Abstract
Vinca alkaloid (VA)-induced ileus, a rare but severe autonomic neuropathy, can be enhanced by concomitant use of antifungal triazole agents. We herein present a case of VA-induced ileus in a 17-year-old girl who was diagnosed with B-cell acute lymphoblastic leukemia. On day 1, the patient received cyclophosphamide, vincristine, and methylprednisolone. On day 2, she began treatment with posaconazole oral suspension at 200 mg three times daily for prophylaxis against invasive fungal infection. On day 5, she began induction therapy consisting of vindesine, methylprednisolone, daunorubicin, and cyclophosphamide. The patient developed severe abdominal pain with marked constipation on day 11 and was diagnosed with incomplete ileus. After switching the antifungal agent to micafungin, performing gastrointestinal decompression, administering parenteral nutrition, and omitting the fourth dose of vindesine, the ileus symptoms were relieved. This case emphasizes the potential interaction between VAs and posaconazole. We also herein present a review of the literature on ileus caused by the combination of VAs and antifungal triazole agents. In clinical practice, physicians and pharmacists should be aware of the possibility of ileus caused by the use of VAs in combination with posaconazole. It is important to reduce complications during chemotherapy to improve patients’ prognosis.
Keywords
Introduction
B-Cell acute lymphoblastic leukemia (ALL) is a hematologic malignancy characterized by impaired differentiation, proliferation, and accumulation of lymphoid progenitor cells in the bone marrow (BM), peripheral blood, and other tissues, and it is the most common acute leukemia among children. 1 Combination chemotherapy is the front-line regimen for the treatment of ALL, with 5-year overall survival of approximately 90% in children and 30% to 40% in adults and elderly patients, respectively. 2 Vinca alkaloids (VAs) are the earliest developed microtubule-targeting agents. They are derived from the periwinkle plant and are widely used in the treatment of malignancies, most prominently leukemia and lymphoma. The VAs currently approved for clinical use are vincristine (VCR), vinblastine, vinorelbine, vindesine (VDS), and vinflunine. 3 Among them, VCR and VDS are important components of front-line therapy for ALL. 4
Neurotoxicity is a common dose-limiting adverse effect of VAs, and VCR is the most neurotoxic among all VAs. 5 VA-induced peripheral neuropathy can be classified into the following four categories: sensory neuropathy, motor neuropathy, autonomic neuropathy, and cranial neuropathy. One study indicated that more than one-third of patients treated with VCR may develop autonomic neuropathy manifesting as orthostatic hypotension, sexual dysfunction, abdominal pain, constipation, dysuria, and paralytic ileus. 6 Paralytic ileus is a rare but severe autonomic toxic effect of VAs that may lead to death in severe cases. 7
Antifungal triazole agents are widely used for the prophylaxis and treatment of invasive fungal infections, an important cause of substantial morbidity and mortality in patients with hematological malignancies. 8 Recent evidence has suggested that concomitant administration of antifungal triazole agents can enhance the risk of VCR-induced neuropathy, and ileus usually occurs when VCR is combined with itraconazole.9,10 Yasu et al. 11 investigated the incidence of and risk factors for VCR-induced ileus caused in Japanese adults with ALL. They found that concomitant use of itraconazole was significantly associated with VCR-induced ileus. 11 However, few reports have addressed ileus caused by the interaction of posaconazole with VAs. We herein report a recently observed case of ileus caused by coadministration of VAs and posaconazole.
Case report
A 17-year-old girl was admitted to the hematological unit of our hospital with a 2-week history of intermittent fever and general body aches. She had no history of gastrointestinal disease, diabetes, peripheral neuropathy, or other diseases. Abdominal computed tomography (CT) showed splenomegaly. A complete blood count revealed anemia (hemoglobin concentration, 65 g/L; reference range, 115–150 g/L) and thrombocytopenia (platelet count, 35 × 109/L; reference range, 125–350 × 109/L). Flow cytometry of the BM showed 52.38% abnormal B lymphoblasts among the nucleated cells, which was considered diagnostic for B-cell ALL (≥20% primitive/immature B lymphocytes in BM). BM aspiration and biopsy confirmed the diagnosis.
The patient was treated with one course of the COP regimen to reduce the leukemic cell burden; this regimen included cyclophosphamide (1 g) and VCR (2 mg) on day 1 and methylprednisolone (80 mg/day) from days 1 to 5. Antifungal prophylaxis with posaconazole oral suspension (200 mg three times daily) was started on day 2. On day 5, the patient’s general body aches had improved, and she received induction therapy consisting of VDS 4 mg (days 5, 12, 19, and 26), methylprednisolone 60 mg/day (days 5 to 32), daunorubicin 60 mg (day 5) and 40 mg (days 6 to 7), and cyclophosphamide 1 g (day 5). On day 11, the patient developed severe abdominal pain with marked constipation and received a glycerol enema. Her laboratory results were as follows: white blood cell count, 0.52 × 109/L (reference range, 3.50–9.50 × 109/L); absolute neutrophil count, 0.03 × 109/L (reference range, 1.80–6.30 × 109/L); hemoglobin, 66 g/L; and platelet count, 26 × 109/L. An abdominal CT scan showed excessive intestinal gas (Figure 1). Incomplete ileus was confirmed clinically as well as radiologically, coinciding with the clinical manifestations of severe VA-induced neurotoxicity. The posaconazole was switched to intravenous caspofungin (loading dose, 70 mg; maintenance dosage, 50 mg/day). During the next 22 days, the patient was treated by fasting, gastrointestinal decompression, parenteral nutrition, and Da-Cheng-Qi Decoction (a traditional Chinese medicine decoction used for enema catharsis). She also continued to receive intravenous broad-spectrum antibiotics because of neutropenia. Flow cytometry of the BM showed 0.43% abnormal B lymphoblasts among the nucleated cells on day 19. The fourth dose of VDS was omitted on day 26. The patient’s abdominal discomfort improved after a few days. On day 31, 4.73% abnormal primitive B lymphocytes were observed by flow cytometry. Abdominal CT showed more severe splenomegaly than before. Considering the possibility of primary disease progression, the patient volunteered to participate in a clinical trial of chimeric antigen receptor T-cell immunotherapy. The patient was found to have a score of 5 (probable) on the Drug Interaction Probability Scale 12 (Table 1). Consequently, a drug–drug interaction between the VAs and posaconazole was likely to have caused the incomplete ileus. A timeline of the treatment-related events is shown in Figure 2.

Abdominal computed tomography scan showing excessive intestinal gas.
Calculation of Drug Interaction Probability Scale score in present case.
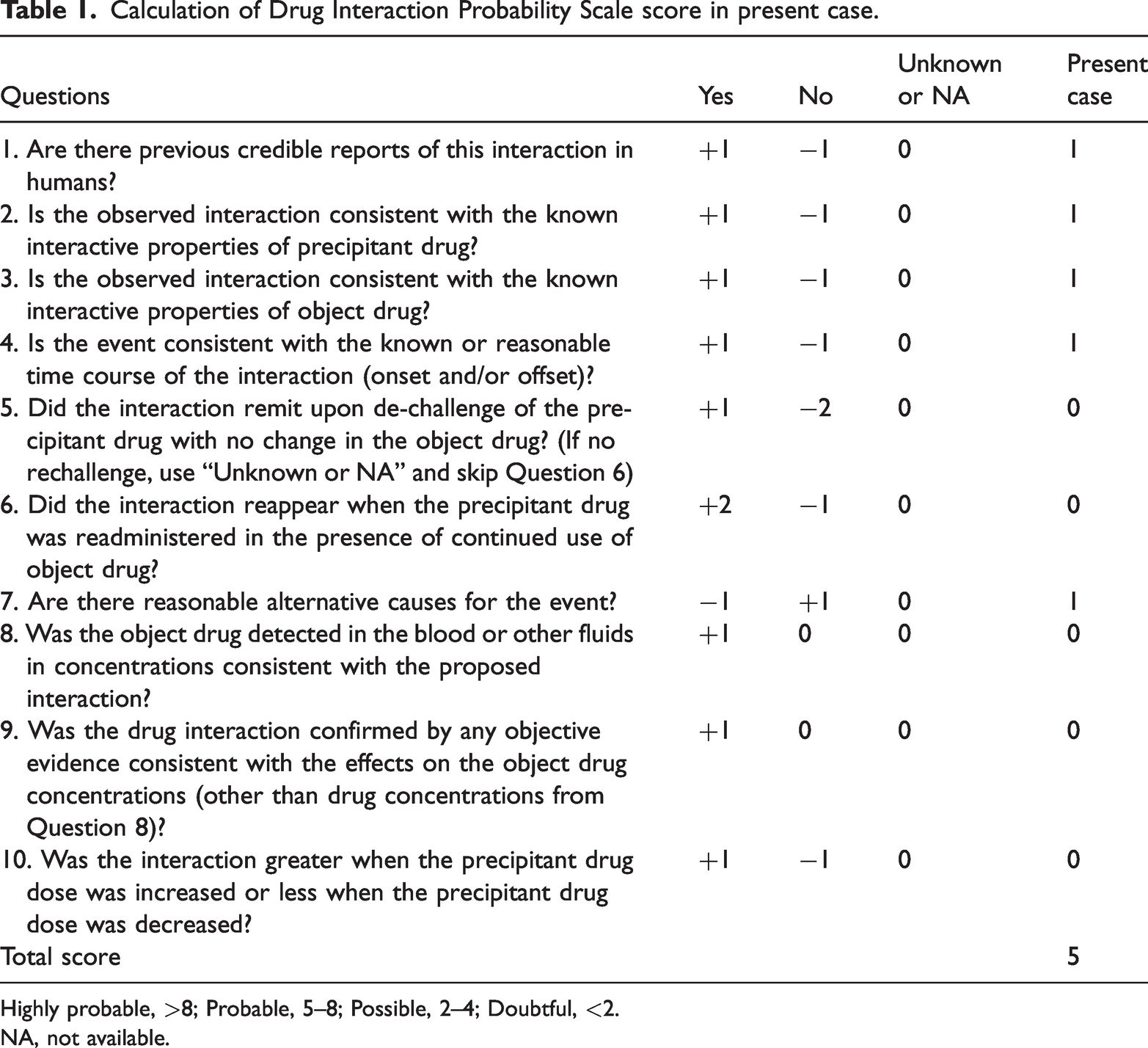
Highly probable, >8; Probable, 5–8; Possible, 2–4; Doubtful, <2.
NA, not available.

Clinical timeline of major interventions.
The reporting of this study conformed to the CARE guidelines, 13 and we obtained the patient’s written consent for treatment. All procedures performed in this study involving human participants were in accordance with the Declaration of Helsinki (as revised in 2013). Although written consent for publication was not obtained, we have de-identified the details such that the identity of the patient may not be ascertained in any way. The requirement for ethics board approval was waived because this was a case report.
Literature review
We searched the PubMed database for available case reports of ileus caused by the combination of VAs and antifungal triazole agents from January 1970 to January 2021. The keywords used were “vinca alkaloids or vincristine or vinblastine or vindesine or vinorelbine” and “antifungal triazole agents or fluconazole or voriconazole or itraconazole or posaconazole” and “ileus or intestinal obstruction.” Overall, 13 publications involving 25 patients were found. We extracted the following details from each article: first author, publication date, number of patients, age, sex, diagnosis, doses of VAs and antifungal triazole agents, day of starting triazole treatment (number of days from onset of VA therapy), interval to reaction (number of days to first appearance of ileus from start of VA therapy), treatment, and outcome.
Discussion
Paralytic ileus has been associated with VA-induced autonomic neuropathy. Paralytic ileus is defined as the temporary cessation of propulsive contraction of the gastrointestinal tract followed by intestinal dilation and accumulation of secretions and gas in the lumen. Clinical manifestations of paralytic ileus include abdominal distension and discomfort, bloating, belching, nausea, vomiting and constipation, and the condition can be fatal in severe cases. 14 This case report describes a patient who was diagnosed with ileus due to the interaction of VAs with posaconazole.
Since Bohme et al. 15 reported the first cases of paralytic ileus associated with VCR and itraconazole in four patients with ALL, there have been 15 additional cases of ileus due to VCR and itraconazole, 3 cases due to VCR and posaconazole, 2 cases due to VDS and itraconazole, and 1 case due to VCR and fluconazole published in PubMed15–27 (Table 2). The sex of 24 of these 25 patients was reported, and they comprised 14 male and 10 female patients. Their median age was 16 years (range, 1.25–68 years). Compared with adolescents and adults, young children showed better tolerance to relatively higher doses of VCR and developed milder neuropathy. 28 A study of adolescents aged ≤18 years suggested that age of ≥10 years is an independent risk factor for the development of VCR-associated toxicity. 29 The patient in our study was 17 years old, and she appeared to be at higher risk of neurotoxicity according to these findings. Aside from age, other suggested predictors of VCR-induced neuropathy include White non-Hispanic race, lymphoma, diabetes, Charcot–Marie–Tooth disease, the number of chemotherapy cycles, a higher single dose of VCR, concomitant azole antifungal therapy, and homozygosity for CYP3A5*3.10,28
Review of ileus caused by coadministration of vinca alkaloids and antifungal triazole agents.

Number of days from onset of vinca alkaloid therapy.
Number of days to first appearance of ileus from start of vinca alkaloid therapy.
VAs, vinca alkaloids; VCR, vincristine; VDS, vindesine; ALL, acute lymphoblastic leukemia; DLBCL, diffuse large B-cell lymphoma; M, male; F, female; d, day.
VCR was administered at weekly intervals at a dose of 1.5 mg/m2 or 2 mg in these previously reported cases. Ileus developed after a median of three doses (range, 2–4 doses) of VCR (the cumulative total dose of VCR was 3 mg/m2 in three patients, 3.48 mg/m2 in two patients, 4 mg in four patients, 4.5 mg/m2 in six patients, 6 mg in two patients, and 8 mg in one patient). Two of 25 patients developed symptoms of ileus after one and two doses of VDS, respectively. One of the patients was a 20-year-old man with ALL who had received itraconazole solution on day 2, and VDS was used on days 4 and 11. Nine days after itraconazole was started, the patient developed symptoms of ileus including abdominal pain, abdominal bloating, cramps, and inability to pass flatus or stool. The other patient was a 37-year-old woman with ALL who was treated with VDS on days 1, 8, and 15. Itraconazole injection was initiated on day 9. Five days after itraconazole was started, the patient developed symptoms of ileus. 22 The patient in our study developed ileus on day 11 after one dose of VCR (2 mg) on day 1 and one dose of VDS (4 mg) on day 5. Posaconazole oral suspension was started on day 2. Thus, it was difficult to determine whether the ileus was caused by VCR or VDS. The most appropriate explanation is that the occurrence of ileus was a result of the combined effect of VCR and VDS. Additionally, the median time to clinical manifestations of ileus due to the VA–azole interaction was 10.5 days (range, 2–28 days). Pekpak et al. 27 described a patient who developed ileus symptoms 7 days after receiving the combination of VCR and posaconazole, whereas this time interval was 10 days in our case. The greatest impact of the VA–posaconazole interaction may occur when the posaconazole drug level reaches a steady state, which is 7 to 10 days after initiation of posaconazole oral suspension. 30 VCR-induced neuropathy can be temporary and reversible, disappearing a few months after discontinuation of VCR in most cases. 31 Among the previously reported cases, the symptoms of ileus were relieved in all but one patient. Importantly, VAs are key drugs in the treatment of ALL, and close attention should be paid to the disease course when completely avoiding the use of VAs.
In our literature review, 21 of 25 patients received itraconazole. The wider literature suggests that itraconazole is more likely to interact with VAs, possibly because it more potently inhibits the CYP3A4 enzyme than do posaconazole and voriconazole. 32 Notably, several cases of ileus caused by coadministration of VAs and posaconazole were observed in recent years.24,26,27 Alavi and Ebadi 24 reported that an 8-year-old boy with T-cell ALL was diagnosed with paralytic ileus after receiving combined treatment with oral posaconazole and VCR. Pekpak et al. 27 described a 14-year-old girl with B-cell ALL who developed abdominal pain and constipation after coadministration of VCR and oral posaconazole. Castells et al. 26 reported a 49-year-old man with ALL who developed paralytic ileus mediated by voriconazole, followed by oral posaconazole alongside VCR. Posaconazole is a second-generation triazole agent with the broadest antifungal spectrum among antifungal triazole agents. It was derived from itraconazole and has the same antifungal mechanism of action as other azole derivatives (i.e., inhibition of lanosterol 14-α-demethylase). 33 Posaconazole oral suspension was first approved by the U.S. Food and Drug Administration in 2006 for prophylaxis against Aspergillus and Candida infections in immunocompromised patients aged ≥13 years at high risk of infection. 34 Compared with most triazole antifungal agents, posaconazole reportedly has advantages of a reduced incidence of invasive fungal infections, lower all-cause mortality, and good cost-effectiveness. 35
The potential interactions between VAs and antifungal triazole agents have been widely reported in recent years. VAs are cell cycle-specific anticancer drugs that exert their antitumor efficiency by inhibiting polymerization of tubulin and incorporation into microtubules, thereby disrupting the mitotic spindle apparatus and directly causing metaphase arrest and apoptosis. 36 The hepatic cytochrome P450 3A (CYP3A) enzyme system, particularly the CYP3A4 and CYP3A5 enzymes, are mainly responsible for the metabolism of VAs. Several membrane transporters, including ABCB1 (P-glycoprotein (P-gp)) and ABCC2 (which mediates bile excretion), also play relevant roles in the transport and clearance of VAs, making VAs susceptible to drug–drug interactions.37–39 Itraconazole and posaconazole are potent inhibitors of CYP3A4. They also inhibit the P-gp efflux pump, which contributes to the increased risk of VA-induced ileus. 32 P-gp is highly expressed on the apical surface of the epithelial cells of the intestine, hepatic bile duct, and proximal tubule of the kidney. The P-gp expressed on intestinal epithelial cells is responsible for efflux, limiting cellular uptake and absorption into intestinal cells, whereas the P-gp expressed on the surface of tubules of hepatocytes and renal tubular cells enhances the elimination of drugs into bile and urine. 40 VAs are mainly eliminated by the hepatobiliary system. When the CYP3A4 enzyme in the liver and the P-gp expressed on the epithelial cells of the hepatic bile duct are inhibited by itraconazole or posaconazole, the metabolism and elimination of VAs slows down, leading to increased exposure of VAs and a higher risk of neurotoxicity. A phase II study conducted by the Japan Adult Leukemia Study Group showed a 3.6% incidence of ileus in patients treated with VCR. 41 However, when VCR was combined with antifungal triazole agents, the incidence of ileus reached 32%. 11 Thus, the inhibitory effect on P-gp might explain why itraconazole and posaconazole are more likely to increase the risk of VA-induced ileus than other triazole agents that do not inhibit P-gp (e.g., fluconazole and voriconazole). In addition, the increased exposure of VAs and risk of neurotoxicity were associated with the route of administration of antifungal triazole agents. The CYP3A4 enzyme is also present in the intestinal mucosa and is therefore involved in the first-pass metabolism of many drugs. Xenobiotics are reportedly capable of increasing or decreasing the enzymatic activity of CYP3A4, which can independently affect both liver and intestinal enzymes. This suggests that intravenous administration may have an inhibitory effect on hepatic CYP3A4, whereas oral administration may produce dual inhibition of both hepatic CYP3A4 and intestinal CYP3A4. In the present case, the patient was treated with posaconazole oral suspension, which inhibited both liver and intestinal enzymes, resulting in the increased exposure of VAs and risk of neurotoxicity. 42
Clinical evidence has shown marked individual differences in VCR metabolism. Wu et al. 43 suggested that the traditional method of basing administration of the body surface area is prone to insufficient exposure of VCR or severe VCR-induced peripheral neuropathy, and they summarized various supportive evidence for the suitability of applying therapeutic drug monitoring (TDM) to VCR therapy. Barnett et al. 44 found that all patients participating in TDM dosing were able to achieve drug exposures equivalent to the areas under the curve observed in older pediatric patients at doses that were well tolerated and subsequently used for the remaining cycles, indicating the usefulness of TDM in neonates and infants. Additionally, posaconazole oral suspension showed significantly variable bioavailability because of its erratic absorption, which was influenced by food, certain medications, and gastric absorption disorders. 45 Higher blood levels might worsen the interaction of posaconazole with VCR and cause more severe clinical symptoms. Therefore, optimizing the dosages of VCR and posaconazole oral suspension using TDM might be of great clinical significance for the management of drug–drug interactions. Furthermore, accumulating evidence has identified the relationship between genetic germline variants and VCR-induced peripheral neuropathy. 46 Egbelakin et al. 47 found that children homozygous for CYP3A5*3 experienced an increased incidence and severity of VCR-induced peripheral neuropathy. Diouf et al. 48 found that an inherited polymorphism in the promoter region of the CEP72 gene was associated with an increased incidence and severity of VCR-induced peripheral neuropathy in children with ALL. This suggests that the combination of pharmacogenomics and TDM is a more reliable means of predicting VA-induced peripheral neuropathy when VAs are combined with itraconazole or posaconazole. 43 Unfortunately, gene polymorphisms were not investigated and the VCR plasma concentration was not monitored in our patient.
Conclusion
We have herein reported a case of ileus due to the combination of VAs and posaconazole and reviewed cases of ileus caused by the interaction of VAs with antifungal triazole agents. We found that the risk of ileus is much greater when VAs are used in combination with itraconazole or posaconazole, necessitating close attention when these drugs are used in the clinical setting.
Footnotes
Author contributions
Yue Li, Ming-Feng Zhao, Xia Xiao, and Xiao-Chen Wei collected the patient’s data from the Department of Hematology. Ming-Feng Zhao and Xia Xiao followed and treated the patient. Yue Li and Xiao-Chen Wei contributed to the data analysis and manuscript writing. Xiao-Chen Wei and Yin-Hua Gong helped perform the data analysis and chart design and engaged in constructive discussions. All authors read and approved the final manuscript.
Data availability statement
All data generated or analyzed during this study are included in this published article and are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
