Abstract
We present the case of a woman of 50 years of age who experienced widespread bone pain along with digestive symptoms, including nausea and vomiting. She had been prescribed tenofovir disoproxil fumarate (TDF) tablets for the treatment of hepatitis B. Laboratory testing revealed low circulating phosphorus and potassium concentrations and acidosis. A whole-body bone scan revealed abnormal bone metabolism. Rheumatologic and urologic conditions were ruled out, and therefore TDF-induced Fanconi syndrome (FS) and related bone pain was diagnosed. After the TDF was discontinued, the patient’s symptoms and laboratory indices significantly improved. In this manuscript, we highlight the clinical manifestations of and laboratory test results associated with FS and summarize the cases of TDF-induced FS reported on PubMed between 2013 and 2022 to improve understanding of FS.
Keywords
Introduction
Fanconi syndrome (FS) is caused by a group of renal tubular functional disorders that develop because of genetically determined or acquired abnormalities of the proximal renal tubules. 1 The features of FS include glycosuria, aminoaciduria, hypercalciuria, renal sodium loss, hypophosphatemia, hypouricemia, proximal renal tubular acidosis, and proteinuria in the renal tubules; and related symptoms, such as muscle weakness, flaccid paralysis, periodic paralysis, and tetany. 2 FS is typically acquired in adulthood, and adult FS can be caused by systemic disease, drugs, and other factors. The systemic diseases that cause FS include amyloidosis, multiple myeloma/light chain disease, paroxysmal nocturnal hemoglobinuria, kidney transplantation, tubulointerstitial nephritis, and membranous nephropathy with anti-tubular basement membrane antibodies. The drugs that induce FS include nucleoside reverse transcriptase inhibitors (NRTIs), anti-cancer drugs, anti-epileptic drugs, antibiotics, DNA polymerase inhibitors, and sulfonylureas. Heavy metals (e.g., lead, cadmium, mercury, copper), L-arginine and L-ornithine, aristolochic acid (a cause of herbal nephropathy), and fumaric acid are some other factors that cause FS.3–11 Of these, drugs are the most frequent cause of FS. 7
Tenofovir disoproxil fumarate (TDF) is an NRTI antiviral drug. It has its antiviral effect when it is converted to tenofovir by esterases and subsequently phosphorylated by cellular enzymes to form tenofovir diphosphate. Tenofovir diphosphate acts by competing with 5′-deoxyadenosine triphosphate, a natural substrate, and integrating within DNA, thereby inhibiting viral reverse transcriptase activity. 12 However, TDF can have various adverse effects, including allergic reactions, hypophosphatemia, hypokalemia, lactic acidosis, severe hepatomegaly with steatosis, respiratory distress, abdominal pain, pancreatitis, severe acute exacerbation of hepatitis B, high circulating liver enzyme activities, and hepatitis. Moreover, the prolonged use of TDF can be associated with additional complications, such as lower bone density, myopathy, new or worsening renal impairment, renal failure, FS, and osteomalacia. 13 Of 1537 patients with human immunodeficiency virus (HIV) who were administered TDF, 13, with a mean age of 43.9 years, developed FS. The mean duration of TDF use prior to the onset of FS was 55 months (range: 12 to 98 months) and the incidence was approximately 1/1000. 14
Here, we present the case of a patient with FS that developed secondary to the administration of TDF for the treatment of hepatitis B. We also discuss the diagnosis and treatment of FS in the present patient in the context of a literature review.
Case report
A woman of approximately 50 years of age was admitted to our hospital because of repeated episodes of nausea and vomiting, and bone pain, of 2 months’ duration. She had sought medical help at several hospitals and had been repeatedly misdiagnosed with acute gastroenteritis and osteoporosis. She presented to our hospital in November 2022, when she reported that she had experienced repeated episodes of nausea and dry heaving since early September, accompanied by significant loss of appetite; constipation; left subcostal, back, shoulder, elbow, and wrist pain; and progressive weight loss of approximately 10 kg. She had a history of viral hepatitis type B that had been diagnosed a year previously and had been taking TDF (300 mg daily per os) since then. She also had a 1-year history of a mental disorder that had been treated with olanzapine (5 mg in the morning, 10 mg at night) and sodium valproate (5 mg in the morning and at night), but she had stopped this medication 6 months previously. She had no history of hypertension or diabetes. At the time of admission, she was in a poor mental and nutritional state and had a body mass index of 17.5 kg/m2. The reporting of this case conforms to the CARE guidelines. 15 The detailed information provided regarding the patient has been de-identified.
The laboratory results (Table 1; Figure 1) for the patient showed high circulating alkaline phosphatase (ALP) activity; low 25-hydroxyvitamin D (25(OH)D), calcium, phosphorus, potassium, uric acid, and total CO2 concentrations; low estimated glomerular filtration rate (eGFR); and positivity for urinary protein, glucose, and ketones. These results indicate abnormalities in electrolyte balance, bone metabolism, and renal excretion, as well as metabolic acidosis. However, the concentrations of rheumatism-related antibodies, tumor markers, and thyroid hormones were not abnormal. A whole-body emission computed tomography (bone) scan suggested abnormal bone metabolism. Because of the patient’s history of hepatitis B, her year of TDF treatment, and her symptoms of bone pain, nausea, vomiting, and muscle tenderness, we considered that the patient most likely had FS with bone pain, secondary to TDF treatment. After obtaining the patient’s consent for treatment, we replaced the TDF with entecavir dispersible tablets for antiviral therapy, and provided symptomatic treatment, including calcium, potassium, and phosphate supplementation. After 5 days, the patient reported no nausea or vomiting after eating, and her pain had improved.
Laboratory results upon admission.

indicates a high value.
*indicates a low value. TP, total protein; ALB, albumin; ALP, alkaline phosphatase; CG, glycine; PA, prealbumin; UA, uric acid; APOA1, apolipoprotein A1; HDL-C, high-density lipoprotein-cholesterol; LDH, lactic dehydrogenase; TCO2, carbon dioxide combining power; AMY, amylase; eGFR, estimated glomerular filtration rate; β-CTX, β-Crosslaps; P1NP, procollagen type IN-terminal propeptide; N-MID, N-MID osteocalcin; PTH, parathyroid hormone; BALP, bone alkaline phosphatase; 25(OH)D, 25-hydroxy-vitamin D; 24-h Ucr, 24-hour urine creatinine; KET, ketones; ACR, albumin/creatinine ratio; UOSM, urine osmolality; SG, specific gravity; A-dsDNA, anti-double-stranded DNA antibody; A-SM, anti-Smith antibody; ANA, anti-nuclear antibody; RNP, ribonucleoprotein; PCNA, proliferating cell nuclear antigen; CENPB, recombinant centromere protein B; A-TPO, antithyroid peroxidase antibody; A-TG, antithyroglobulin antibody; TG, thyroglobulin; TRAb, TSH receptor Antibody; LH, luteinizing hormone; FSH, follicular stimulating hormone; E2, estradiol; PRL, prolactin; TESTO, testosterone; PROG, progesterone; IgA, immunoglobin A; IgG, immunoglobin G; IgM, immunoglobin M; C3, complement 3; C4,complement 4; ASO, anti-streptolysin O; RF, rheumatoid factor; CRP, C-reactive protein; CER, ceruloplasmin; AFP, alpha fetoprotein; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; MPO-ANCA, anti-myeloperoxidase antibody; PR3-ANCA, anti-proteinase 3 antibody; cANCA, cytoplasm anti-neutrophil cytoplasmic antibody; pANCA, pronuclear anti-neutrophil cytoplasmic antibody; A-GBM, anti-glomerular basement membrane antibody.
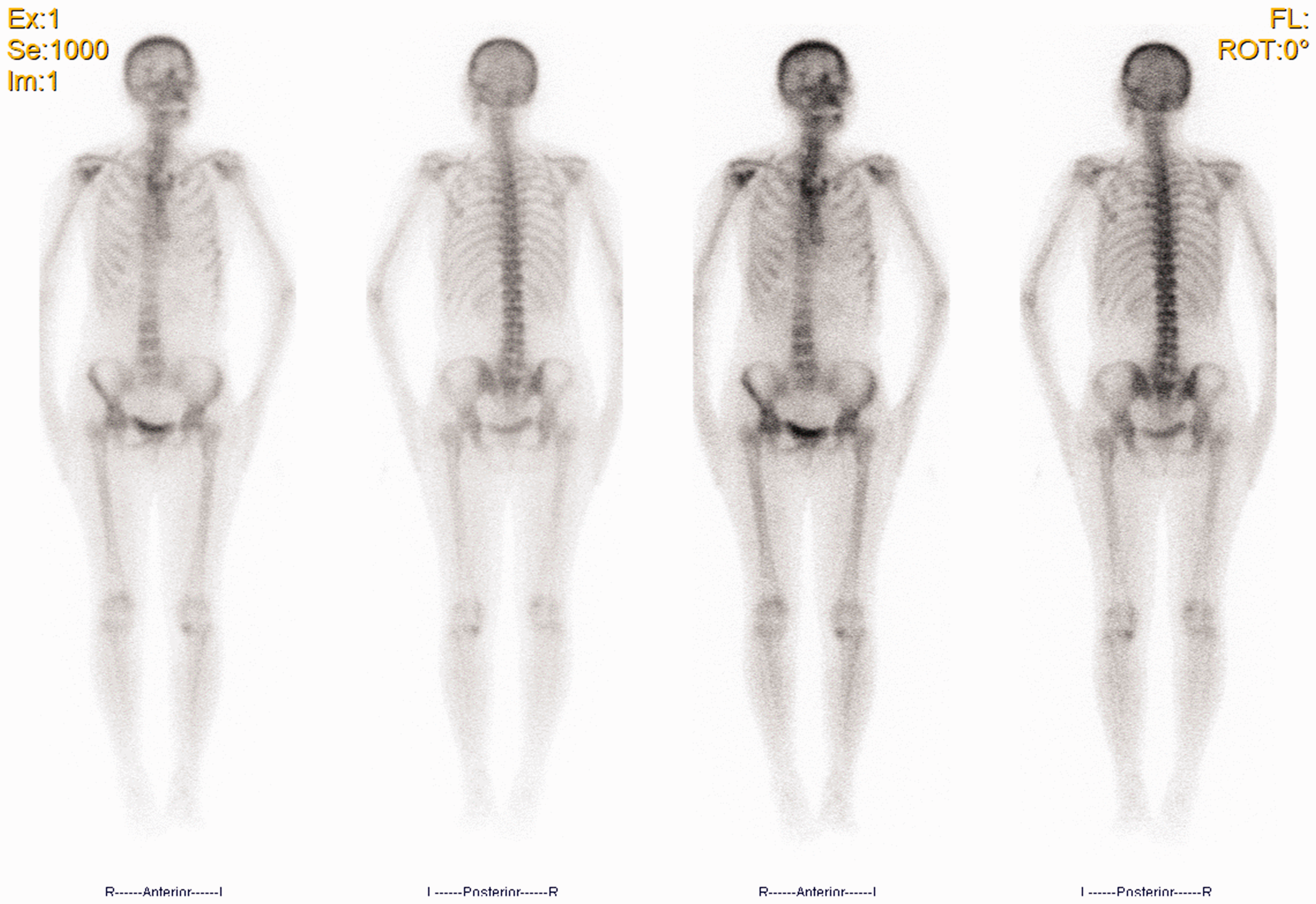
Whole-body emission computed tomography (bone) scan image, showing abnormal metabolic activity in the right shoulder joint and left sixth rib.
The patient was re-examined 10 days after discontinuing the medication (Table 2), when her circulating potassium concentration had slightly decreased and her circulating phosphate concentration had increased. In addition, her circulating chloride, calcium, and total CO2 concentrations had returned to normal, and she reported improvements in her symptoms of nausea, vomiting, bone pain, and muscle pain.
Laboratory data 10 days after the discontinuation of the medication.

indicates a high value;
*indicates a low value. TCO2, carbon dioxide combining power.
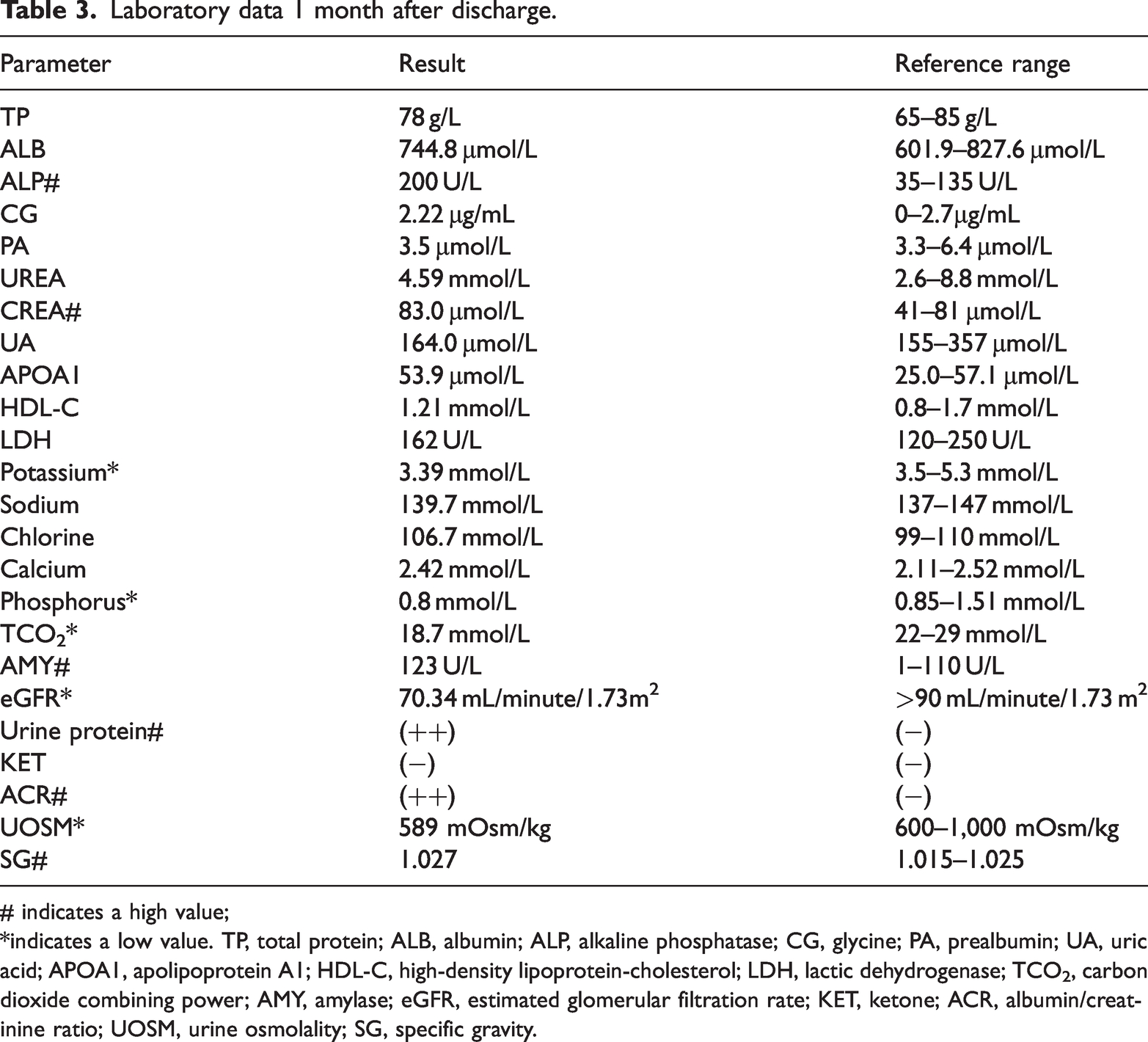
One-and-a-half months after the medication had been discontinued, the patient showed significant improvements in her circulating potassium and phosphorus concentrations, creatinine clearance rate, and urine osmotic pressure (Table 3). In addition, her urinary ketone body and circulating protein concentrations had normalized. However, her circulating ALP activity and urinary protein concentration remained high. Thus, the bone metabolism of the patient appeared not to have completely returned to normal. Moreover, the patient seemed to have developed drug-related kidney damage.
Laboratory data 1 month after discharge.
# indicates a high value;
*indicates a low value. TP, total protein; ALB, albumin; ALP, alkaline phosphatase; CG, glycine; PA, prealbumin; UA, uric acid; APOA1, apolipoprotein A1; HDL-C, high-density lipoprotein-cholesterol; LDH, lactic dehydrogenase; TCO2, carbon dioxide combining power; AMY, amylase; eGFR, estimated glomerular filtration rate; KET, ketone; ACR, albumin/creatinine ratio; UOSM, urine osmolality; SG, specific gravity.
Two-and-a-half months after the medication had been discontinued, the circulating phosphorus concentration of the patient had returned to normal and her potassium concentration had slightly decreased. In addition, her circulating ALP activity had decreased. However, her circulating creatinine concentration had further increased. The patient’s Ca2+ concentration had normalized, but bone density testing showed ongoing osteoporosis, suggesting that the bone metabolism of the patient had not fully returned to normal (Table 4).
Laboratory data 2 months after discharge.

indicates a high value;
*indicates a low value. ALP, alkaline phosphatase; CG, glycine; PA, prealbumin; UA, uric acid; TCO2, carbon dioxide combining power.
Discussion
FS is a disease characterized by a variety of genetically determined or acquired abnormalities of the proximal renal tubules. It involves impairments in the reabsorption of sodium, potassium, calcium, phosphate, glucose, water, amino acids, bicarbonate, uric acid, citrate, and low-molecular-weight proteins in the kidney. 16 The prolonged use of nucleoside analogs can cause renal tubular dysfunction, including FS,17,18 and although the mechanism remains unclear, the most likely mechanism is the inhibition of DNA synthesis in renal tubular cell mitochondria, leading to mitochondrial DNA loss in the proximal tubules. In addition, the large molecular weight of nucleoside analogs may predispose toward their accumulation in the renal tubules, disrupting the function of transporter proteins and ultimately resulting in proximal tubular dysfunction, which leads to a series of functional deficits.19,20
In total, reports of 27 cases of TDF-induced FS were published between 2013 and 2022.21–27 The ages of the patients ranged from 17 to 75 years and their duration of drug use ranged from 0.6 to 4 years. Laboratory testing revealed varying reductions in circulating bicarbonate, phosphorus (0.37 to 2.0 mmol/L), potassium (2.3 to 3.5 mmol/L), and calcium (0.44 to 0.5 mmol/L) concentrations, and ALP activity (288 to 1032 U/L). Ten percent of the patients had normal parathyroid hormone (PTH) and 25(OH)D concentrations, and almost all had proteinuria and diabetes. After a period of medication, some of the patients developed hyperglycemia, proteinuria, and high circulating creatinine concentrations. However, because TDF was not identified as the cause, the medication was continued, resulting in severe renal tubular acidosis. The electrolyte imbalance caused by FS-induced renal tubular acidosis can be ameliorated within 8 weeks. However, the recovery of renal function takes longer and the damage may persist for >5 years. In addition, nearly half of such patients develop permanent renal functional deficits. 28 Mild proteinuria following the discontinuation of TDF imply minor kidney damage and is a predictor of eventual recovery of renal function. 24 Ten weeks after the present patient had discontinued her medication, her circulating phosphorus and calcium concentrations had returned to normal, but her creatinine concentrations had increased, indicating that her TDF-induced renal functional impairment remained. The patient’s urine sample was positive for protein, which was quantified as 0.64 g/24 hours at admission, indicating mild proteinuria. Further follow-up will be essential to monitor her renal function. Of the previously studied 27 patients, eight actively sought hospital treatment, of whom six were admitted for the treatment of bone pain. Bone scans of all eight of these patients revealed osteoporosis, and some had experienced vertebral compression fractures, probably secondary to hypophosphatemia and parathyroid dysfunction. Laboratory testing revealed high ALP activities, but approximately 50% of these patients had normal PTH and 25(OH)D concentrations. The present patient presented with bone pain, showed high ALP activity and low 25(OH)D concentration, but a normal PTH concentration upon admission. These findings are consistent with those for previously reported cases. During the follow-up period, after she discontinued her medication, her Ca2+ concentration returned to normal. Although a bone scan showed ongoing osteoporosis, the patient’s severe bone pain had significantly improved. However, the patient had discontinued her medication relatively recently, and therefore further follow-up is required.
Because the understanding of FS among physicians is limited, misdiagnoses and missed diagnoses are common. Therefore, we discuss the clinical manifestations, diagnosis, and treatment of FS in detail here. The clinical manifestations of FS include bone pain at multiple sites, and especially in joints, and this is dull and persistent in character. Bone fracture, rickets, and growth retardation can also develop. 1 The laboratory findings are principally characterized by electrolyte imbalance, including low potassium, phosphorus, and calcium concentrations, and metabolic acidosis. Other abnormalities that may develop during FS include high urinary sodium concentration, low-molecular-weight proteinuria, renal glycosuria, aminoaciduria, phosphaturia, and urate excretion. The hypophosphatemia can be associated with bone demineralization because it affects the activation of vitamin D, resulting in relative calcitriol deficiency, which leads to osteomalacia. X-ray examination may reveal osteoporosis and skeletal deformities. 29 FS can also cause renal tubular acidosis, which in severe cases can progress to kidney failure. When a patient presents with compatible symptoms, the differential diagnoses include rheumatoid arthritis, other bone and joint diseases, and disease of the urinary system, along with FS. If a patient has a medical history of contact with certain medications or poisons, their electrolyte and acid-base imbalances can be corrected after discontinuing administration or exposure, thereby confirming a diagnosis of FS. The treatment of FS should involve the identification and treatment of the underlying cause, followed by symptomatic treatment, with the principal goal of preventing complications caused by the urinary loss of electrolytes. Salt consumption can lead to mineral loss from bones, but oral phosphate supplementation and a high-phosphate diet can help compensate for the urinary losses. 16 The bone disorders can be treated by the administration of vitamin D2, vitamin D3, or hydroxycholecalciferol. Dehydration, acidosis, and electrolyte imbalances may occur, and therefore adequate fluid intake is required from the commencement of treatment. Potassium citrate and sodium-containing fluids may be used to correct the hypokalemia, and potassium citrate is a more suitable choice than potassium chloride for patients with renal tubular acidosis, owing to its ability to increase urine pH. In addition, the administration of potassium ions may help relieve the polyuria. In patients with significant renal tubular acidosis, simultaneous potassium, sodium, and magnesium supplementation is necessary. 16 Drug-induced FS usually resolves spontaneously after the administration of the drug is discontinued; and the bone pain, muscle pain, electrolyte imbalances, including the low potassium and phosphate concentrations, and acidosis resolve without specific treatment. 30
The patients who were previously studied were middle-aged women whose osteoporosis was severe for their age. These women had a good quality of life, did not smoke or drink alcohol, had no long-term eating disorders, and had no lack of exposure to sunlight, which thus eliminates potential causes such as alcohol-induced and vitamin D deficiency-induced osteoporosis. Laboratory testing indicated high circulating ALP activity and low phosphate and potassium concentrations, which are not consistent with primary osteoporosis, multiple myeloma, metastatic bone disease, or hereditary osteogenesis imperfecta. Following a comprehensive evaluation of their clinical symptoms, laboratory data, and medical history (including 6 months of TDF use), and after ruling out other suspected causes, the women were diagnosed with FS. Their treatment consisted of discontinuing the TDF and administering symptomatic treatment to alleviate their symptoms. This approach significantly improved their symptoms and increased their circulating concentrations of electrolytes, such as phosphorus and potassium. Therefore, secondary FS and bone pain caused by TDF was diagnosed.
Conclusion
This case report reminds us that long-term TDF use can cause renal tubular damage, especially in patients with existing or potential risk of renal impairment. In severe cases, the condition can progress to FS. According to the European Liver Research Society, serum creatinine and phosphate concentrations should be monitored every 3 months during the first year of TDF treatment, and every 6 months thereafter to recognize such complications. For high-risk patients, the society recommends monthly monitoring during the first 3 months of TDF treatment, followed by 3-monthly examinations until the end of the first year, and 6-monthly examinations thereafter. 31
Footnotes
Acknowledgements
The authors would like to thank all the participants discussed in the review and PubMed for their support.
Author contributions
The authors accept responsibility for the entire content of the manuscript and approved its submission. Jun L and HG are responsible for the integrity of the work as a whole. Jiayi L, XZ, and HH organized the case files. XL, HG, and Jun L designed the study.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
The study was approved by the Ethics Committee of Xuzhou Central Hospital (approval number XZXY-LJ-20170217-008). We obtained verbal informed consent from the patient for the publication of their details. All the personal information is anonymized.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
