Abstract
Only a few cases of renal vein thrombosis (RVT) occurring in patients with vasculitis have been reported. RVT associated with vasculitis and hemolytic anemia has not been reported yet. We describe here a patient with RVT complicated by pulmonary embolism, autoimmune hemolytic anemia, and eosinophilic granulomatous polyangiitis. A 69-year-old Japanese man who had been treated with corticosteroids was referred to our department for severe proteinuria (4.32 g/gCr). Abdominal ultrasonography showed bilateral RVT, and contrast-enhanced computed tomography showed bilateral pulmonary embolism. Therefore, the patient was diagnosed with RVT complicated by pulmonary embolism. Anticoagulation therapy with heparin followed by apixaban was started. Thereafter, the D-dimer concentration decreased from 8.3 to 1.2 μg/mL, and urinary protein excretion improved to 0.62 g/gCr. Renal function was unchanged with an estimated glomerular filtration rate of 68.8 mL/minute/1.73 m2. The thrombi in both renal veins and pulmonary arteries gradually regressed. Clinicians should be aware of this complication when worsening proteinuria is observed during steroid therapy in patients with autoimmune hemolytic anemia and eosinophilic granulomatous polyangiitis.
Keywords
Introduction
Renal vein thrombosis (RVT) is a rare but serious condition in which a blood clot forms in the renal vein, resulting in various sequelae, such as hematuria, proteinuria, renal dysfunction, and pulmonary embolism. 1 RVT is sometimes observed as a complication of nephrotic syndrome, malignancy, and coagulopathy. 2 Vasculitis and hemolytic anemia reportedly increase the risk of thrombosis;3,4 therefore, they are considered risk factors for RVT. 1 However, only a few cases of RVT occurring in patients with vasculitis have been reported.5,6 Furthermore, RVT associated with hemolytic anemia has not been reported yet. We report a case of RVT complicated by pulmonary embolism in a patient with autoimmune hemolytic anemia (AIHA) and eosinophilic granulomatous polyangiitis (EGPA).
Case report
The reporting of this study conforms to the CARE guidelines. 7 The patient was a 69-year-old Japanese man who had been treated with corticosteroids and immunosuppressants for AIHA and EGPA since the age of 68 years. He also had a 60-year history of asthma. He was taking prednisone at 15 mg/day, tacrolimus at 3.5 mg/day, trimethoprim-sulfamethoxazole at 480 mg/day, and vonoprazan at 20 mg/day. Eight weeks before admission to hospital, he developed dyspnea.
On the admission day, he visited our hospital for a regular medical examination and urinalysis showed proteinuria (3+). Therefore, he was referred to our department for further examinations. His blood pressure was 164/100 mmHg, heart rate was 118 beats/minute, respiratory rate was 12 breaths/minute, and oxygen saturation was 94% in room air. Fine crackles were audible in the back lower lung fields and mild leg edema was detected. Laboratory data showed an elevation of the C-reactive protein concentration (44.6 µg/L), white blood cell count (11,530/µL), and D-dimer concentration (8.3 μg/mL), as well as hypoalbuminemia (27 g/L). The serum creatinine level was 76.93 µmol/L, and the estimated glomerular filtration rate was 67.3 mL/minute/1.73 m2. Urinalysis showed proteinuria (4.32 g/gCr). No microscopic hematuria was present. The laboratory data obtained at the time of referral are shown in Table 1. No abnormalities were found in the patient’s chest X-ray or electrocardiogram. Abdominal ultrasonography showed bilateral RVT with enlarged kidneys. Contrast-enhanced computed tomography (CT) was then performed, which showed bilateral pulmonary embolism and bilateral RVT. Therefore, the patient was diagnosed with RVT complicated by pulmonary embolism and was admitted to our department.
Patient’s laboratory results at the time of referral to our department
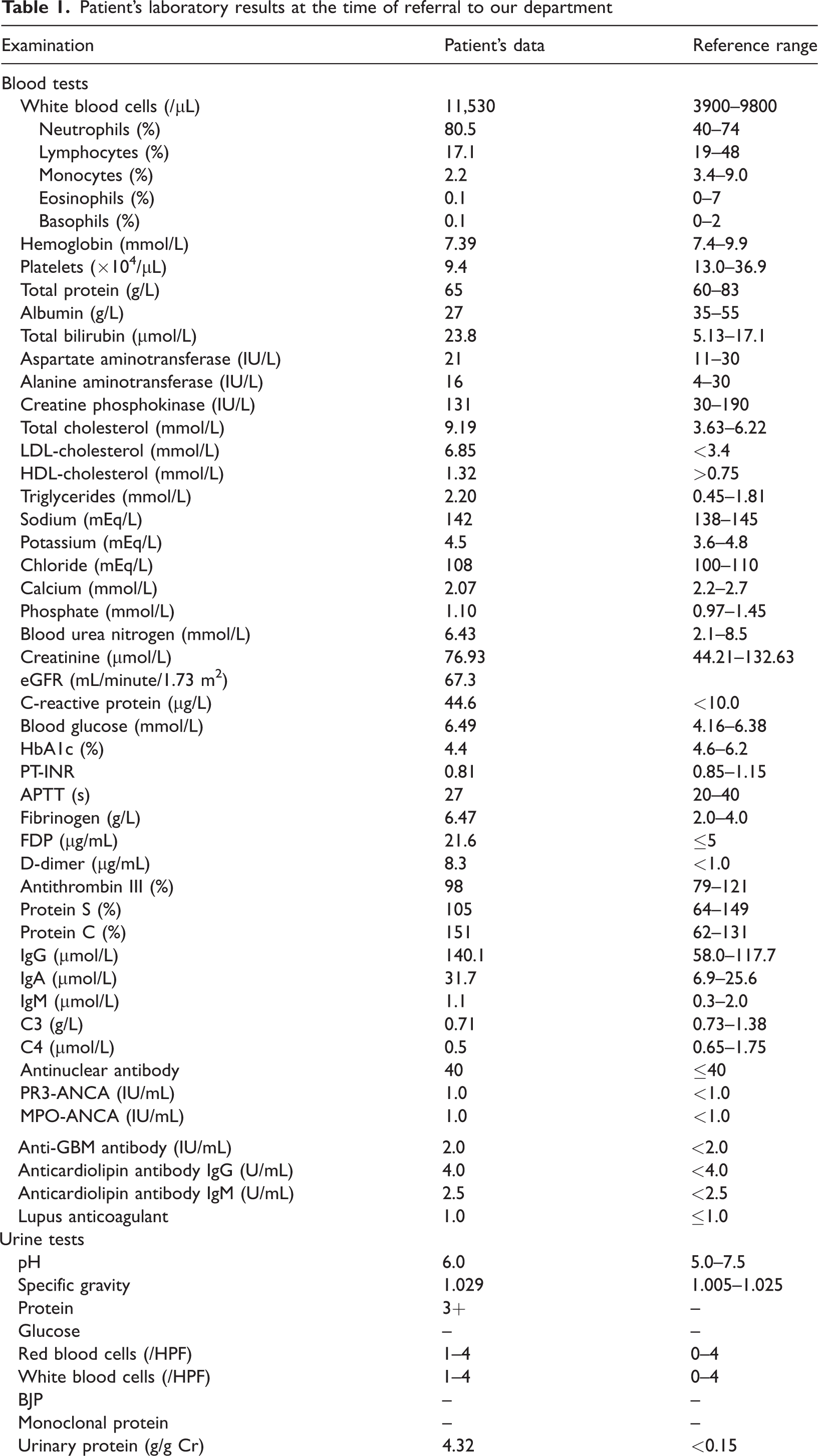
LDL, low-density lipoprotein; HDL, high-density lipoprotein; eGFR, estimated glomerular filtration rate; HbA1c, hemoglobin A1c; PT-INR, prothrombin time-international normalized ratio; APTT, activated partial thromboplastin time; FDP, fibrinogen/fibrin degradation products; IgG, immunoglobulin G; IgA, immunoglobulin A; IgM, immunoglobulin M; PR3, proteinase-3; ANCA, anti-neutrophil cytoplasmic antibody; MPO, myeloperoxidase; anti-GBM, anti-glomerular basement membrane antibody; HPF, high-power field; BJP, Bence-Jones protein.
Note: the eGFR was calculated using a modified version of the Modification of Diet in Renal Disease formula of the Japanese Society of Nephrology: eGFR (mL/minute/1.73 m2) = 194 × −0.287 × serum creatinine−1.094 (×0.739 for women).
Consent to have treatment performed was obtained from the patient. His clinical course after admission is shown in Figure 1. Anticoagulation therapy with heparin was started. The patient was negative for anticardiolipin antibody and lupus anticoagulant. Protein C and protein S were within the normal range. A follow-up contrast-enhanced CT scan on hospital day 9 showed no improvement of the thrombosis. The heparin was switched to apixaban on hospital day 10 because his vital signs were stable, and he was discharged on hospital day 11. At the 191-day follow-up visit, the patient’s D-dimer concentration had decreased to 1.2 μg/mL and urinary protein excretion had decreased to 0.62 g/gCr. However, his renal function was unchanged with an estimated glomerular filtration rate of 68.8 mL/minute/1.73 m2. Contrast-enhanced CT showed regression of the thrombi in both renal veins and disappearance of the thrombi in both pulmonary arteries. He then underwent regular outpatient treatment in our department.

Patient’s clinical course. The horizontal axis shows the number of days from the initiation of anticoagulation therapy. Renal vein thrombosis can be seen on axial ultrasonography and axial CT (arrows). Pulmonary embolism can also be seen on axial CT (arrows). No improvement was observed until day 130, but CT on day 191 showed shrinkage of the renal vein thrombosis and disappearance of the pulmonary embolism. CT, computed tomography; eGFR, estimated glomerular filtration rate.
Discussion
RVT is a rare disease in which thrombi form in the renal veins, and it is mainly observed in men with cancer, nephrotic syndrome, infection, coagulation abnormality, or a history of kidney surgery. 2 However, our patient had none of these risk factors in his background. He had been receiving steroid therapy for AIHA and EGPA for 1 year. AIHA and EGPA promote platelet activation,3,4 and corticosteroid therapy induces thrombus formation by increasing clotting factors. 8 These combined effects might have led to the RVT in our case (Figure 2). Further investigation is required to clarify the mechanism of development of RVT in patients with AIHA and EGPA.

Suggested mechanism of the development of renal vein thrombosis in patients with EGPA and AIHA. AIHA, autoimmune hemolytic anemia; EGPA, eosinophilic granulomatosis with polyangiitis.
RVT generally causes abdominal pain, microscopic hematuria, proteinuria, renal dysfunction, and pulmonary embolism. 1 Our patient had nephrotic-range proteinuria at the onset of the disease, and his urinary protein excretion was improved by anticoagulation therapy; therefore, we considered that the proteinuria was due to RVT. However, he did not have microscopic hematuria, renal dysfunction, or abdominal pain. Ultrasonography showed thrombi in both renal veins with protrusion into the inferior vena cava, and contrast-enhanced CT showed thrombi in both pulmonary arteries. The presence of multiple risk factors for the development of thrombosis may have contributed to the progression of the thrombi in our case. Vasculitis can be complicated by thrombotic microangiopathy, which leads to hematuria, proteinuria, and renal dysfunction. 9 Our patient might have had thrombotic microangiopathy because he showed nephrotic-range proteinuria and mild renal dysfunction. However, we could not perform a renal biopsy owing to a severe bleeding risk because the patient was receiving anticoagulation therapy for RVT complicated by pulmonary embolism. Further investigations are necessary to clarify the pathogenesis of proteinuria and renal dysfunction in patients with AIHA and EGPA.
Recently, to decrease complications, treatment of RVT has shifted from surgical treatment to medical treatment, starting with intravenous anticoagulants and then shifting to oral anticoagulants. 1 Anticoagulation therapy is not necessarily required in patients with asymptomatic RVT, but is indicated when the condition is complicated by pulmonary embolism. 1 The duration of anticoagulation therapy has not been clearly defined yet. One report suggested that anticoagulants should be continued until the primary disease achieves complete remission because the discontinuation of anticoagulation therapy may cause recurrence of RVT in patients with highly active primary disease. 10 Another report recommended continuing the anticoagulant until the serum albumin concentration reaches ≥25 g/L. 1 No literature to date has shown an association between an improvement in thrombosis and the duration of treatment. In this case, RVT was discovered incidentally, and treatment with intravenous heparin was started because of coexistent pulmonary embolism. The patient was then switched to a direct oral anticoagulant and showed slow regression of the thrombi in both renal veins and both pulmonary arteries. Lifetime anticoagulation therapy might be necessary for this patient because of his steroid therapy for AIHA and EGPA and the presence of residual thrombi in both renal veins.
The life prognosis of RVT due to malignancy is poor, and the reported 5-year survival rate is approximately 20%. 2 In contrast, the prognosis of RVT without malignancy is favorable, with a 5-year survival rate of 80%. 2 In addition, one study showed that reopening of the renal vein following anticoagulation therapy was not associated with a better prognosis in patients with RVT, and that the most important factor associated with a favorable prognosis was renal function at the time of onset of RVT. 11 Our patient’s prognosis was expected to be favorable because no malignancy was observed and his renal function was normal at the time of diagnosing RVT. However, even at the time of this writing, careful monitoring was still required because the patient’s condition was complicated by pulmonary embolism and thrombi still remained in his renal veins.
In conclusion, we report a case of RVT complicated by pulmonary embolism in a patient with AIHA and EGPA. Clinicians should be aware of this complication when worsening proteinuria is observed during steroid therapy in patients with AIHA and EGPA.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231200272 - Supplemental material for A case of renal vein thrombosis associated with autoimmune hemolytic anemia and eosinophilic granulomatous polyangiitis
Supplemental material, sj-pdf-1-imr-10.1177_03000605231200272 for A case of renal vein thrombosis associated with autoimmune hemolytic anemia and eosinophilic granulomatous polyangiitis by Yusaku Watanabe, Keiji Hirai, Momoko Matsuyama, Taisuke Kitano, Kiyonori Ito, Tsuyoshi Kobashigawa, Susumu Ookawara and Yoshiyuki Morishita in Journal of International Medical Research
Footnotes
Author contributions
Tsuyoshi Kobashigawa and Momoko Matsuyama diagnosed the patient and Keiji Hirai supervised this case report. Yusaku Watanabe wrote the manuscript. Taisuke Kitano and Kiyonori Ito reviewed and edited the manuscript. Susumu Ookawara made critical revisions. Yoshiyuki Morishita approved the final version. All authors read and approved the manuscript.
Data availability statement
The data supporting the findings of this case are available from the corresponding author upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
Written informed consent was obtained from the patient for publication of this case report. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Approval for publication was obtained from the institutional review board of Saitama Medical Center, Jichi Medical University, Japan (approval no. RIN 15-33).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
