Abstract
Laser refractive surgery (LRS) is a specialized surgical discipline within ophthalmology that focuses on vision correction via laser techniques. LRS requires a high rate of accuracy and exactitude to improve the visual outcome and minimize complications, which may lead to delayed visual recovery. Keratitis, either infectious or noninfectious, is a post-LRS complication that requires early diagnosis and proper interventional measures. In this narrative review, we summarize different aspects of keratitis following LRS. This literature review aims to provide a thorough understanding of the causes of post-LRS infectious keratitis and its appropriate management for successful outcomes.
Keywords
Introduction
Laser refractive surgery (LRS) is a surgical subspecialty within ophthalmology that uses photorefractive laser technology to improve visual outcomes, and it requires a high level of precision to achieve this goal while minimizing ocular complications. 1 Laser-assisted in situ keratomileusis (LASIK), photorefractive keratectomy (PRK), and small-incision lenticule extraction (SMILE) are common keratorefractive surgical interventions that are thought to provide reliable and predictable outcomes. Despite the application of modern technologies and meticulous patient selection, complications occasionally occur, leading to unsatisfactory visual acuity.2–5 Although keratitis is an uncommon complication following LRS, it may have catastrophic outcomes. Early diagnosis of keratitis is necessary to prevent extensive corneal destruction and preserve visual acuity in affected patients.4,6 Compared with other forms of microbial keratitis such as contact lens-associated keratitis, post-LRS keratitis is characterized by a distinct spectrum of pathogens and a particular management approach. Typically, a broad-spectrum topical antibiotic such as a fluoroquinolone is administered postoperatively. However, such antibiotics are not effective against fungi, Nocardia, Acanthamoeba, and some atypical mycobacteria. 7 Furthermore, flap lifting and amputation may be needed in some patients with post-LASIK keratitis. In this narrative review, we provide an overview of the epidemiology, risk factors, management principles, and prognosis associated with keratitis following LRS in an effort to improve its understanding and management.
Epidemiology
Post-LRS keratitis is a rare complication that can be divided into infectious and noninfectious keratitis. The occurrence of noninfectious keratitis (2.34%) is reportedly 7.5 times higher than that of infectious keratitis (0.31%). 8 The statistics regarding the rate of keratitis after refractive surgery seem to possess some degree of inaccuracy because affected patients may present to another physician/center instead of the primary physician/center.9–11 Both infectious keratitis and noninfectious keratitis (sterile infiltrations) have been reported to manifest within a few days after the procedure, with a prevalence of 0.0013% 8 after all types of LRS.
Table 1 shows the incidence rate of infectious keratitis following LASIK and PRK in different studies. A 30-year model showed that the risk of microbial keratitis-related vision loss following LASIK was lower than that following 15 years of using extended-wear contact lenses and daily-wear soft contact lenses. However, rigid gas-permeable lenses showed a safer profile than LASIK. 12 The occurrence of infectious keratitis during the first week after PRK is higher than that after LASIK because of the presence of corneal epithelial defects and the utilization of soft contact lenses. An article published in 2022 reported that the incidence of infectious keratitis following PRK increased by nine-fold after the COVID-19 pandemic. 13 Although it has been suggested that the occurrence of infectious keratitis following SMILE is extremely low, this may have been underestimated.14–16 Incidence rates of 0.003% and 0.004% have been reported for post-SMILE keratitis in different studies.17,18
Incidence of infectious keratitis after LASIK and PRK.

LASIK, laser-assisted in situ keratomileusis; PRK, photorefractive keratectomy.
Diffuse lamellar keratitis (DLK) and staphylococcal marginal hypersensitivity (SMH) are the two main types of noninfectious keratitis, with DLK being the most frequent. 19 The incidence of DLK as an inflammatory response after photorefractive procedures is estimated to range from 0.40% to 4.38%. Studies have indicated that DLK occurs more frequently after femtosecond LASIK than after microkeratome LASIK (0.4%–37.5% vs. 0.1%–12.1%), possibly because of the higher amount of laser energy delivered in the former procedure.20,21 Additionally, DLK is more often observed following LASIK than SMILE; however, it has a distinctive appearance following SMILE with a frequency of 0.20% to 0.45%. 21 The occurrence of SMH as an immune response to ocular microflora is more common following PRK than LASIK. 19 The occurrence of SMH following SMILE is infrequent and benign, and it probably develops secondary to a type 1 hypersensitivity immune reaction. 22
Microbiology
Pathogens responsible for infectious keratitis include bacterial species (staphylococci, streptococci, mycobacteria, Pseudomonas, and Nocardia), fungal species (Aspergillus flavus, Aspergillus fumigatus, Candida, Alternaria, Fusarium, Acremonium, and Curvularia), viruses (herpes simplex virus (HSV), Adenovirus), and amoebae (Acanthamoeba).7,23 The prevalence of mycotic keratitis is lower than that of other infectious etiologies. 11 Post-LASIK Gram-negative keratitis is less common than contact lens-related cases of keratitis. 9
Gram-positive bacteria and atypical mycobacteria are the most common causative organisms of post-LASIK microbial keratitis. It has been proposed that mycobacterial keratitis is related to contamination of steamers used to clean microkeratomes. During recent years, femtosecond laser technology has gained significant attention in lamellar flap creation, leading to a decreasing trend in mycobacterial infections. 24 Better adherence to sterility protocols and prescription of fourth-generation fluoroquinolones are other contributing factors in this regard.
Gram-positive organisms, especially coagulase-negative staphylococci, Staphylococcus aureus, and Streptococcus species, are the most common pathogens that cause complications following surface ablation procedures. 25 Working in healthcare facilities appears to increase the risk of methicillin-resistant Staphylococcus aureus (MRSA) keratitis. 26 In contrast to LASIK, surface laser ablation is rarely associated with postoperative atypical mycobacteria infection. 23 Previous reports have described outbreaks of Nocardia keratitis following LASIK and PRK due to usage of the same blade/microkeratome and breakage in operating room sterility, respectively.27,28
Predisposing factors
Contact lens usage or manipulation, xerophthalmia, meibomitis/blepharitis, dacryocystitis, and an immunocompromised status are the major risk factors for infectious keratitis; thus, a comprehensive preoperative ophthalmic examination is critical. 16 Inappropriate postoperative prescription of eye drops containing corticosteroids can also accelerate the spread of infection. 29 A history of a corneal foreign body can be a predisposing factor for keratitis. 15 Trauma from vegetable materials such as soil, tree branches, or wind-borne foreign particles increases the risk of mycotic keratitis. 30 Furthermore, conditions that may lead to poor healing such as neutropenia should be noted prior to surgery. 24 The SPEED Questionnaire for ocular surface disease can assist ophthalmologists in recognizing these issues preoperatively. 24 Notably, however, predisposing factors are not always detectable in all patients presenting with post-LRS keratitis. 15
Adherence to hygiene regulations and sterilization procedures can significantly reduce the likelihood of post-LRS infectious keratitis. 31 Proper case selection, application of antiviral medications for patients with a history of HSV keratitis, prophylactic antibiotic administration, and perioperative irrigation of the eyelashes, lid margins, and conjunctiva can significantly reduce the incidence of post-LRS keratitis. 24
General assessments
The presence of pain, photophobia, discomfort, discharge, reduced vision, and signs of corneal infiltration and opacification should raise clinicians’ suspicion for post-LRS keratitis. In these patients, corneal scrapings should be performed for smear examination and culture. In post-LASIK patients (excluding those with an epithelial defect and accessible infiltration site), the flap should be lifted to allow access to the infiltration site. Culture media can be inoculated directly, including blood agar, chocolate agar, Sabouraud agar, thioglycolate broth, Lowenstein–Jensen medium for atypical mycobacteria, and non-nutrient agar with Escherichia coli overlay for Acanthamoeba. Polymerase chain reaction can be used for suspected viral cases as well as Acanthamoeba, fungi, culture-negative bacterial cases, and atypical mycobacteria. 9 Corneal biopsy should be considered for cases that are refractory to medical therapy and have a negative culture. Flap amputation can provide both diagnostic and therapeutic benefits in LASIK cases. 32 Confocal microscopy, another diagnostic tool especially for fungal, Acanthamoeba, Candida, and Nocardia species, shows remarkable rapidity and sensitivity, although it is observer-dependent. 33
Infectious keratitis
Post-LASIK
Post-LASIK infectious keratitis may have an early or delayed onset. The early-onset type occurs within the first 2 weeks after the procedure and is mostly caused by MRSA and streptococci. Notably, MRSA cases are more likely to be seen in patients with a history of exposure to healthcare environments. The late-onset type occurs from the third week to third month after the procedure and is commonly caused by fungi and atypical mycobacteria. 24 However, a very late-onset Acanthamoeba keratitis (1 year after LASIK) was reported by Kaur et al. 34 Approximately 47% of post-LASIK cases are caused by mycobacteria (mostly Mycobacterium chelonae and Mycobacterium fortuitum). 26 Reports suggest that most cases of post-LASIK mycobacterial keratitis are caused by inadequate sterilization of operating rooms and equipment and reutilization of disposable microkeratome blades. Nocardia, which is an uncommon microorganism, remains endemic in Southeast Asia among patients who have undergone LASIK.26,35
Post-PRK
Comparative studies have estimated that the risk of developing infectious keratitis after PRK is 2.92 to 6.00 times higher than that after LASIK. 24 Clinicians must maintain a high degree of suspicion when managing patients with keratitis following PRK to ensure an early diagnosis and adequate management, which will likely lead to a better visual outcome and final prognosis.
Post-SMILE
SMILE is a flapless keratorefractive procedure that was approved by the US Food and Drug Administration in 2016; thus, there are few available documented reports of its complications. 14 Most of the reported cases of infectious keratitis following SMILE were unilateral; nevertheless, pneumococci and non-tuberculous mycobacteria might lead to bilateral keratitis.36,37 Infectious keratitis is typically expected to present within the first week of SMILE; however, atypical mycobacterial and fungal keratitis tend to manifest with delayed presentations.29,37,38
Non-infectious keratitis
Diffuse lamellar keratitis
DLK is a sterile inflammatory keratitis that is usually associated with LASIK, but it can also develop following SMILE. Its frequency varies according to the type of surgery, different operation settings, and patients’ clinical status.22,39–41 DLK usually occurs on the first postoperative day; however, it may rarely manifest 2 to 3 days after LASIK or almost 5 days after SMILE.22,39 The precise cause of DLK remains unclear; however, several potential risk factors have been identified, including glove talc, marker pens, high-power laser exposure, intraoperative epithelial damage, chemical toxins or bacterial endotoxins on surgical equipment, and meibomian gland secretions.42,43 In their case report, Dan et al. 22 concluded that IgA nephropathy might be a risk factor for delayed DLK following SMILE; therefore, a precise history along with a complete urinalysis and kidney function tests would be helpful before SMILE. In slit lamp biomicroscopy, DLK is classically described as “sands of Sahara” because of the appearance of wavy white lines of granular inflammatory cells. Upon recognizing diffuse, multifocal, and sand-like granular infiltration, ophthalmologists should consider potential microbial infections of the cornea and interfacial inclusions as differential diagnoses of DLK. 39
SMH
SMH is a self-limiting benign enhanced immune response in immunocompetent patients secondary to staphylococcal blepharoconjunctivitis, meibomitis, or rosacea. 44 It mainly occurs following PRK and less frequently following LASIK. The manifestations often appear 1 to 4 days postoperatively. The general pathophysiology of SMH is based on a type III hypersensitivity reaction.24,44 SMH following SMILE is a rare condition that seems to develop under specific clinical circumstances.44,45 Hypopyon, though infrequent, may manifest in severe cases of SMH, complicating the diagnostic process. Therefore, endophthalmitis should be ruled out as a differential diagnosis. 46 Differentiation from infectious keratitis is possible by focusing on anterior stromal involvement and lack of focal inflammatory lesions. 24
Management
LASIK
According to different reviews, the recommended management of post-LASIK keratitis follows a systematic approach consisting of elevation of the flap, corneal scraping coupled with culture, and irrigation with a tailored antibiotic solution. Discontinuation of corticosteroids is essential and should be accompanied by the administration of a mixture of local antibiotics. Early-onset cases should be treated with a combination of topical vancomycin 5% and either topical gatifloxacin or moxifloxacin (fourth-generation fluoroquinolone) every 30 minutes. For delayed-onset cases, a combination of local vancomycin 5% with amikacin or local clarithromycin with a fluoroquinolone (fourth-generation) are two viable options. However, the antibiotic regimen should be altered based on the results of culture isolation or antibiogram sensitivity results. Theoretically, oral doxycycline has the ability to decrease corneal melt. 24 In severe cases, amputation of the flap is necessary to achieve both diagnostic and therapeutic objectives. 47
Atypical mycobacterial keratitis should be seriously considered if the patient is not responsive to routine post-LASIK keratitis treatment. Late-onset keratitis in conjunction with crystalline accumulation is another indicator of mycobacterial keratitis. In cases of suspected mycobacterial keratitis, immediate treatment with local amikacin, clarithromycin, and fluoroquinolone should be initiated. 48 Topical linezolid is reportedly effective for multidrug-resistant mycobacterial keratitis following LASIK (Mycobacterium abscessus). DLK, HSV, Nocardia, Acanthamoeba, and infectious crystalline keratopathy are among the other differential diagnoses in patients with lack of response to routine treatment.26,35,47
Fungal keratitis after LASIK is very rare, and most cases are caused by Aspergillus, Candida, and Fusarium (Figure 1). A very-late-onset case of acremonium mycotic keratitis following LASIK has been reported; it was diagnosed by metagenomic deep sequencing, and the origin was confirmed to be livestock hay. 49 Hence, the authors recommended metagenomic deep sequencing as a novel method to improve the corneal culture sensitivity, especially in mycotic etiologies. 49 Immediate flap amputation and hourly topical antifungal medication such as natamycin, amphotericin B, or voriconazole are recommended. 49 Generally, management of interface mycotic keratitis is highly challenging with potentially poor outcomes related to poor drug bioavailability and sequestration of the involved microorganisms.50,51

A case of post-LASIK fungal keratitis.
PRK
Bacterial culture of contact lenses and scraping of the cornea should be performed in cases of probable corneal infection following PRK (Figure 2). For mild cases, topical eye drops containing fluoroquinolones should be administered immediately. In severe cases, topical tobramycin 1.5% or cefazolin 5% should be administered every 30 minutes. Additionally, topical corticosteroids should be discontinued. 52
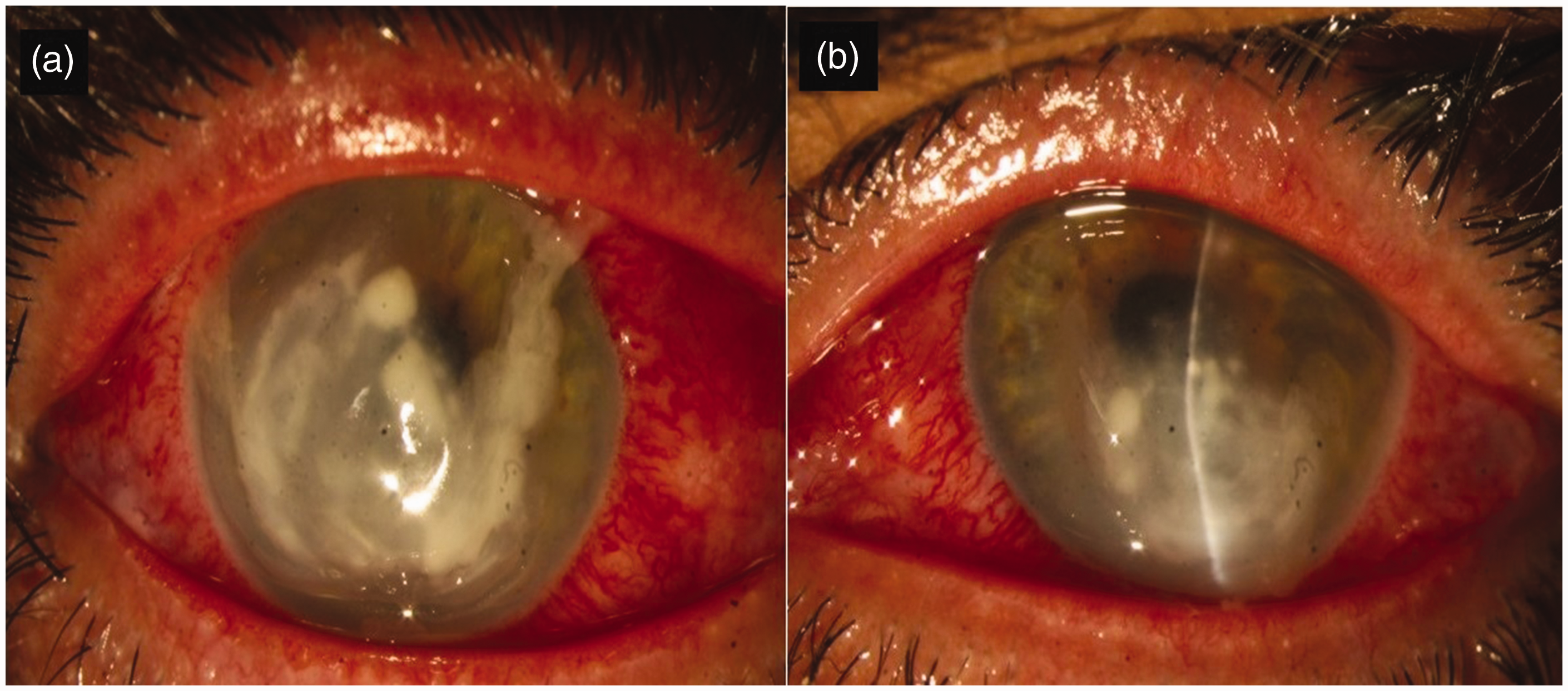
Bilateral staphylococcal keratitis following PRK. (a) Right eye. (b) Left eye. PRK, photorefractive keratectomy.
The following prophylactic antiviral regimen was recently recommended in patients with a positive history of herpetic keratitis without several recurrences: 400 mg oral acyclovir every 12 hours or 500 mg oral valacyclovir daily from 2 weeks preoperatively to at least 2 weeks postoperatively or while on topical steroids. The patient should have not experienced herpetic keratitis during the last year. This guideline has also been generalized to candidates for LASIK and SMILE. 53
Post-PRK fungal keratitis is a rare and clinically challenging diagnosis (Figure 3). Immediate initiation of proper medication is important to avoid severe complications and save the patient’s vision. Various antifungal medications are available, including natamycin, amphotericin B, imidazole, econazole, ketoconazole, fluconazole, miconazole, and terbinafine, and the most appropriate medication may be selected based on the type of fungus identified.7,54 Corneal collagen crosslinking and surgical corneal debridement are two other possible treatments. In cases that are particularly resistant to treatment, surgical resection of corneal lesions may be necessary. 54

Bilateral fungal keratitis following PRK. (a) Right eye. (b) Left eye. PRK, photorefractive keratectomy.
SMILE
Post-SMILE infectious keratitis can threaten vision if not diagnosed early and appropriately managed. 55 Treatment of post-SMILE keratitis is more challenging than treatment of post-LASIK keratitis because of the closed intrastromal junction, which can act as a nest for sequestration of pathogens.29,37 Furthermore, access to this area is highly restricted. 38 Li et al. 55 recently published a step-by-step protocol for managing bacterial keratitis. They recommend topical antibiotics for mild, limited cases of infection. In cases of lesion shrinkage, topical medication should be continued. In cases of lesion expansion, the corneal cap should be converted to a flap, and necrotic tissue should be scraped off for smear and culture followed by irrigation of the stromal bed and flap with antibiotic solutions. In cases of cap involvement, a complete intrastromal interface washout should be performed after scraping the necrotic tissue off. For multiple lesions or drastic infections, all the above-mentioned steps should be performed as soon as possible, and both systemic and topical antibiotics should be administered. As the last step, application of local corticosteroids is recommended to minimize scar formation. 55
Fungal organisms exhibit higher virulence than bacteria and are capable of penetrating the inner layers of the corneal tissue, resulting in more severe damage than bacterial keratitis. This can potentially lead to corneal perforation.36,38 Moreover, the need for interventional procedures is highly probable because of resistance to standard medications.29,47 In patients with a perforated cornea following fungal keratitis, which is more likely to occur than after bacterial keratitis, penetrating keratoplasty would be a lasting and durable intervention. 39
A case of a post-SMILE unilateral HSV keratitis was reported in 2016; it was indistinguishable from DLK and successfully managed by subconjunctival injections of dexamethasone and valacyclovir for 2 weeks. 56 Another case of unilateral HSV keratitis following SMILE was published in 2021; the authors recommended initiating empiric medication before collecting smears and cultivating samples because of the atypical presentation of herpetic keratitis, the similarity of signs between bacterial and viral keratitis, and the possibility of coincident mixed bacterial and viral keratitis. 57
DLK
Depending on the degree of inflammation and the patient’s clinical presentation, DLK is divided into four stages ranging from a mild peripheral infiltration of white granular cells without visual impairment (stage 1) to severe scarring, corneal melting, and visual impairment (stage 4). 58 Management is based on intensive topical corticosteroid and antibiotic therapy. Fluoroquinolones plus dexamethasone 0.2%, prednisolone acetate 1%, or rimexolone 1% are the main treatment choices in mild peripheral cases; however, flap lifting and interface washout with a mixed solution of saline, a steroid, and an antibiotic should be immediately performed in severely advanced cases. Notably, some data have shown that interface irrigation can increase the probability of corneal melting and focal infiltrations. 59 Furthermore, oral high-dose corticosteroids can reportedly improve the final outcome without flap raising and interface washing. 60 Hence, postponing the washout procedure for 1 month after steroid administration has been recommended. 22 Oral doxycycline can speed up wound healing by decreasing inflammatory processes (e.g., reducing macrophage recruitment and inhibiting the expression of endothelial growth factor C, interleukin-1b, nitric oxide synthetase, and tumor necrosis factor-α). 60 The occurrence of keratomycosis as a consequence of DLK management by intensive antibiotic and corticosteroid therapy should be kept in mind at follow-up visits.61,62 The same medical principles have been recommended for both late- and early-onset DLK following SMILE, and severe late-onset cases may respond to topical corticosteroids without flap lifting and interface washout. 22 However, Rana et al. 63 reported that severe delayed-onset DLK following LASIK should be treated by flap raising and irrigation in patients with a poor response to high-dose topical corticosteroids.
SMH
Although the disease course of SMH is self-limiting, administration of local corticosteroids and antibiotics can lead to quick resolution and relief.44,46 Use of a topical steroid/antibiotic mix can also enhance patient compliance. Moreover, a maintenance nightly prescription of an eyelid-cleaning routine with an antibiotic effective against Staphylococcus decreases the probability of SMH recurrence. Patients with refractory SMH should be treated with oral tetracyclines and topical fourth-generation macrolides. 64
Outcome
Infectious keratitis usually has worse visual consequences than noninfectious keratitis. Atypical organisms that are associated with a limited therapeutic response usually result in a poor prognosis and undesirable visual outcome.24,25 Noninfectious keratitis following PRK generally has a favorable prognosis with effective corticosteroid therapy, and most patients do not require interventional procedures. Approximately 50% to 75% of patients with post-LASIK infectious keratitis gain a best corrected visual acuity (BCVA) of 20/40 or better. More than 50% and 90% of patients with post-PRK infectious keratitis achieve a BCVA of 20/20 and 20/40 or better, respectively. In a large study of 1500 patients who underwent SMILE, 5 developed infectious keratitis; none experienced loss of BCVA, and all 5 had a final vision of 20/20. 43 However, there is a significant lack of data about post-SMILE cases.
Most cases of DLK resolve with complete restoration of vision after treatment. 11 Among patients with post-LASIK DLK, no lasting visual impact is expected if early diagnosis and proper management are achieved. 64 Furthermore, the incidence of DLK secondary to LASIK has been decreased because of the availability of low-energy femtosecond lasers. 25 The visual outcome of patients who develop SMH is often acceptable because of the frequent peripheral scars and eventual good visual acuity. 24 Progressive keratolysis, known as corneal melt, is a rare but serious complication of keratorefractive surgeries and negatively influences patients’ final visual outcome. Corneal melt is more frequent following LASIK than following other types of LRS. The active type of keratolysis is associated with underlying immunological or systemic diseases, DLK, and infectious or ulcerative keratitis. Treatment is based on prevention and management of the underlying causes. The most common type of infectious keratitis associated with corneal melt is HSV keratitis; however, a history of HSV is not an absolute contraindication for keratorefractive surgeries.65,66 Various factors can reportedly affect the final visual outcome of keratorefractive procedures and increase the need for retreatment. Older age, male sex, a high degree of mixed astigmatism, hyperopia, and a low temperature in the operating room are some predisposing factors.67,68
Conclusion
Although efforts have been made to decrease the incidence of post-LRS infectious and noninfectious keratitis, postoperative keratitis has not been eradicated. Early diagnosis and immediate interventional measures would absolutely improve the ultimate outcome. The early phase of each type of keratitis must be identified and differentiated by precise physical examination.
Footnotes
Acknowledgement
We would like to express special thanks to Soraya Banz (University of Edinburgh, UK) for his efforts in assisting with the preparation of this report.
Author contributions
Mohammad Soleimani and ARD conceived the idea and supervised the study. MA and SAT performed the literature search. SSS and KC wrote the first draft. KC, Mohammad Sargolzaeimoghaddam, and Maral Sargolzaeimoghaddam performed the editing. All authors created and approved the revised manuscript. SSS and KC should be considered joint first authors.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Ethics
The requirement for ethics approval was waived because of the nature of the study (review article).
Funding
This research received no specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
