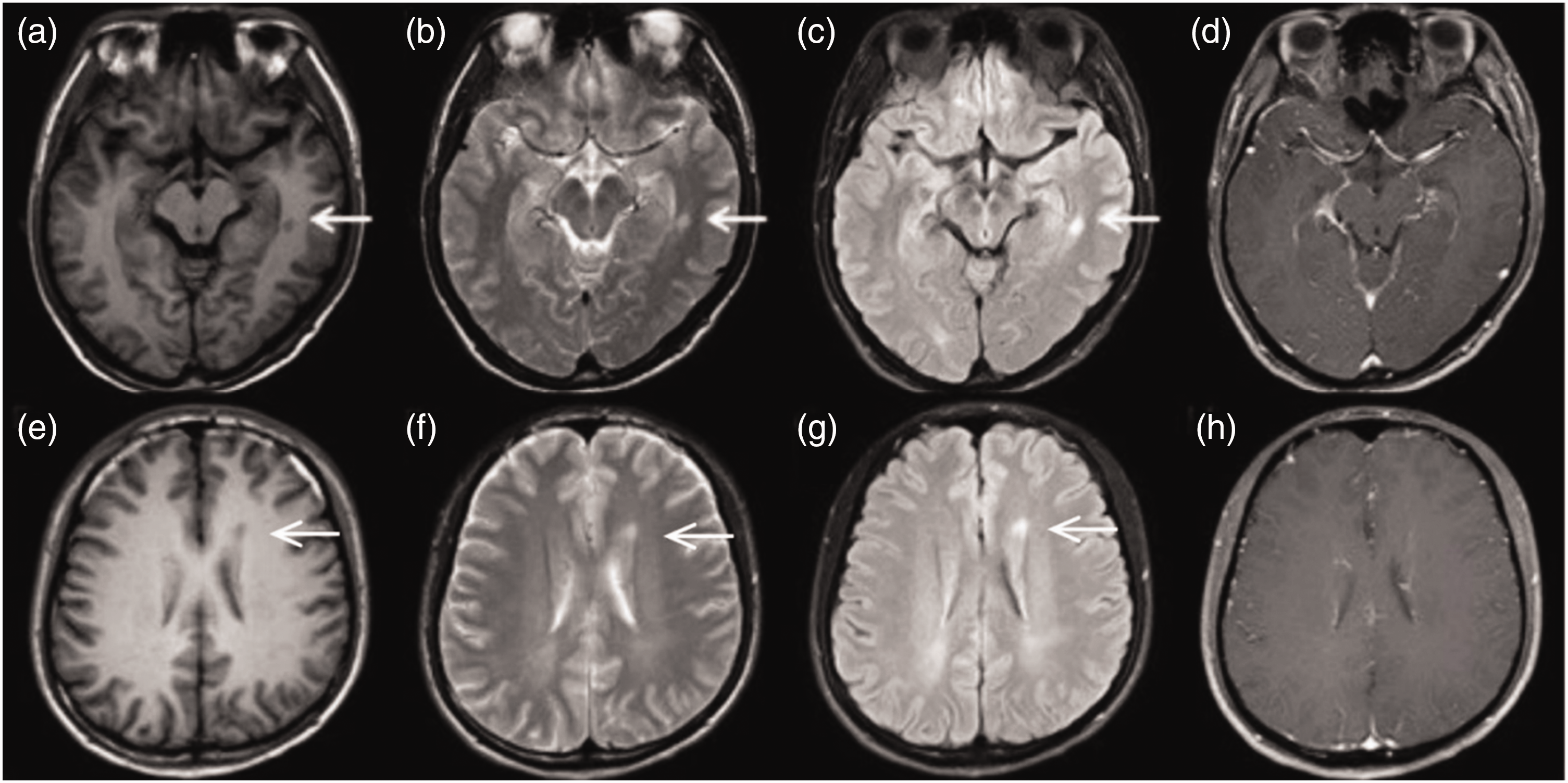
Magnetic resonance imaging performed at the 3-year follow-up revealed new lesions (without significant enhancement; d) in the white matter of the left temporal lobe (arrows, a–c). Previously identified lesions around the anterior and posterior horns of the lateral ventricles (e–g) were inconspicuous compared with previous findings (see Figure 2e–g). Lesions without significant enhancement (h) were unchanged relative to previous findings (see Figure 2h).
Discussion
Although the initially vague diagnosis was clarified over 3 years of follow-up, some aspects merit further discussion, such as the qualitative diagnosis of the initial specific focal lesion in the interpeduncular fossa because OBs were absent from serum and CSF, and the role of TB infection in the early onset of MS.
At the onset of disease, the patient presented with bilateral incomplete extraocular muscle palsy, as well as a specific focal lesion in the interpeduncular fossa. The presence of the lesion in the ventromedial region of the midbrain explained the clinical manifestations of third cranial nerve palsy. The first challenge was to clarify the nature of the lesion and establish a treatment plan. Tests to identify autoimmune etiologies of ophthalmoplegia (e.g., thyroid dysfunction and myasthenia gravis) involved the detection of specific antibodies. Laboratory investigations revealed intracranial TB infection, although the patient had no symptoms of pulmonary TB or tuberculous poisoning. Considering the site of the lesion, pathological examination could not be performed. Because PCR-based detection of M. tuberculosis in CSF is a highly sensitive and specific test,5,6 we suspected that the ophthalmoplegia was associated with focal inflammation or an unknown TB-related immune disorder. Because of this suspicion, short-term corticosteroids were administered. Corticosteroids are generally accepted in the treatment of central nervous system TB. 7 We believe that short-term corticosteroids can complement antitubercular medication. Although we did not have access to pathological data in this case, we hypothesized that TB shares antigens with MBP; thus, lymphocytes that recognize TB may also react to myelin. MS may develop via TB-related antigens. This antigenicity hypothesis requires further experimental confirmation, particularly involving reliable animal models.
The role of infections, such as TB, in the pathogenesis of MS has been discussed but remains unclear. Graf et al. explored infectious risk stratification among patients with MS; they found that latent TB infection was frequent (>1%), suggesting that screening should be performed before the initiation of immunomodulatory therapies to reduce the risk of reactivation during treatment. 8 Associations between MS/neuromyelitis optica spectrum disorder and TB have been suggested by the authors of multiple case reports and case series; the most likely mechanism is regarded as immune-mediated inflammatory demyelination of the central nervous system triggered by TB infection.9–11
In this case, OBs were detected in CSF 16 months after initial symptom onset, and MRI revealed the presence of new lesions in the brain. Accordingly, the patient was diagnosed with MS according to the 2017 revised McDonald criteria.3,4 Follow-up revealed the presence of additional lesions in the brain, without symptoms of TB recurrence. In most patients, there is insufficient evidence of temporal and spatial multiplicity during the first episode of MS, hindering diagnosis. If a patient is initially suspected to exhibit an immune-related inflammatory demyelinating disease, close monitoring should be implemented to clarify the diagnosis, thereby reducing the interval between onset and diagnosis, guiding subsequent treatment, and improving patient prognosis. The repeated PCR results indicating TB in CSF are somewhat suspicious, considering the absence of TB symptoms. We speculate that the patient’s asymptomatic status was related to his history of possible TB in childhood, as well as his good immune status during the course of admission and follow-up. However, the two positive test results cannot entirely rule out the possibility of laboratory error, considering the absence of TB symptoms. We plan to continue monitoring this patient for further insights.
The findings in this case, during 3 years of neuroimaging follow-up, revealed progression from atypical solitary lesions to multifocal lesions in the white matter of the brain. Moreover, key immunological markers were observed in CSF during follow-up, suggesting the evolution of MS. This case demonstrates that TB is a potentially important but overlooked factor involved in the pathogenesis of MS. Upon initial detection, atypical lesions should receive sufficient attention and patients should undergo systematic screening to identify TB and its associated immunological abnormalities.
We followed the CARE guidelines when writing this report. 12
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231208570 - Supplemental material for Ophthalmoplegia with a focal lesion in the interpeduncular fossa as the initial symptoms of multiple sclerosis: 3-year follow-up
Supplemental material, sj-pdf-1-imr-10.1177_03000605231208570 for Ophthalmoplegia with a focal lesion in the interpeduncular fossa as the initial symptoms of multiple sclerosis: 3-year follow-up by Kai Han, Yixuan Wang, Shasha Li, Tianshu Ye, Miao Li and Jie Wang in Journal of International Medical Research
Footnotes
Acknowledgements
We thank Jie Wang and Miao Li for assistance with reviewing and revising this article.
Author contributions
KH and JW wrote the main manuscript. SL and TY prepared the imaging data. YW and ML revised the manuscript. All authors reviewed the manuscript.
Data availability statement
The original data obtained in this report are included in the article. Inquiries for additional information should be directed to the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
Because this is a case report, ethical approval was not applicable. The patient in this case provided written informed consent to participate and consented to publication of the case report (including the imaging results). We followed the CARE guidelines when writing this report.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
