Abstract
Objective
Hepatitis E virus (HEV) infections are common, self-limiting causes of acute viral hepatitis. This study aimed to analyze hepatic injury, viremia, and chronicity rates in patients with acute HEV infection receiving immunosuppressive (IS) therapy taking into account ribavirin treatment.
Methods
In this retrospective, single-center, observational study, we analyzed the disease course of 25 non-cirrhotic patients receiving IS therapy who were diagnosed with acute HEV viremia. Forty-four patients with acute HEV viremia without IS therapy were controls.
Results
Demographics, symptoms at presentation, and extrahepatic manifestations were not different between patients with and without IS therapy, but liver injury at presentation was less severe in patients with IS therapy. Among the patients with IS therapy, 18 (72%) received ribavirin for a median of 56 days. Sustained viral clearance was observed in 21 patients with IS therapy, whereas 3 patients relapsed after ribavirin, and 1 patient had viral persistence. Among patients with sustained viral clearance, there was a longer duration of viremia in patients with IS therapy than in those without.
Conclusions
In this cohort of non-cirrhotic patient with IS, early treatment with ribavirin for acute HEV infection did not improve viral clearance rates, but may have shortened the duration of viremia.
Introduction
Hepatitis E virus (HEV), a 27- to 34-nm-sized RNA virus from the family of Hepeviridae, is currently the most common cause of acute viral hepatitis worldwide.1,2 HEV was first described as an enterally-transmitted non-A non-B hepatitis virus following outbreaks in the early 1980s. 3 However, HEV, particularly genotype 3, has now become endemic in Europe, North Asia, Australia, and the Americas.4,5 Consumption of raw or undercooked meat, offal, and meat products or shellfish products, and occupational contact with pigs or sewage are considered relevant risk factors for autochthonous HEV infection.6,7
The reported seroprevalence of HEV in Europe ranges from 8% to 23%, and is the highest in southern France.8,9 In Germany, the prevalence of HEV is estimated at approximately 17%, and it exceeds 25% in hexagenerians.10,11 Approximately 1 in 3800 blood donors have HEV viremia, 12 and this prevalence ranges from 1:600 in the Netherlands 13 to 1:14,500 in Scotland. 14
Acute HEV infection is often asymptomatic or self-limiting. However, the prevalence of HE infection makes it one of the most common causes of severe liver injury, accounting for up to 15% of patients with transaminase concentrations ≥1000 U/mL. 15 In patients with hepatitis B virus-related cirrhosis, acute HEV infection increases mortality by five fold. 16 In addition, extrahepatic complications, such as neuralgic amyotrophy, Guillain–Barré syndrome, glomerulonephritis, pancreatitis, thyroiditis, myocarditis, and lymphoma, have been reported.17–19 In immunocompromised patients after solid organ transplantation (SOT) 20 or with hematological malignancies, 21 infections with HEV genotypes 3 and 4, as well as infections with rat HEV, can lead to chronic infection with considerable morbidity and liver-related mortality.
Guidelines published in 201822,23 recommend ribavirin treatment in patients with persisting HEV replication 3 months after detecting HEV RNA for at least 3 months. The recommendation of this delayed treatment is based on the observation that spontaneous HEV clearance is predominantly observed, although spontaneous clearance between 3 and 6 months has also been observed.24,25 Ribavirin treatment may also be considered in patients with severe or complicated acute HEV, such as patients who develop severe liver dysfunction, acute-on-chronic liver failure, or extra-hepatic manifestations, although the currently available evidence is limited. Retrospective cohort studies and case series have shown that ribavirin is safe and effective for the treatment of HEV in patients receiving immunosuppressive (IS) therapy, even in the acute phase of this disease. 26
A recent retrospective, multicenter cohort study of 50 patients with IS therapy for hematological malignancies recently showed lower mortality in patients with HEV infection when ribavirin was initiated within 24 weeks after diagnosing HEV infection than non-treated patients. 21 However, earlier treatment within the first 12 weeks did not result in a significantly improved mortality. Data on early short-term treatment with ribavirin for acute HEV infection in patients with IS therapy are sparse. 23 Therefore, this study aimed to identify non-cirrhotic patients with IS therapy who were treated for acute HEV infection at our tertiary center to analyze hepatic injury, viremia, and chronicity rates.
Methods
Study population
In this retrospective cohort study, all patients with a positive serum HEV RNA nucleic acid test who were treated at Jena University Hospital between March 2014 and October 2021 were retrospectively identified via our clinical data system. Age, sex, concomitant diseases, immunosuppressants and other concomitant medications, symptoms, cause of infection, travel before hospitalization, and blood transfusions were determined from routine documentation. Immunosuppression was defined as the use of glucocorticoids (≥5 mg prednisolone or equivalent/day), calcineurin inhibitors, mycophenolate mofetil, thiopurines, biologicals, anti-metabolites, or cytostatic chemotherapeutic agents. Laboratory parameters used to assess liver function and the disease course were alanine transaminase (ALT), total bilirubin, the international normalized ratio, HEV RNA, anti-HEV immunoglobulin M, and liver autoantibodies. Patients with pre-existing advanced liver disease at the diagnosis of HEV infection as defined by the medical history and imaging and histological results were excluded from the analysis. In addition, patients with chronic HEV infection (i.e., patients with persistent viremia for longer than 3–6 months)22,25 or likely chronic HEV infection (i.e., patients with prior positive anti-HEV antibodies or with elevated transaminase concentrations >90 days before inclusion) were excluded. Missing data were imputed by the last observation carried forward method.
The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Ethics Committee of Friedrich Schiller University Jena at the Faculty of Medicine (number: 2020-1900-Daten). The need for written informed consent was waived owing to the retrospective nature of the study and the exclusive use of routine data only. The reporting of this study conforms to the STROBE guidelines. 27
IS therapy
The reasons for performing IS therapy were SOT in 13 patients (10 kidney, 1 lung, 1 heart and lung, and 1 liver), hematopoietic stem-cell transplantation (HSCT) in 1 patient, cancer in 2 patients (lung cancer, glioblastoma), hematological malignancies in 4 patients (3 with B cell chronic lymphocytic leukemia and 1 with follicular lymphoma), and immune-mediated diseases (membranoproliferative glomerulonephritis, myasthenia gravis, rheumatoid arthritis, tuberous sclerosis, and CREST syndrome). After SOT or HSCT, the patients received triple IS therapy with calcineurin inhibitors, mycophenolate mofetil, and corticosteroids (n = 10) or dual IS therapy with calcineurin inhibitors and mycophenolate mofetil (n = 2), a calcineurin inhibitor and corticosteroids (n = 1), or an mTOR inhibitor and corticosteroids (n = 1). Patients who received IS therapy for other indications received rituximab alone (n = 2) or in combination (n = 2), everolimus (n = 1), mycophenolate (n = 1), azathioprine plus corticosteroids (n = 1), or methotrexate plus leflunomide (n = 1). Additionally, some patients received antineoplastic therapy with vinorelbine (n = 1), temozolomide (n = 1), lomustine and corticosteroids (n = 1), or the tyrosine kinase inhibitor ibrutinib (n = 1) (Supplementary Table 1).
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, versions 26 and 29 (IBM Corp., Armonk, NY, USA), and the visualization of data was performed using Prism 8 (GraphPad, La Jolla, CA, USA). Statistical differences were evaluated with the nonparametric Mann–Whitney U test for the comparison of continuous data or Fisher's exact test for discrete variables as appropriate. To identify predictors of sustained viral clearance, univariate and multivariable logistic regression analyses were used. Tests at a significance level of p < 0.05 in two-sided tests were considered significant.
Results
Clinical presentation of patients with acute HEV infection
Sixty-nine patients without pre-existing cirrhosis who were diagnosed with acute HEV viremia were identified in the study period. Reverse transcription-polymerase chain reaction (RT-PCR) to identify HEV was performed at the first or second visit as an outpatient (n = 10) or as part of the diagnostic work-up as an inpatient (n = 59). The majority of patients were men, and the median age was 59 years. Twenty-five (36.2%) patients received IS therapy (Table 1).
Presentation, clinical course, and outcomes of patients with acute HEV viremia.
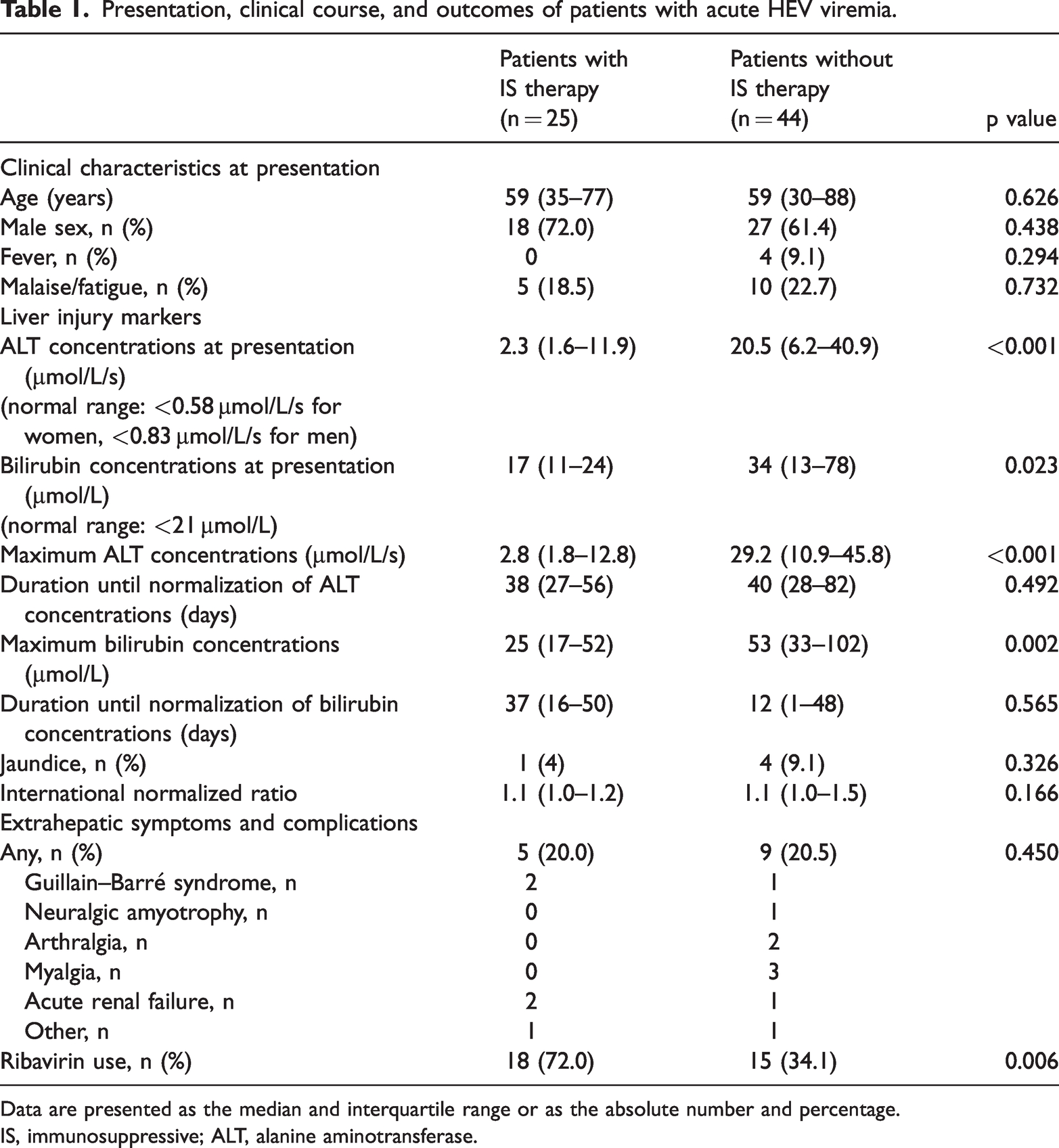
Data are presented as the median and interquartile range or as the absolute number and percentage.
IS, immunosuppressive; ALT, alanine aminotransferase.
All patients with acute HEV infection had elevated ALT concentrations, indicating hepatocellular damage, at inclusion in the study. ALT concentrations at presentation and the maximum ALT concentration were significantly lower in patients with IS therapy than in those without IS therapy (both p < 0.001). In addition, total serum bilirubin concentrations at the diagnosis were lower in patients with IS therapy than in those without IS therapy (p = 0.023). Clinical characteristics and complications, including neurological complications, were not different between patients with and without IS therapy (Table 1).
Treatment of acute HEV infection in patients receiving IS therapy
The median follow-up in patients with acute HEV infection and IS therapy was 655 days. One patient was lost to follow-up 17 days after the diagnosis, and one patient died 577 days after the diagnosis.
In 18 (72%) of the 25 patients with IS therapy, ribavirin was initiated on the basis of the decision by the treating physician. In these patients, the median time from diagnosis of viremia to initiating therapy was 8 days (range: 2–61 days), and 94% patients were treated within the first 21 days after the diagnosis (Figure 1). Patients who received ribavirin had lower bilirubin concentrations at presentation than those who did not receive ribavirin (p = 0.012), but other biochemical or demographic parameters were not different between the groups of patients (Table 2). The median ribavirin dose was 600 mg/day. One patient received combination therapy with ribavirin and sofosbuvir (400 mg each).

Schematic representation of viremia, liver injury, ribavirin treatment, and virological outcome of 25 patients with immunosuppression and acute hepatitis E virus infection.
Baseline characteristics of patients with acute hepatitis E virus viremia who received IS medication stratified for ribavirin use.
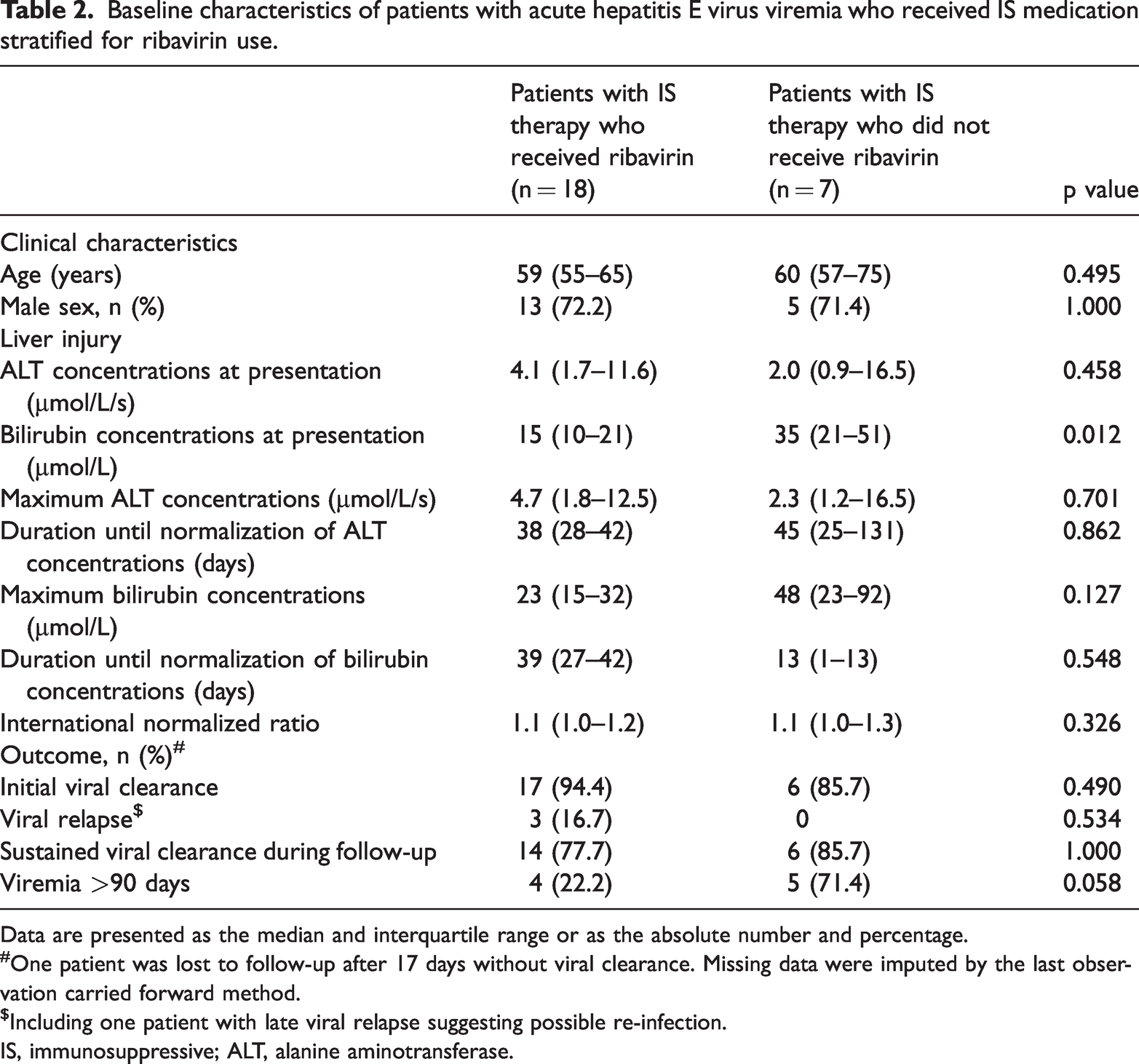
Data are presented as the median and interquartile range or as the absolute number and percentage.
One patient was lost to follow-up after 17 days without viral clearance. Missing data were imputed by the last observation carried forward method.
Including one patient with late viral relapse suggesting possible re-infection.
IS, immunosuppressive; ALT, alanine aminotransferase.
Overall, the initial viral clearance was documented in 23 (92%) patients without any difference in ribavirin use. In 11 patients, viral clearance from serum was confirmed during treatment after a median of 47 days of therapy with ribavirin (range: 23–84 days). In six patients, ribavirin therapy was stopped before viral clearance was achieved, which was after a median of 36 days (range: 14–70 days). One patient who was treated with ibrutinib failed to show HEV clearance, but ribavirin was stopped after 48 days because of side effects, and HEV-RNA was still detectable at the last contact 222 days after the discontinuation of ribavirin (Figure 1).
Six of seven (86%) patients who did not receive ribavirin achieved viral clearance owing to the modulation of immunosuppression. However, one patient who did not have HEV infection resolved was lost to-follow up 17 days after the first diagnosis and thus could not be assessed adequately. In patients who were not treated with ribavirin, the median time from presentation to the normalization of ALT concentrations was 61 days.
In all patients, ALT concentrations returned to normal values after viral clearance. Hepatic decompensation with ascites or hepatic encephalopathy was not observed in patients with acute HEV infection who received IS therapy.
Three ribavirin-treated patients suffered from a relapse of their HEV infection, including two kidney transplant recipients and a patient with glomerulonephritis during follow-up (Table 2). The first documentation of a positive RT-PCR result was made 90, 110, and 609 days after the first negative RT-PCR result in these three patients. Re-infection with HEV was a possible differential diagnosis in one patient with a long interval of a positive result. In all three patients, ribavirin was stopped before the first clearance of HEV RNA (3, 30, and 100 days before clearance) from serum.
There was a trend toward a lower number of patients with viremia >90 days in ribavirin-treated versus untreated patients (22% vs. 71%; p = 0.058). When only patients who achieved a long-term viral response were considered, a duration of viremia >90 days was observed in 22.3% with ribavirin treatment compared with 80.0% without ribavirin treatment (p = 0.049) (Table 3).
Patients using IS therapy with and without relapse of failure of viral clearance.

Data are presented as the median and interquartile range or as the absolute number and percentage.
Indicators for sustained viral clearance
Patients who failed to achieve long-term clearance of HEV infection tended to be younger than patients with successful clearance, but this was not significant (58 vs. 61 years; p = 0.097). Despite only a slight difference in the maximum ALT concentration between patients with and without long-term clearance, higher maximum ALT concentrations were significantly correlated with a shorter duration of viremia (Spearman’s Rho: −0.492, p = 0.012), and a delay in treatment was associated with a prolonged viremia (Spearman’s Rho: 0.589; p = 0.010).
A reduction in IS medication was heterogenous because it was based on the decision of the treating physician. Overall, in the first 4 weeks after the diagnosis of HEV, the IS medication remained unchanged in 11 patients. Additionally, in three patients who received rituximab, there was no scheduled dose in these 4 weeks. In 11 patients, the IS agents were reduced. Mycophenolate mofetil was reduced in seven patients (6 kidney transplant recipients and 1 patient with autoimmune disease) and completely stopped in another kidney transplant recipient. In one patient, azathioprine was stopped, while prednisolone remained unchanged. However, in two patients (1 patient with everolimus and 1 patient with methotrexate and leflunomide), the IS therapy was completely stopped. Tacrolimus and prednisolone doses remained largely unaltered in all patients. A reduction in IS therapy was associated with a nonsignificant change in the odds ratio for sustained virological clearance, which was most likely due to the small number of patients who did not achieve it (Table 4).
Predictors of achieving sustained virological clearance* in patients receiving IS therapy.
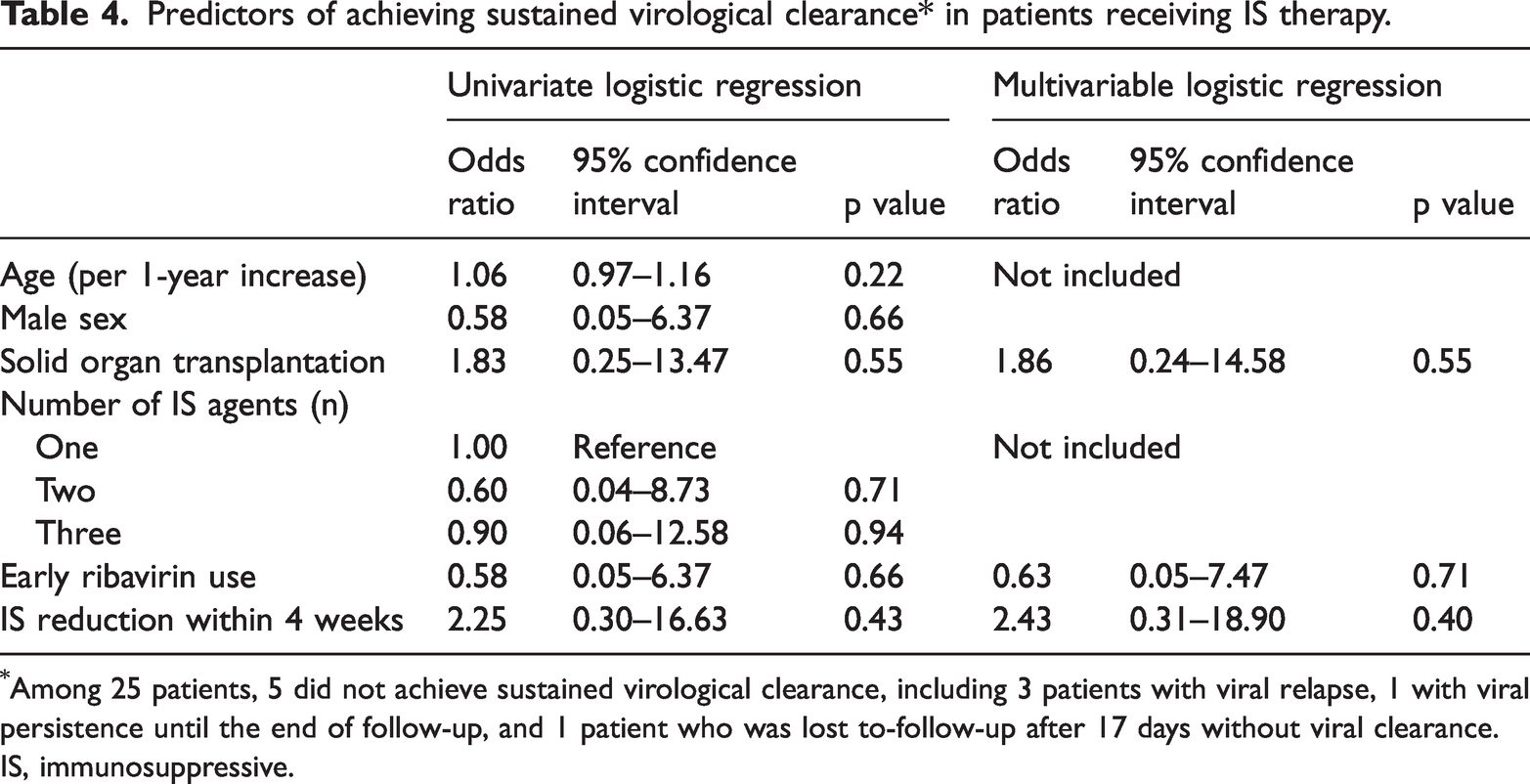
Among 25 patients, 5 did not achieve sustained virological clearance, including 3 patients with viral relapse, 1 with viral persistence until the end of follow-up, and 1 patient who was lost to-follow-up after 17 days without viral clearance.
IS, immunosuppressive.
The odds ratio for achieving sustained viral clearance with early ribavirin use was 0.58 (95% confidence interval: 0.05–6.37) in the univariate logistic regression analysis (Table 4). After adjustment for SOT as a cause of IS and for whether IS therapy was reduced within the first 4 weeks after the diagnosis, the odds ratio was 0.63 (95% confidence interval 0.05–7.47) in the multivariable logistic regression analysis (Table 4).
Ribavirin use for acute HEV infection in patients who did not receive IS medication
Seventeen of 44 (38.6%) patients who did not receive IS therapy received ribavirin for the treatment of HEV. Ribavirin was started after a median of 3 days after diagnosing viremia (range: 0–59 days) on the basis of the decision of the treating physician. One patient without IS therapy developed ascites owing to the acute HEV viremia, and no patients suffered from hepatic encephalopathy.
The median duration of ribavirin treatment was shorter in patients without IS therapy than in patients with IS therapy (42 vs. 56 days), but this difference was not significant (p = 0.220). Treatment with ribavirin in patients without IS therapy did not result in a quicker normalization of ALT concentrations (39 days in treated patients vs. 41 days in untreated patients, p = 0.560) or quicker clearance of HEV RNA (41 days in treated patients vs. 38 days in untreated patients, p = 0.997).
One patient with chronic lymphatic leukemia who received the B cell depleting agent rituximab 6 years before presentation and the phosphoinositide 3-kinase inhibitor idelalisib until 4 months before the diagnosis was treated with ribavirin and sofosbuvir because of severe hepatitis. This patient cleared HEV 30 days after the diagnosis, but relapsed 112 days later. According to the study protocol, this patient was classified as being without IS therapy.
Discussion
To date, there is controversy regarding which patients with HEV infection should receive antiviral treatment with ribavirin. While ribavirin therapy is recommended for immunocompromised patients with persistent HEV infection >3 months, whether earlier treatment provides a benefit is controversial.22,23 Treatment of acute HEV has been advocated and performed in patients with underlying cirrhosis, especially in hepatic decompensation.15,28 Treatment of acute HEV is also recommended in patients with severe or potentially severe hepatitis, in those with an age ≥70 years, and in those with IS therapy for autoimmune disease or chemotherapy for malignancy.21,26
This study showed that ribavirin therapy was a safe treatment option in patients with acute HEV infection who received IS therapy for SOT, malignancies, or immune-mediated disease. In our cohort, 72% of non-cirrhotic patients with IS therapy received ribavirin at a median time of 8 days after diagnosing acute HEV infection. Although ribavirin therapy was not always tailored to HEV RNA serum levels, and continued beyond viral clearance in only 50% of patients, an initial viral clearance was observed in 94% and sustained viral clearance was observed in 78% of patients. Viral relapse in ribavirin-treated acute HEV infection was diagnosed in 17% of patients after a median time of 110 days, and one patient did not show HEV clearance after 222 days at the end of the follow-up.
Although the rate of sustained viral response was not significantly different between patients who received ribavirin and those who did not, patients who cleared HEV did so more frequently within 90 days when treated with ribavirin. Notably, this was achieved with a comparably low median dose of 600 mg/day. Because chronicity of HEV is a complication in patients with immunosuppression, especially in patients after SOT, prevention of viral persistence beyond 3 months appears to be a reasonable therapeutic goal for acute HEV infection in patients with IS therapy.
Spontaneous HEV clearance in SOT recipients usually occurs within the first 3 months after infection, although it has also been observed 3 to 6 months after infection.24,25 Persistent viremia beyond this time frame is considered chronic HEV infection.22,23 Previous case series have suggested that the rate of chronicity in immunosuppressed individuals is approximately 30% and varies from 20% to 60%.21,24,29–31 In our cohort, viremia for >90 days was observed in 11 (44%) patients and that >180 days was observed in 3 (12%) patients. However, notably, the intervals of the nucleic acid test were at the discretion of the treating physician, and the last observation carried forward method was used for the imputation of the data. This method may have resulted in the overestimation of the actual viremia intervals.
In our cohort, the majority of the patients received combination IS therapy with two or more substances. Tacrolimus and mTOR inhibitors are thought to promote HEV persistence, whereas mycophenolate suppresses HEV replication in vitro.20,32,33 Current guidelines recommend a reduction in immunosuppression as the first step of therapy before starting ribavirin. 23 However, in some situations, this is not possible and may even endanger the patient. A recent study on 50 patients with hematological malignancy from 11 European centers reported a survival benefit in patients treated within 24 weeks after HEV infection and a trend towards better survival when treated within 12 weeks. 21 However, among the eight patients who died, six received allogeneic HSCT, two had chronic HEV infection at presentation, and three had cirrhosis. We excluded patients with cirrhosis and only one patient received HSCT. Therefore, we did not observe any relevant mortality in our study (one patient died 19 months after the diagnosis). However, our study suggests that early ribavirin therapy is beneficial in patients with IS therapy because viral persistence beyond 3 months was less frequently observed with early ribavirin treatment, and a shorter duration to initiation was correlated with a shorter duration of viremia.
It should be noted that in non-cirrhotic patients who did not receive IS drugs, ribavirin treatment was not associated with a faster viral clearance or a significant difference in ALT concentrations, bilirubin concentrations, or the international normalized ratio than in those who received IS drugs in this study. The risk of chronic HEV infection differs between different types of immunosuppression and is thought to be highest in patients after SOT than in those with autoimmune disease or malignancies. This difference may have affected the generalizability of our results. However, because IS regimens and underlying disease were heterogenous in our cohort, we decided against further subgroup analysis. This issue should be addressed in future, prospective studies.
Our study has some limitations. First, this was a retrospective, uncontrolled, single-center study. However, the baseline characteristics of patients who received ribavirin and those who did not were well balanced. Second, the sample size of the cohort was relatively small. Early treatment for patients with non-severe acute HEV infection is currently not recommended by guidelines;22,23 therefore, published data on this type of treatment are sparse. Third, the treatment decision, dose, duration, and discontinuation criteria, as well as the timing of laboratory test intervals, were based on a decision by the treating physician. We attempted to compensate for this last aspect in particular by imputing missing data, but this may have introduced bias, as described above. Additionally, some samples were only analyzed with a qualitative assay. Therefore, we could not analyze the viral load between treated and non-treated patients. Fourth, we could not assess the incidence of HEV infection in our center because we could not estimate the total number of patients with IS therapy, and RT-PCR was performed only in the presence of elevated transaminases or symptoms. Fifth, we cannot exclude the possibility that some of the patients described here already had chronic HEV infection. Although we excluded patients with positive HEV RNA, positive anti-HEV antibodies, or elevated transaminase concentrations >90 days before admission, a few of the cases that we detected may have been the first diagnosis of persistent HEV infection.
In conclusion, our study provides no evidence that early ribavirin treatment for acute HEV infection reduces viral persistence, reduces hepatic damage, or improves the clinical outcome in patients receiving IS therapy. Despite the limitations of this retrospective, observational cohort study, early ribavirin treatment could result in a shorter time of viremia. However, the efficacy of ribavirin in reducing the duration of viremia in immunosuppressed individuals needs to be validated in a randomized, controlled trial. If ribavirin is used for the treatment of acute HEV infection in immunosuppressed individuals, repeated testing for HEV RNA after the first viral clearance should be performed to detect patients with a viral relapse.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231187941 - Supplemental material for Early ribavirin for hepatitis E virus infection in patients receiving immunosuppressive therapy: a retrospective, observational study
Supplemental material, sj-pdf-1-imr-10.1177_03000605231187941 for Early ribavirin for hepatitis E virus infection in patients receiving immunosuppressive therapy: a retrospective, observational study by Anika Franz, Philipp A Reuken, Sura Guliyeva, Michael Rose, Katharina Boden, Andreas Stallmach and Tony Bruns in Journal of International Medical Research
Footnotes
Author contributions
TB designed and supervised the study. SG, AF, and PAR collected the clinical data. MR and KB identified the clinical cases. AF, PAR, and TB analyzed the clinical data, performed statistical analysis, interpreted the results, conducted the literature search, and wrote the manuscript. AS and TB critically revised the manuscript for important intellectual content. All authors read and approved the final version of the manuscript.
Data availability statement
The authors declare that the data supporting the findings of this study are available within the article and its supplementary information files.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
Tony Bruns was supported by the German Research Foundation (SFB1382 Project ID 403224013/B07).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
