Abstract
Objective
We aimed to investigate the frequency of dysnatremia among patients admitted with COVID-19 infection and its association with inpatient mortality.
Methods
This retrospective longitudinal study was conducted for 12 weeks. Serum sodium levels were recorded at admission, during the hospital stay, and within 48 hours of discharge or death. Logistic regression was used to determine the predictors of mortality.
Results
This study included 574 patients (69.7% men, age 55.6 ± 14.4 years). On admission, mean sodium was 135.9 ± 6.4 mEq/L; 39% had hyponatremia and 4.7% had hypernatremia. During admission, hypernatremia increased to 18.8%; maximum sodium in patients who survived was 140.6 ± 5.0 mEq/L versus 151.0 ± 9.9 mEq/L in those who died. The final sodium was 145.4 ± 9.4 mEq/L in patients who died versus 137.7 ± 3.7 mEq/L in those who survived (odds ratio [OR]: 1.22, 95% confidence interval [CI]: 1.13–1.32). Other predictors of mortality included ischemic heart disease (OR: 3.65, 95% CI: 1.39–9.61), acute kidney injury (OR: 6.07, 95% CI: 2.39–15.42), invasive ventilation (OR: 28.4, 95% CI: 11.14–72.40), and length of stay (OR: 0.91, 95% CI: 0.86–0.97).
Conclusion
Hypernatremia was frequently observed in patients who were critically ill and died and may be considered a predictor of mortality in COVID-19 infection.
Introduction
Numerous studies have shown that both hyponatremia and hypernatremia (defined as serum sodium <135 mEq/L and >145 mEq/L, respectively) are independently associated with mortality. 1 Hyponatremia is frequently observed in patients admitted with community-acquired pneumonia and is associated with both lengthened hospital stay as well as poor survival. 2 In patients infected with COVID-19, severe life-threatening pneumonia can develop, and mortality in these patients can be as high as 20.3% to 27.9%. 3
Whereas respiratory tract involvement is one of the main manifestations of COVID-19 infection, many patients also have associated dysnatremia (hyponatremia or hypernatremia). The reported prevalence of hyponatremia and hypernatremia in patients with COVID-19 ranges from 25% to 45% and 3% to 7%, respectively.4–6 Recently, it has also been observed that patients with COVID-19 infection can develop severe treatment-resistant hypernatremia exceeding >150 mEq/L, which is difficult to correct. 7
A recent review reported that hyponatremia is highly prevalent in patients who are hospitalized with COVID-19 infection and is associated with severe disease, intensive care unit admission, invasive ventilation, and mortality. 8 Another prospective observational study found that both hyponatremia and hypernatremia are associated with longer hospitalization and mechanical ventilation. 5 Recent observational studies have also reported that hyponatremia is frequently seen in patients with COVID-19 infection upon hospital admission,9–11 and both hyponatremia and hypernatremia are linked to poor outcomes. However, longitudinal data are lacking, and additional research is needed to clarify the association of dysnatremia with mortality. Therefore, the primary objective of our study was to determine the frequency of hyponatremia and hypernatremia at different stages of hospital admission in patients with COVID-19 infection.
Methods
This retrospective longitudinal study was conducted at Aga Khan University Hospital in Karachi, Pakistan for 12 weeks during 2020. The current work was conducted according to the principles laid down in the Declaration of Helsinki and was approved by the Ethical Review Committee of Aga Khan University Hospital, Karachi, Pakistan (protocol no. 2021-5803-16608 2021/02/04). All patient details were de-identified to protect patient privacy. Owing to the retrospective nature of this study, consent was not required. The current study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 12 A total of 626 patients were enrolled, among which 52 patients were excluded because they were admitted for reasons other than COVID-19 infection or were asymptomatic despite being positive for COVID-19 infection. Patients discharged home from the emergency room were also excluded. A specialized COVID-19 unit was established within our hospital for COVID-19-positive patients. All patients referred from the emergency department or the outpatient clinic were admitted to this designated COVID-19 unit.
In the study, patients’ serum sodium levels were recorded at three stages: 1) at admission, 2) at the maximum level observed during the hospital stay, and 3) within 48 hours of discharge or death. Serum sodium levels were categorized as follows:13,14 1) hyponatremia with serum sodium <135 mEq/L; 2) hypernatremia with serum sodium >145 mEq/L; and 3) normonatremia with serum sodium within the normal range (135–145 mEq/L). Hypernatremia was further categorized into three categories: serum sodium 146–154 mEq/L, 155–164 mEq/L, and >164 mEq/L.
Data were collected from patient discharge summaries, electronic records, and health records. A detailed patient history was obtained, focusing on comorbid conditions, the severity of COVID-19 infection, and medications (steroids, remdesivir, tocilizumab, and furosemide) given during the hospital stay. Information regarding the need for invasive or non-invasive ventilation was also recorded, along with renal replacement therapy, if required.
Serum creatinine levels at the time of admission and the maximum level during the hospital stay were recorded. Acute kidney injury was defined according to Kidney Disease Improving Global Outcome 2012 criteria. 15
The severity of COVID-19 infection was classified as non-severe, severe, or critical. 16 Non-severe COVID-19 included patients with mild to moderate disease. Mild disease referred to patients with diverse signs and symptoms but without breathlessness or abnormal chest imaging. Moderate disease referred to evidence of lower respiratory tract involvement with oxygen saturation (SpO2) ≥94%. Severe COVID-19 was defined as the presence of SpO2 ≤94% on room air, respiratory rate >30 breaths/minute, and >50% involvement with lung infiltrates on chest imaging. Critical disease referred to the presence of acute respiratory distress syndrome, aggravated inflammatory response, presence of septic or cardiogenic shock, thrombotic disease, and aggravation of co-existing comorbid conditions.
The main outcome measure in this study was inpatient mortality. The outcome for each patient was recorded as survived and discharged home or died.
The data were analyzed using Stata version 14 (StataCorp LLC, College Station, TX, USA). Continuous variables are reported as mean with standard deviation, and frequency with percentage are reported for categorical variables. The Student t-test, chi-square test, or Fischer exact p-values were used to assess the association of variables with mortality. Applied logistic regression was used to determine the predictors of mortality and acute kidney injury (AKI). A p-value of <0.05 was considered statistically significant.
Results
A total of 574 patients aged ≥18 years who tested positive for SARS-COV-2 via polymerase chain reaction testing of nasal swab samples and were admitted to the dedicated COVID unit were included in this study. Of the 574 enrolled patients, more men than women were included (69.7% vs. 30.3%). The mean patient age was 55.6 ± 14.4 years. Patients who died had more comorbidities as compared with those who survived, including ischemic heart disease (34.9 vs. 14.7%, p < 0.001) and chronic kidney disease (CKD) (19.1% vs. 6.7%, p = 0.008). The proportion of critical COVID-19 cases was higher among patients who did not survive than among those who survived (85.7% vs. 22.1%, p < 0.001) (Table 1).
Demographic characteristics of patients admitted with COVID-19.
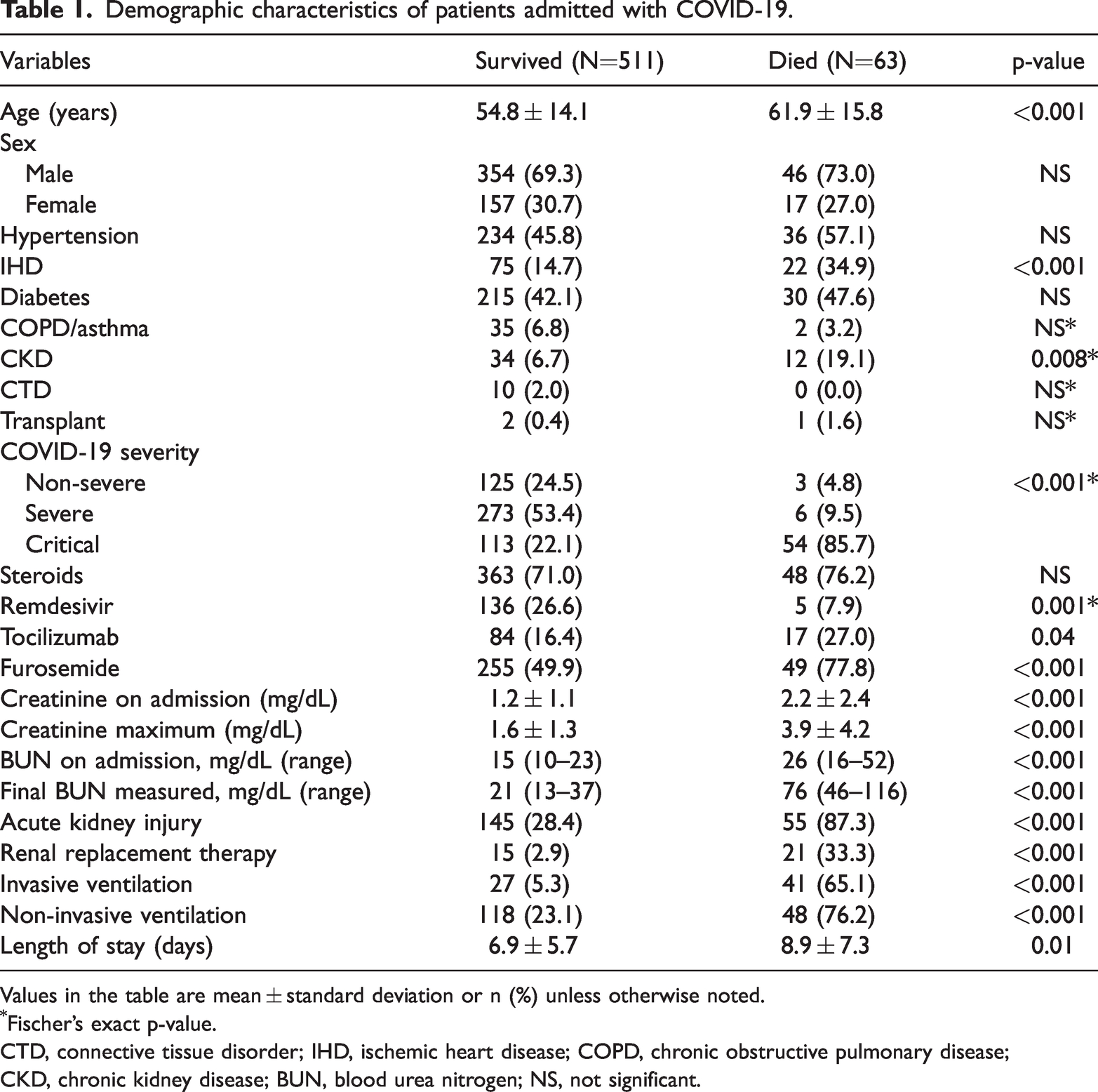
Values in the table are mean ± standard deviation or n (%) unless otherwise noted.
Fischer’s exact p-value.
CTD, connective tissue disorder; IHD, ischemic heart disease; COPD, chronic obstructive pulmonary disease; CKD, chronic kidney disease; BUN, blood urea nitrogen; NS, not significant.
Based on the severity of COVID-19 infection, patients were treated conservatively or administered remdesivir, steroids, or tocilizumab (Table 2). During the hospital stay, 107 (18.6%) patients developed hypernatremia, among which nine (8.4%) patients had sodium levels >165 mEq/L (Table 3). The maximum serum sodium at any time during the hospital stay was 140.6 ± 5.0 mEq/L; however, this was 151 ± 9.9 mEq/L among patients who died. Similarly, the mean final serum sodium at the time of discharge or death (within 48 hours) was 145.4 ± 9.4 mEq/L in patients who died as compared with 137.7 ± 3.7 mEq/L in patients who survived.
Drugs administered according to COVID-19 severity.

Comparison of sodium levels at different stages in patients who survived or died.

Values in the table are mean ± standard deviation or n (%).
Fischer’s exact p-value.
The mean serum creatinine on admission was 1.3 ± 1.3 mg/dL. Overall, 34.8% of patients developed AKI (18.5% had underlying CKD); among them, 50.5% of patients developed stage 1 AKI, 24.5% had stage 2, and 25% had stage 3 AKI. Approximately 6.3% of patients required renal replacement therapy. AKI was significantly higher in patients who died as compared with those who survived (87.3% vs. 28.4%, p < 0.001). We also assessed the predictors of mortality and AKI (Table 4). The final serum sodium level, checked within 48 hours prior to discharge or death, was found to be significant in patients who died (odds ratio [OR]: 1.22, 95% confidence interval [CI]: 1.13–1.32, p < 0.001). Admission and maximum sodium levels did not show any significant association with mortality. Final serum sodium levels did not show a significant association with the development of AKI (OR: 1.49, 95% CI: 1.00–2.23). Other predictors of mortality included ischemic heart disease (OR: 3.65, 95% CI: 1.39–9.61), acute kidney injury (OR: 6.07, 95% CI: 2.39–15.42), invasive ventilation (OR: 28.4, 95% CI: 11.14–72.40), and length of stay (OR: 0.91, 95% CI: 0.86–0.97).
Predictors of mortality and acute kidney injury.
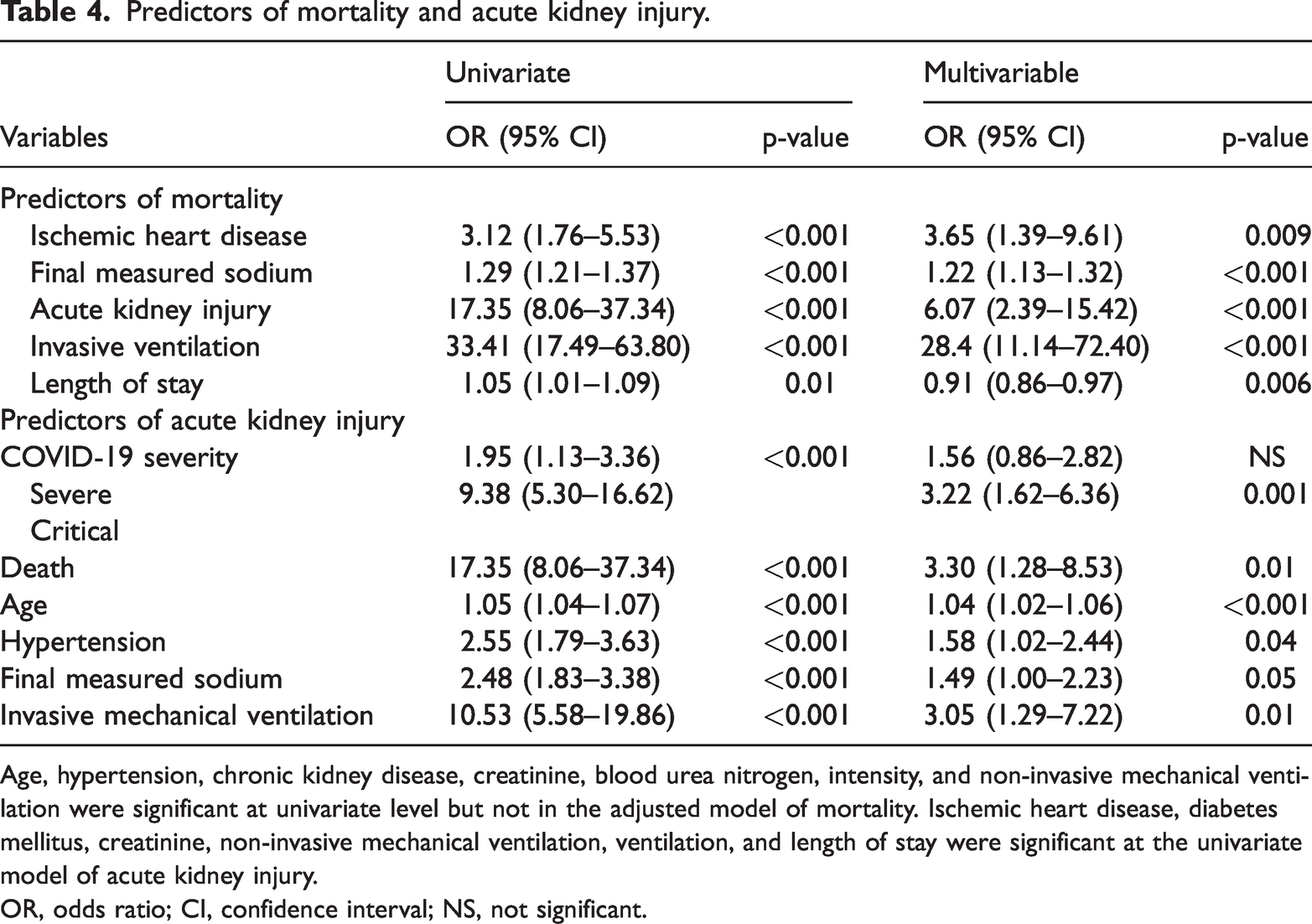
Age, hypertension, chronic kidney disease, creatinine, blood urea nitrogen, intensity, and non-invasive mechanical ventilation were significant at univariate level but not in the adjusted model of mortality. Ischemic heart disease, diabetes mellitus, creatinine, non-invasive mechanical ventilation, ventilation, and length of stay were significant at the univariate model of acute kidney injury.
OR, odds ratio; CI, confidence interval; NS, not significant.
Discussion
In this retrospective study of 574 patients with COVID-19, we observed a significant difference in serum sodium levels at the time of admission, as well as at any point during their hospital stay, between patients who survived (135.4 ± 5.7 mEq/L) and those who did not survive (140.3 ± 9.7 mEq/L). The principal finding in our study was the development of hypernatremia during the hospital stay (increasing to 18.6% from 4.7%), with a significant presence (42.9%) among patients who did not survive.
In our study, the proportion of patients with dysnatremia on admission was approximately 43.7%, with 39% exhibiting hyponatremia and only 4.7% with hypernatremia. In a retrospective longitudinal study by Tzoulis et al., dysnatremia was present in approximately 29.9% of patients on admission, and most patients (24.6%) had hyponatremia, which is comparable to our study. 4 Similarly, Hirsh et al. found dysnatremia in 51.7% of patients on admission, 44.6% with hyponatremia and 7% with hypernatremia. 6 Another case series of 12 patients with critical COVID-19 showed treatment-resistant hypernatremia in 50% of patients. 7 In a recent systematic review and meta-analysis regarding the association of outcomes in COVID-19 patients with hypernatremia, the incidence of hypernatremia ranged from 10.69% to 12.17%. This difference in the incidence of hypernatremia could be attributed to the threshold used to define hypernatremia (145 mEq/L to >147 mEq/L) as well as the timing of sodium measurements. 17
The increased development of hypernatremia in patients with COVID-19 could be attributed to low water intake owing to loss of taste, anorexia, nausea, or other gastrointestinal complaints. Additionally, increased insensible losses owing to continuous fever could be a contributing factor. Moreover, it has been proposed that there is increased absorption of sodium by the renal tubules owing to activation of the renin–angiotensin–aldosterone system in patients with COVID-19 infection. 18 These patients usually need good hydration; however, owing to lung involvement in COVID-19, a conservative approach to fluid management is often preferred to protect the lungs. This conservative approach, along with intermittent furosemide administration given to patients with increasing shortness of breath, reduced urine output, or worsening lung infiltrates on chest X-ray, can further predispose to or exacerbate hypernatremia. A significant improvement in oxygenation after intravenous furosemide in patients with tomographic evidence of pulmonary edema and fluid overload was demonstrated by Santos et al. using a “negative fluid balance approach.” 19 Notably, none of the patients in this study required mechanical ventilation. Additionally, a separate retrospective analysis highlighted a reduced likelihood of mechanical ventilation and 28-day mortality in patients treated with a combination of steroids and furosemide. 20
We observed a significant increase in hypernatremia during the hospital stay, rising from 4.7% to 18.6%, as opposed to hyponatremia, where the percentage dropped to 5.4% from 39%. Remarkably, among patients who died, the proportion with hypernatremia further rose to 42.9%, in sharp contrast to the presence of hyponatremia, which was recorded in just 11.1% of cases. This observation coincides with the findings of Tzoulis et al. where hyponatremia on admission was present in 24.6% of patients; however, this was not identified as a contributing factor for increased mortality among patients with COVID-19. 4 Conversely, hypernatremia at any time during the hospital stay is associated with a three-times greater risk of death. 4 Several different factors have been presumed to play a role in hyponatremia among patients with COVID-19 such as inflammation, 21 syndrome of inappropriate antidiuretic hormone secretion, 5 and most frequently, volume depletion. 4 As per Hirsch et al., the odds of death in patients with moderate to severe hyponatremia is 1.26 (95% CI: 1.05–1.51) as compared with 2.06 (95% CI: 1.57–2.70) in hypernatremic patients. 6
Our study findings confirmed that AKI is frequent in patients with COVID-19 infection, with a recorded rate of 34.8%, which is comparable to the 36.9% and 36.6% reported for cohorts in the United Kingdom and New York, respectively.8,22 The association between AKI and hypernatremia is not yet clear. One study showed that hypernatremia is an independent predictor of mortality in patients with COVID-19 infection, irrespective of AKI. 23 Similarly, another study revealed no association between serum sodium at any point and the risk of AKI. 4 In our subgroup analysis, neither hypernatremia nor hyponatremia was an independent predictor of AKI. However, the final sodium measurement was a statistically significant predictor of mortality in our patient population.
Several studies have identified increasing age as a risk factor for both disease progression and mortality in patients infected with COVID-19. A study from China showed the mortality rate in patients aged 59 years or older to be 5.1 times higher than in patients aged between 30 and 59 years. 24 In our study, although the average age of patients who died was older than that among patients who survived, this factor did not reach statistical significance in multivariate analysis.
COVID-19 infection demonstrates a predisposition toward male individuals. Based on a meta-analysis comprising 36,470 patients, the probability of male patients being diagnosed with COVID-19 infection was 8% higher than that of their female counterparts. 25 Our study also showed an increased frequency of COVID-19 infection in male (69.6%) versus female (30.3%) individuals. This higher predisposition in male individuals may be explained by lifestyle factors such as smoking, 26 a weakened immune system owing to genetic and hormonal factors 26 or increased levels of angiotensin-converting enzyme 2 receptors in the pulmonary endothelium. 27
Another important finding in our study was the low use of remdesivir among patients who died. The use of remdesivir in patients who have critical COVID-19 requiring invasive ventilation, or in those requiring high-flow oxygen or non-invasive ventilation, has not been clearly defined. Similarly, the World Health Organization has not recommended use of remdesivir in all patients with COVID-19 infection as its role in reducing the mortality rate or hospital stay is not certain. 28
The main strength of this study is its longitudinal design; dynamic serum sodium levels were available throughout the admission period and the effects of hyponatremia, hypernatremia, and normonatremia were analyzed at different stages of hospitalization.
This study also has several limitations. First, the findings may not be generalizable as this was a single-center study. Second, we did not have data on urine and serum osmolality and urinary electrolytes, which could be helpful to accurately determine the cause of hyponatremia. In addition, owing to unavailability of an electronic health record system, we did not have data regarding fluid resuscitation administered to the included patients. Furthermore, whereas furosemide was frequently used in our patients, the criteria for its use were not well defined; the dosage ranged from 20–40 mg intermittently to continuous intravenous furosemide infusion, according to the physician’s discretion. This lack of accurate dosing information is another limitation. Lastly, although we found an association between hypernatremia and mortality, a causal relationship cannot be established because this study was observational and retrospective in nature.
Conclusion
In the present study, we observed that hypernatremia was more common than hyponatremia among critically ill patients with COVID-19 infection. Further research is required to establish a clear association between hypernatremia and mortality. Furthermore, whereas the serum sodium level at any point during hospitalization was not found to be a predictor of AKI, AKI emerged as a main predictor of mortality in patients with COVID-19 infection.
Footnotes
Author contributions
FG and MMY contributed to the study concept and design; FG, SS, NV and MA performed analysis and interpretation of the results. All authors have read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of conflict of interests
The authors declare there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
