Abstract
Various immunity-related adverse events have been reported to be associated with the inhibition of programmed cell death receptor 1. We report a rare case of a relapse of lupus nephritis (LN), involving rapidly progressive glomerulonephritis, which was induced by nivolumab treatment in a patient with oral cancer. The patient had a history of systemic lupus erythematosus and underwent treatment with steroids, rituximab, and plasmapheresis. However, her renal function did not improve, and she died of multiple organ failure. To our knowledge, this is the first description of severe LN induced by nivolumab.
Keywords
Introduction
Immune checkpoint inhibitors (ICIs) are a recent class of anti-cancer agents that are effective treatments for a variety of malignancies. Programmed cell death receptor-1 (PD-1) is an immune checkpoint receptor that is expressed on activated lymphocytes and suppresses immune responses. Medications such as nivolumab that block the PD-1/PD-1 ligand interaction and promote anti-tumor T-cell activation have become important tools in cancer therapy. 1 ICIs increase the activity of the immune system by inhibiting the immune escape of tumor cells, sometimes at the cost of the deregulation of immune tolerance, leading to immunity-related adverse events. 2 Here, we present a case of lupus nephritis (LN) induced by nivolumab in an 84-year-old patient with oral cancer.
Case presentation
A woman in her early 80 s was referred to our hospital in April 2021 because of palatal pain of 3 months’ duration. She had a history of systemic lupus erythematosus (SLE) that had been diagnosed 32 years previously, but during the preceding 10 years, she had not experienced any serious symptoms or taken any medication.
On admission, intraoral examination of the patient revealed a multilobular mass measuring approximately 40 × 40 mm in the right hard palate, which extended to the maxillary gingiva. The results of imaging were consistent with an oral malignant tumor (Figure 1). She underwent a lumpectomy, and was diagnosed with oral cancer (stage IV, cTXNXM1) on the basis of histopathology (Figure 2).

Computed tomography images showing a mass in the right hard palate (arrow). (a) Median sagittal section and (b) Transverse section.
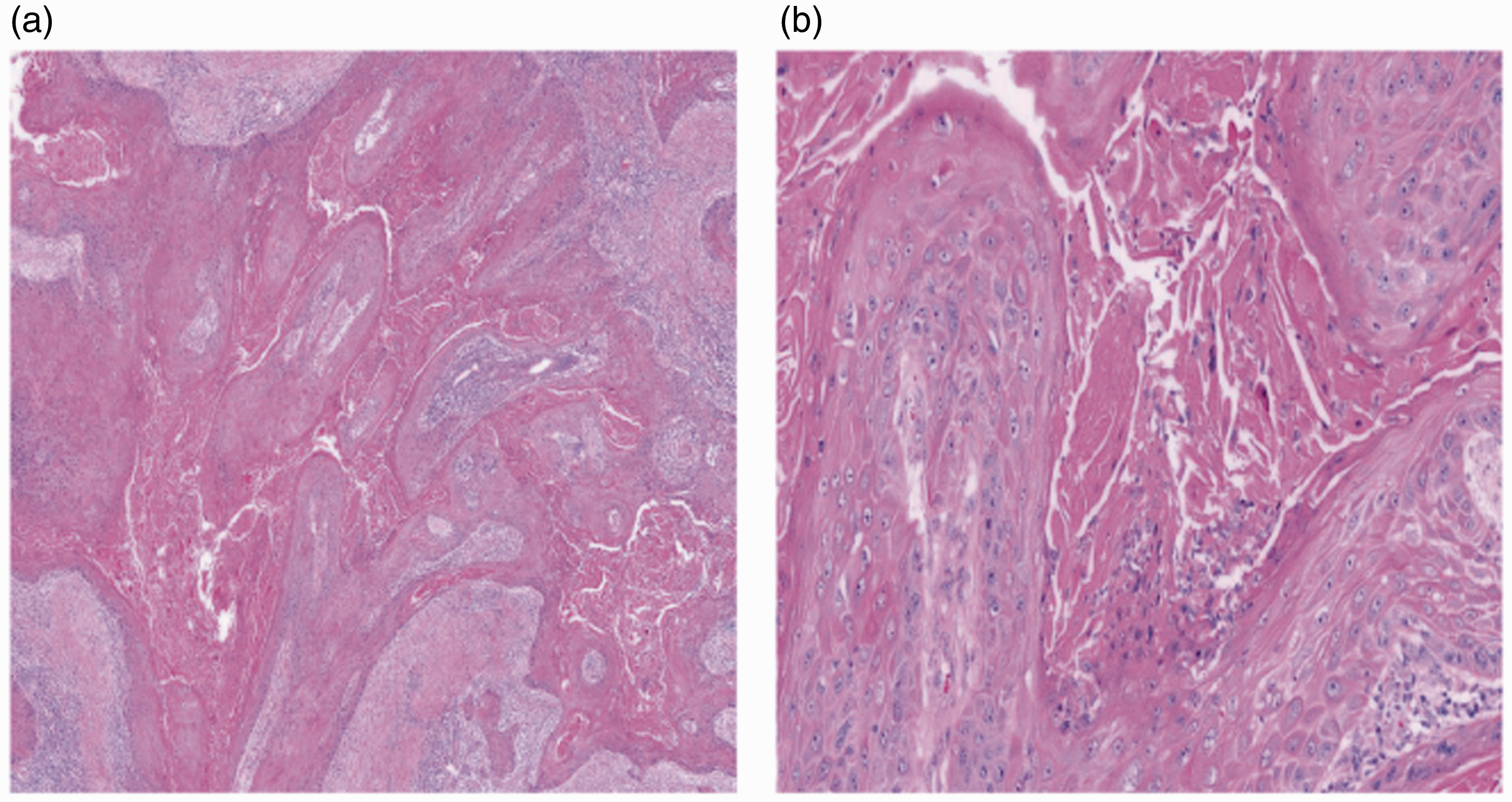
Light micrographs showing an oral squamous cell carcinoma. Hematoxylin and eosin staining. Magnifications: (a) ×20 and (b) ×100.
Postoperative chemotherapy (cisplatin and gemcitabine) and radiotherapy (30 Gy in 10 fractions) were administered. Ten months postoperatively, computed tomography showed local recurrence and distant metastases. The patient underwent a further lumpectomy and nivolumab therapy (140 mg every 2 weeks; 3 doses). Her kidney function was normal at that time, and she showed no microscopic hematuria or proteinuria.
One month after her first nivolumab treatment, the patient presented to our department with systemic edema and oliguria. Her serum creatinine concentration was 5.6 mg/dL, her serum albumin concentration was 24 g/L, and she had a proteinuria of 4.73 g/24 hours, along with hematuria. Immunological testing revealed hypocomplementemia and an anti-nuclear antibody titer of 1:640, with positivity for anti-ds-DNA and anti-Smith antibodies. The examination of a kidney biopsy, as shown in Figure 3, revealed findings suggestive of active LN. Specifically, there was proliferative glomerulonephritis and cellular or fibrocellular crescents involving 60% of the glomeruli, immunopathological testing showed a “full house” pattern, typical of LN, and electron microscopy revealed electron-dense deposits in the mesangial and subendothelial areas.

Photomicrographs of sections through renal biopsy samples obtained from the patient. (a) Immunofluorescence staining for immunoglobulin G (+++). (b) Immunofluorescence staining for complement C3 (+++). (c) Immunofluorescence staining for complement C1q (+++). (d) Cellular crescent in a glomerulus (periodic acid-Schiff staining). (e) Endothelial cell proliferation in a glomerulus (periodic acid-Schiff staining) and (f) Fibrinoid necrosis in a glomerulus (period acid-silver methenamine staining). Magnification ×100.
The patient had experienced a relapse of lupus. In addition to LN, she had a low hemoglobin concentration, a low platelet count, and new-onset butterfly erythema. Because of oliguria and a rapid deterioration of renal function, hemodialysis and plasma exchange (50 ml/kg/48 hours) were performed. In addition, she was administered methylprednisolone (MP) 500 mg/day for 3 days, followed by MP 40 mg/day, and rituximab (375 mg/m2 once).
Despite this therapy, the patient’s kidney function did not improve, and pulmonary edema and pulmonary infection developed. Her condition continued to worsen, and she decided to discontinue treatment other than supportive care. She died 12 days after hospital admission because of multiple organ failure.
This study was approved by the Ethics Committee of Lishui Central Hospital (approval no. 2022034). We obtained the written informed consent of the patient for treatment and the informed consent of her relatives for the publication of the case report. The reporting of this case conforms to the CARE guidelines. 3
Discussion
Anti-PD-1 agents are widely used to treat several late-stage malignancies, such as melanoma and lung cancer, by inhibiting the interaction between the PD-1 and its ligand. In recent years, these agents yield durable responses and to improve the survival of patients with recurrent or metastatic head and neck squamous cell carcinoma, including primary oral malignancies, and are now being evaluated for the treatment of locoregionally advanced tumors. 4 , 5 However, there have been few studies regarding the safety of anti-PD-1 agents for the treatment of patients with autoimmune disorders.
Patients with a medical history of autoimmune disease are at risk of the reactivation or exacerbation of their disease when taking ICIs, because these drugs can increase the activity of the immune system. In a letter, Fadel et al. reported a case of LN induced by ipilimumab in a patient with metastatic melanoma, and warned clinicians to be aware of this potentially severe complication. 6 A systematic review of the use of ICIs in 123 patients with pre-existing autoimmune disease showed that 50% experience an exacerbation of their disease and 34% experience de novo immunity-related adverse events. 7 Furthermore, only two of the patients with SLE did not experience disease exacerbation or immunity-related adverse events. Cutaneous lupus erythematosus in the setting of pembrolizumab therapy for metastatic melanoma has been described in some previous case reports, including one of a patient who was undergoing nivolumab therapy for metastatic lung cancer.8–11 The clinical course of the present patient suggested severe SLE and a flare-up of LN following nivolumab therapy. The use of nivolumab has rarely been described to be associated with glomerulonephritis; the most common renal pathology reported has been acute tubulointerstitial nephritis. 12 To the best of our knowledge, this is the first case of LN-related, rapidly progressive glomerulonephritis to be reported that was induced by nivolumab. Nivolumab might induce LN directly or indirectly, through the activation of SLE-related antibodies. The severity of ICI-related glomerulonephritis and responses to treatments differ, at least in part because of variations between cases. It is possible that the poor outcome in the present patient was the results of severe SLE/LN, the malignancy, infection, and/or old age.
In summary, we have reported a case of a patient with oral cancer who presented with LN-related, rapidly progressive glomerulonephritis following nivolumab treatment. In our review of the literature, we did not find previous reports of LN induced or exacerbated during ICI treatment in patients with oral cancer or other conditions. Therefore, the present case might be the first described, and it will be through the study of similar cases in the future that our hypothesis may be confirmed.
Footnotes
Author Contributions
Z.Z. and X.W. drafted the manuscript; X.W. revised the manuscript; and Q.Z. was responsible for the microbiological identification of the pathogen and helped to draft the manuscript. All the authors have approved the final version of the manuscript and agree with its publication.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
This study was approved by the Ethics Committee of Lishui Central Hospital (approval no. 2022034). We obtained the written informed consent of the patient for treatment and the informed consent of her relatives for the publication of this case report.
Funding
This study was supported by funds from the Public Benefit Project of Lishui (2021GYX19), the Medical and Health Project of Zhejiang (2021427592, 2023583325), the Project of the Zhejiang Science and Technology Department (2022C03172), and the National-Zhejiang Administration of Traditional Chinese Medicine cooperation project (GZY-ZJ-KJ-23099).
