Abstract
Objective
This study was performed to evaluate the effect of a homemade autotransfusion pressure-control system on the regulation of negative pressure and to clarify the influence of different negative pressures on the recovered erythrocytes.
Methods
Fifty patients were randomly divided into five groups, and five different suction-generated negative pressures were applied. Before suction, 6 mL of blood was collected from the surgical field; after suction, 6 mL of blood was collected from the blood storage tank. The hemoglobin, hematocrit, mean corpuscular volume, newly generated standardized plasma free hemoglobin, and change in the hemolysis rate of erythrocytes before and after suction were compared. Additionally, the erythrocyte morphology was observed.
Results
The hemoglobin and hematocrit were significantly different before and after suction in all five groups. As the suction pressure increased, gradual increases were noted in the number of abnormal erythrocytes in the field of view, the newly generated standardized plasma free hemoglobin, and the change in the hemolysis rate.
Conclusions
The destruction rate of erythrocytes increased as the suction-generated negative pressure increased. When using a pressure-control system, a negative pressure of <200 mmHg should be applied to reduce the damage to the autotransfused blood.
Introduction
Blood transfusion has become a necessary method to ensure the safety of patients with severe intraoperative blood loss. However, adverse reactions caused by allogeneic transfusion are not rare. 1 Therefore, in the current situation of limited blood resources, autotransfusion has been playing an increasingly prominent role in blood conservation strategies. 2 Autotransfusion allows the lost blood to be quickly and effectively reused, improving the success rate of first aid in the case of hemorrhage. This can reduce the burden of medical blood consumption and avoid various complications caused by allogeneic transfusion. 3
It should be noted that research has shown that the quality of recycled blood is affected by many factors, such as negative-pressure suction devices, filtration devices, and washing liquids used during the autotransfusion process. 4 Among these, negative-pressure suction is the most important factor in declining erythrocyte quality. 5 Our group found that most operating rooms adopt a central suction negative-pressure system and that the negative pressure is generally fixed at 0.040 to 0.055 MPa (300.0–412.5 mmHg); however, the current clinically recommended suction-generated negative pressure should not exceed 0.02 MPa (150 mmHg). 6 To reduce erythrocyte destruction, we developed a pressure-control pipe to regulate the suction-generated negative pressure during autotransfusion using a simple reconstructive device (Patent No. ZL 201410666009.6). This autologous blood recovery pressure-control system was used to accurately regulate the suction-generated negative pressure, clarify the impact of different negative pressures on red blood cells, and ensure the quality of recovered blood. In the present study, we developed an updated autotransfusion pressure-control system to precisely control the suction-generated negative pressure and investigated the influence of different negative pressures on erythrocytes using this new system.
Methods
Patients
This study was approved by the Medical Ethics Committee of Lishui People’s Hospital (23 January 2019, approval no. 2019-01), and written informed consent was provided by the patients and their families. The study involved 50 orthopedic and neurosurgical patients who were undergoing autotransfusion in Lishui People’s Hospital and were expected to bleed more than 400 mL. The patients were aged 48 to 78 years, and they had a preoperative American Society of Anesthesiologists physical status of I or II, preoperative hemoglobin (Hb) concentration of >100 to <150 g/L, hematocrit (HCT) >30%, and platelet count of >100 × 109/L. All patients had normal platelet function and coagulation indices, and no patients had severe visceral diseases (severe impairment of heart, brain, liver, or kidney function), infectious fever, bacteremia, or hematopoietic diseases. The random number table method was used to divide the patients into Groups A, B, C, D, and E, in which the suction-generated negative pressure was 100, 150, 200, 250, and 300 mmHg, respectively.
Experimental procedure
Anesthesia preparation
The patients were given oxygen supplementation after entering the operating room, and electrocardiography, oxygen saturation, blood pressure, and hemodynamic indices (arterial blood pressure and central venous pressure) were monitored. Local anesthesia was administered via inferior right internal jugular puncture and catheterization and radial artery puncture and catheterization. All patients received slow intravenous injections of 0.2 to 0.3 µg/kg sufentanil, 0.2 mg/kg cisatracurium, and 1 to 2 mg/kg propofol. After tracheal intubation, mechanical ventilation was initiated, and the partial pressure of end-tidal carbon dioxide was maintained at 30 to 40 mmHg. During the operation, anesthesia was maintained with sevoflurane at 1% to 2%, propofol at 100 to 200 mg/hour, remifentanil at 0.1 to 0.3 µg/kg/minute, dexmedetomidine at 0.02 to 0.05 µg/kg/minute, and a bispectral index of 40 to 60.
Pressure-control system
The pressure-control device was set between the suction pipe (connected to the central negative-pressure suction system) and the blood storage tank. This device was able to stably control the negative pressure in the blood storage tank externally (Figure 1).
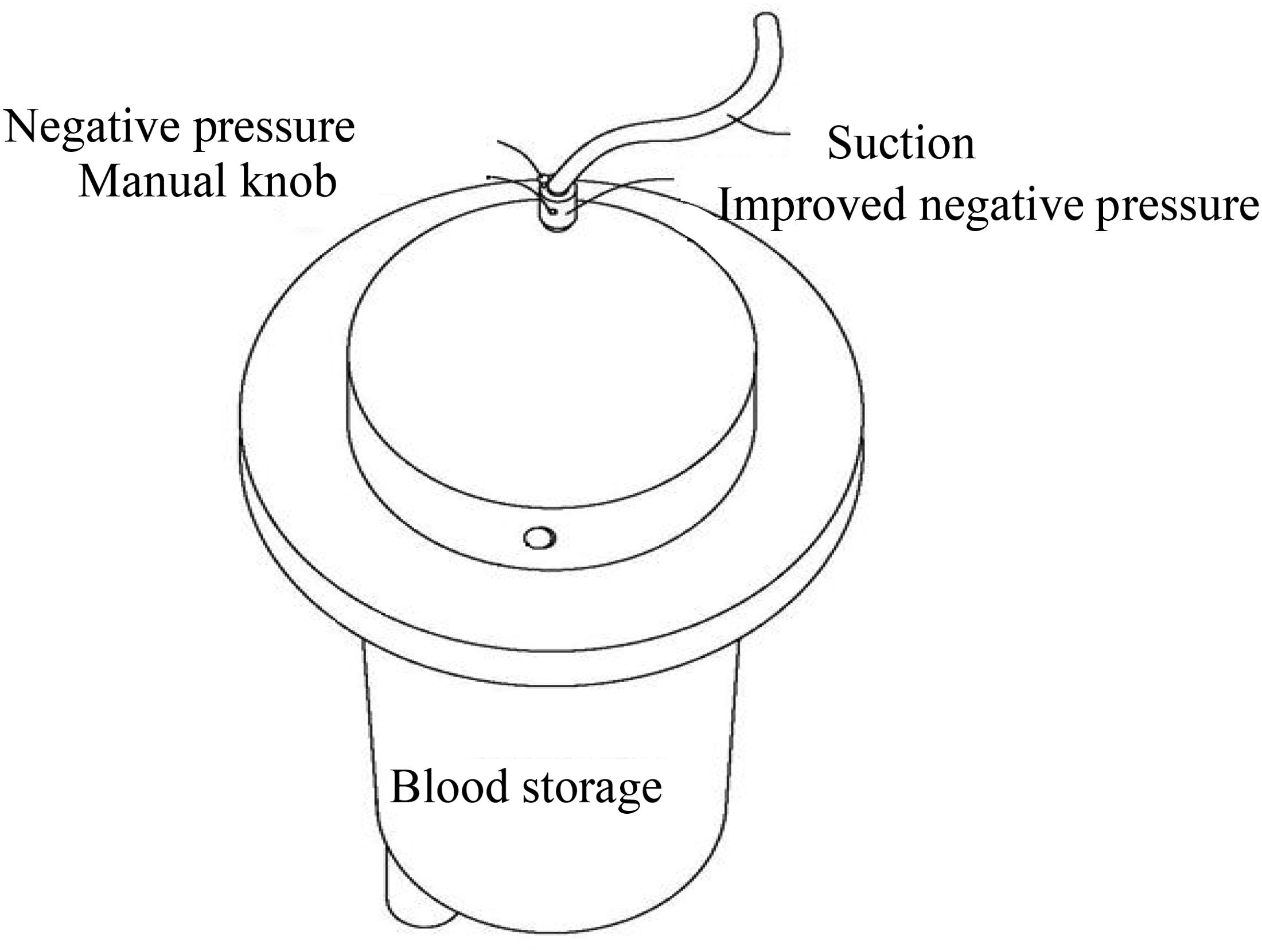
Schematic diagram of the connection structure of the external pressure control device.
This pressure-control device is characterized by a dynamic decompression system and pressure-measuring device added to the negative-pressure suction port of the blood storage tank. The knob is rotated according to the monitoring value of the negative-pressure gauge, causing the decompression membrane to cover the negative-pressure suction port and thus regulate the suction-generated negative pressure. The rest of the pipeline is installed according to the operating instructions (Beijing Jingjing Medical Company, Beijing, China). A schematic diagram of the principle of this autotransfusion dynamic pressure-control system is shown in Figure 2. This homemade negative-pressure suction system has been commercialized by Beijing Jingjing Medical Company (Patent No. ZL 201710839066.3).

Schematic diagram of the principle of the autotransfusion dynamic pressure-control system.
Autotransfusion process
Heparin (12,500 U) was diluted with 500 mL saline, and the recovery line was prefilled with 100 mL of the heparin diluent. During the recovery process, the heparin drip rate was adjusted according to the amount and rate of bleeding to maintain the ratio of blood recovery to heparin diluent at about 100:15 mL. The anesthetist used an improved external pressure-control system to manually and accurately adjust the suction-generated negative pressure at 100, 150, 200, 250, or 300 mmHg according to the study group.
Specimen collection
Within 1 hour of the operation, the assistant slowly drew 6 mL of blood from the surgical field. After suction, 6 mL of blood was drawn from the blood storage tank; the blood storage tank was gently shaken before the blood was drawn. The blood samples were placed in ethylenediaminetetraacetic acid anticoagulation tubes and processed within 2 hours. Erythrocyte morphology was observed with the Wright–Giemsa staining method. The Hb concentration, HCT, and mean corpuscular volume (MCV) of erythrocytes before and after the suction were measured. The newly generated standardized plasma free Hb (plfHb) was quantitatively determined using a Hemoglobin Colorimetric Assay Kit (Beijing Red-bio Company, Beijing, China). The rate of change in hemolysis (Δ hemolysis rate) was calculated by subtracting the hemolysis rate before suction from the hemolysis rate after suction. The hemolysis rate (%) was defined as the ratio of plfHb to whole blood total Hb.
Statistical analysis
Statistical analyses were performed using SPSS 20.0 software (IBM Corp., Armonk, NY, USA), and all measurement data were tested for normality using the Shapiro–Wilk test. Normally distributed data are expressed as mean ± standard deviation. One-way analysis of variance was used for comparison among groups, and a paired t-test was used for comparison within groups. The chi-square test was used for count data. Statistical significance was defined as P < 0.05, and strong statistical significance was defined as P < 0.01.
Results
Basic characteristics of patients in the five study groups
There were no statistically significant differences in sex, age, or American Society of Anesthesiologists physical status of the five groups of patients, as shown in Table 1.
General characteristics of the five groups of patients.

Data are presented as mean ± standard deviation or numbers of patients.
ASA, American Society of Anesthesiologists.
Comparison of erythrocyte morphology among the five groups
The erythrocyte morphology was observed in a blood smear under an optical microscope with an oil lens at 1000× magnification. Before negative-pressure suction, the erythrocytes in all groups exhibited a normal shape and uniform size; they were about 6 to 9 µm in diameter, and they showed a physiological central lightly stained area occupying about one-third of the cell diameter surrounded by a thick periphery (Figure 3).

Blood smears of the patients in the five groups processed by Wright–Giemsa staining method. The arrows indicate poikilocytes.
In Group A, stomatocytes were occasionally observed after negative-pressure suction, and the original central lightly stained area was fish mouth-shaped. Most of the erythrocytes in the visual field were the same as before suction and were uniform in size.
In Group B, a few teardrop-shaped erythrocytes and elliptocytes were observed in the visual field after suction, and most of the cells were disc-shaped.
In Group C, there were more stomatocytes in the visual field after suction, and occasional acanthocytes of uniform size were observed. Many normal disc-shaped erythrocytes were still present.
In Group D, erythrocytes of different sizes were irregularly gathered in the visual field after suction. Stomatocytes, serrated erythrocytes, and spherocytes were observed, and a few disc-shaped erythrocytes were still present.
In Group E, most of the cells in the visual field were abnormal poikilocytes after negative-pressure suction, and many were acanthocytes with many needles or protrusions on the surface of the cells. Elliptocytes were occasionally observed, and disc-shaped erythrocytes were rare.
Hb, HCT, and MCV of erythrocytes before and after suction
As shown in Table 2, there was no statistically significant difference in the Hb concentration, HCT, or MCV of erythrocytes among the five groups either before or after suction, and there was also no statistically significant difference in the MCV after suction. However, paired comparisons of the Hb concentration and HCT before and after suction within each group showed significant differences (P < 0.01).
Hb, HCT, and MCV before and after suction.

Data are presented as mean ± standard deviation.
Note: aComparison before and after suction is statistically significant (P < 0.01).
Hb, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume.
Comparison of newly generated standardized plfHb and Δ hemolysis rate
Under different negative pressures, the differences in the newly generated standardized plfHb and Δ hemolysis rate were statistically significant (P < 0.01). As the negative pressure increased by 50 mmHg, the plfHb and Δ hemolysis rate of each group increased accordingly. Both the plfHb and Δ hemolysis rate exhibited a strong exponential correlation with the negative pressure (correlation coefficient r2 > 0.996 for both) (Figure 4). The exponential correlation indicated that as the negative pressure increased, the plfHb and Δ hemolysis rate increased more rapidly. The average plfHb in Groups B, C, D, and E was approximately 3, 5, 18, and 41 times higher than that in Group A, respectively. The average Δ hemolysis rate in Groups B, C, D and E was approximately 3, 5, 15, and 33 times higher than that in Group A.

Line chart of (a) newly generated standardized plasma free hemoglobin and (b) hemolysis rate under different negative pressures. Note: Compared with Group A, aP < 0.05; compared with Group B, bP < 0.05; compared with Group C, cP < 0.05; compared with Group D, dP < 0.05; and compared with Group E, eP < 0.05.
Surgeon satisfaction
As shown in Table 3, the pairwise comparative analysis showed no significant difference between Groups A and B; showed a significant difference between Groups C, D, and E and Groups A and B (P < 0.05); and showed no significant difference between Groups C, D, and E.
Comparison of surgeon satisfaction
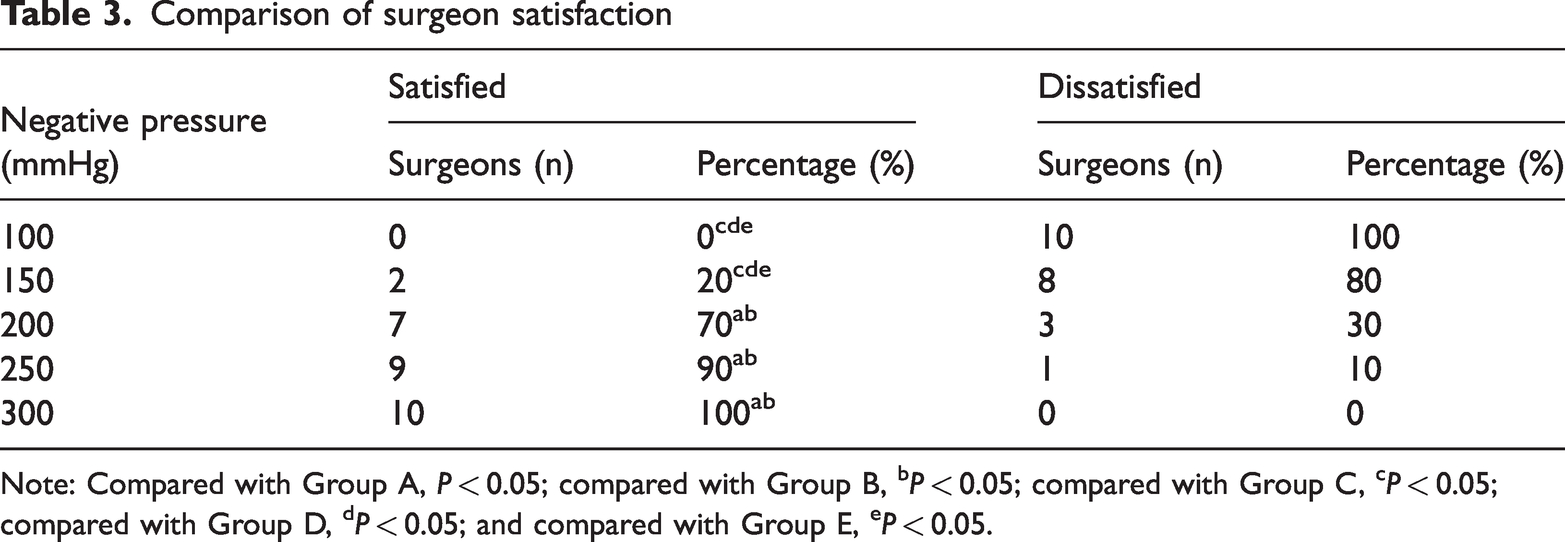
Note: Compared with Group A, P < 0.05; compared with Group B, bP < 0.05; compared with Group C, cP < 0.05; compared with Group D, dP < 0.05; and compared with Group E, eP < 0.05.
Discussion
Intraoperative cell-washing autotransfusion is the most commonly used autotransfusion technology in clinical practice and can effectively reduce the need for allogeneic transfusion. However, the quality of autotransfused blood is affected by many factors, among which negative-pressure suction is the most important factor in declining erythrocyte quality. During negative-pressure suction, erythrocytes rupture and dissolve under the combined effects of air–blood interaction and shear forces. 7 Waters et al. 8 showed that using appropriately low negative-pressure suction could reduce hemolysis in saline-diluted blood by 60%. Yazer et al. 9 demonstrated that the hemolysis rate could be effectively reduced by using a device that automatically adjusted the suction pressure according to the air pressure of the suction tip. Thus, precisely controlling the suction-generated negative pressure at a low value is a practical method to reduce the damage to recycled erythrocytes.
In our preliminary research, we found that erythrocytes were likely to be damaged when the suction-generated negative pressure of autotransfused blood was too high because of the use of autotransfusion negative pressure together with central suction negative pressure (300.0–412.5 mmHg). We developed a pressure-control pipe to regulate the negative pressure. However, some difficulties were encountered in the operation of this device. First, during the actual suction process, many factors (such as mixing of air, fluctuation in the central suction negative pressure, and others) led to pressure fluctuation in the outlet pipe (or piezometer). Therefore, the surgeon frequently had to manually adjust the opening of the decompression membrane to maintain a constant pressure in the outlet pipe (or piezometer), which was difficult to accomplish. Second, the pressure-control tube was in direct contact with the patient’s blood and was required to be sterile; thus, it could not be reused and was therefore wasteful. Third, during the operation, the surgeon required quick and efficient suction to keep the surgical field clean. To accomplish this, the anesthetist had to continue informing the surgeon of the negative-pressure gauge reading. This not only failed to effectively control the suction-generated negative pressure in a timely manner but also increased the workload of the surgeon and affected the operation process.
Therefore, to compensate for these disadvantages of the pressure-control pipe for autotransfusion, we made improvements to the device and developed a negative-pressure suction system for blood storage hemofilters. The improved pressure-control system has three major advantages. First, the overall control mechanism can adjust the degree of closure of the decompression membrane according to the incoming negative pressure (Figure 2). This is a mature computer-controlled process with the characteristics of real-time system monitoring and regulation of suction-generated negative pressure. Second, the negative-pressure meter is peripherally located for safety. The knob can be rotated manually when the negative-pressure gauge index is too high. The coexistence of two adjustment modes avoids the occurrence of manual control errors and ensures the conversion to manual control under special circumstances. Third, the operation is simple; the anesthetist can monitor the negative pressure alone without the intervention of the surgeon, and the operation process is not affected.
Normal mature erythrocytes, which exhibit a biconcave disc shape, can maintain their unique shape when passing through narrow blood vessels. This ability is mainly attributed to the characteristics of the cell membrane. 10 In this study, most of the erythrocytes that were recovered in Groups A and B maintained their biconcave disc shape; their morphology was not significantly different from that before suction. However, poikilocytes (acanthocytes, mouth-shaped cells, teardrop-shaped cells, spherocytes, and others) of various sizes began to appear in the blood that was recovered in Groups C, D, and E. Furthermore, as the negative pressure increased, the numbers of all types of poikilocytes gradually increased; this was especially true in the blood recovered in Group E, which was characterized by many acanthocytes. The changes in the morphology of these erythrocytes indicate that the cell membrane is damaged during the process of negative-pressure suction and its stability is reduced. These poikilocytes easily rupture and readily dissolve under the action of high-speed centrifugal force during washing, resulting in a decreased recovery rate.
After an erythrocyte ruptures, the Hb is freed and enters the plasma, becoming plfHb. 11 Therefore, the amount of newly generated plfHb can reflect the extent of erythrocyte destruction. Figure 2 shows that as the suction-generated negative pressure increased, the newly generated standardized plfHb gradually increased, and an exponential correlation was observed between the negative pressure and the newly generated standardized plfHb. This means the extent of erythrocyte rupture dramatically rises as the negative pressure increases. For every 50-mmHg increase in suction-generated negative pressure, the plfHb of Groups B, C, D, and E increased by 2, 2, 13, and 23 times the amount of plfHb in Group A. The Δ hemolysis rate calculated from the concentration of plfHb intuitively expressed the quality of the autotransfused blood, and its variation with suction-generated negative pressure was basically the same as that of plfHb. With the increase in suction-generated negative pressure, the Δ hemolysis rates of the five groups were 0.038%, 0.102%, 0.180%, 0.567%, and 1.258%, respectively. When the suction-generated negative pressure was <200 mmHg, the Δ hemolysis rate did not substantially change, and the erythrocytes were less severely damaged. With excessive suction-generated negative pressure (≥250 mmHg), the efficiency of blood suction improved; however, the mechanical damage to erythrocytes also increased. Moreover, both the blood flow velocity and shear stress markedly increased, leading to a significant increase in plfHb in both Groups D and E. Patients who undergo excessive plfHb blood transfusion have been shown to be prone to postoperative macroscopic hemoglobinuria, which is associated with the incidence of renal failure. 12
Research has also confirmed that excessive shear stress on erythrocytes causes increased release of adenosine triphosphate, leading an insufficient supply of adenosine triphosphate for erythrocytes and directly affecting the sodium pump exchange on the erythrocyte membrane. This results in decreased intracellular potassium and increased intracellular sodium followed by swelling of the cells to a spherical shape, reduced deformability, and even direct dissolution and fragmentation. 13 The surface area of mature red blood cells is generally 135 µm2 and the volume is 84 to 94 µm3; therefore, the ratio of area to volume is large, which is conducive to the exchange of oxygen and carbon dioxide throughout the body. In the present study, the MCV of erythrocytes after negative-pressure suction was within the normal range in all five groups, and there were no significant differences either among or within the groups before and after suction. Unexpectedly, the erythrocyte volume did not increase or decrease after suction. Two possible reasons for this were considered. First, the sample size was insufficient for this clinical trial; second, considering that the human body is a complex environment, there were many uncontrollable factors. Therefore, the sample size should be increased in future studies, and the selection criteria should be strictly controlled.
The results of this study show that our self-developed autologous blood transfusion pressure-control system can accurately control the required suction-generated negative pressure through a simple operation with low cost and high clinical practicality. As the suction-generated negative pressure increases, the destruction of red blood cells gradually increases. Considering both the suction speed and surgeon’s satisfaction, the use of a negative pressure of ≤200 mmHg can reduce cell damage during autologous blood recovery.
Footnotes
Acknowledgements
We thank our colleagues in the departments of anesthesiology, orthopedics, and neurosurgical surgery for their cooperation in facilitating this trial.
Authors’ contributions
Jimin Wu, Yini Wu, and Jiaqun He designed the study. Xin Han, Qiaomin Xu, and Hong Dai recruited the patients. Yini Wu and Jiaqun He performed the statistical analysis and wrote the manuscript. Jimin Wu revised the manuscript. All authors are aware of and responsible for the research data. All authors read and approved the manuscript in its final version.
Data sharing statement
All data generated or analyzed during this study were included in the published article. Further inquiries about the datasets can be directed to the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This article was funded by the Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (grant number 2019PY092) and the Lishui Science and Technology Plan Project (grant number 2023GYX27).
