Abstract
A 29-year-old woman with chronic, prolonged pustular psoriasis was admitted to our hospital because of high-grade fever and a systemic skin rash. General examination revealed a whole-body skin rash and superficial lymphadenopathy. Peripheral blood examination showed unclassified cells positive for CD3, CD4, and T-cell receptor αβ, and negative for CD20 and CD56. Soon after administration, she developed acute respiratory failure and required artificial ventilation. Bronchoalveolar lavage fluid showed increased numbers of eosinophils and abnormal lymphocytes of the same phenotype in peripheral blood and skin. She was diagnosed with eosinophilic pneumonia, and her respiratory failure was improved by corticosteroid therapy. Based on the histological findings of skin, lymph node, and bone marrow biopsies, a diagnosis of peripheral T-cell lymphoma not otherwise specified (PTCL-NOS), with positivity for CC chemokine receptor 4 was made. She received chemotherapy followed by allogeneic stem cell transplantation, which resulted in complete remission of her PTCL-NOS. She remained alive and disease-free 6 years later. This is the first reported case of PTCL-NOS developing during the clinical course of pustular psoriasis. The clinical manifestations of PTCL-NOS are complex, but an accurate diagnosis and appropriate therapy may produce a good clinical outcome in patients with PTCL-NOS.
Keywords
Introduction
Peripheral T-cell lymphoma not otherwise specified (PTCL-NOS) is a heterogeneous category of nodal and mature T-cell lymphomas that do not correspond to any of the specifically defined entities of mature T-cell lymphoma in the current classification.1,2 PTCL-NOS is the most common PTCL subtype, accounting for at least 25% of all cases of PTCL. 3 Although PTCL-NOS is a clinically rare disease, it has various clinical manifestations, including impaired immune surveillance,1–3 secondary malignancies, 4 Epstein–Barr virus (EBV) infection,1–4 and/or peripheral eosinophilia.1–3,5 In addition, the prognosis of PTCL-NOS is generally poor, with a 5-year overall survival of only 20%–30%.1–3 Furthermore, expression of CC chemokine receptor 4 (CCR4) was shown to be associated with a shorter survival time, compared with patients with CCR4-negative PTCL-NOS.6,7 The diagnosis and optimal treatment of PTCL-NOS are thus challenging because of the complexity of the clinical manifestations and the potentially poor prognosis.
We report a case of CCR4-positive PTCL-NOS that developed from chronic and prolonged pustular psoriasis, which had been treated with prednisolone and cyclosporine A. The patient presented with acute eosinophilic pneumonia at the initial diagnosis. Respiratory failure was improved by corticosteroid therapy, and subsequent chemotherapy followed by allogeneic stem cell transplantation resulted in complete remission of her PTCL-NOS for over 5 years. We here describe the clinical course and review the relevant literature.
Case report
A 29-year-old woman was admitted to our hospital with general fatigue, high fever, and a skin rash. She had suffered from pustular psoriasis since she was less than 1 year old, based on typical dermatological and histopathological findings. She had been treated with prednisolone and cyclosporine A to control the disease for over 25 years. She gave birth when she was 27, and oral prednisolone was gradually reduced and stopped 6 months after the birth. However, she developed low-grade fever and skin rash on her face 15 months after the delivery. She received oral antibiotics, but her fever did not improve and her skin rash worsened. She was admitted to our hospital for further examination. Physical examination revealed remittent fever (38.0°C), red papules and nodules on her skin (Figure 1), hepatomegaly, and superficial lymphadenopathies. Laboratory findings revealed leukocytosis (17,460/µL) and 30% abnormal and unclassified cells in her peripheral blood. Other findings included lactate dehydrogenase 945 IU/L, and soluble interleukin (IL)-2 receptor 2,079 U/mL (normal range, 135–421 U/mL). Human immunodeficiency virus and human T-cell lymphotropic virus antibodies were negative, and EBV DNA was undetectable by real-time polymerase chain reaction. Contrast-enhanced computed tomography (CT) scan revealed hepatosplenomegaly and superficial lymphadenopathy (Figure 2). (18F)-Fluorodeoxyglucose (FDG) positron emission tomography/CT scan showed increased FDG uptake in whole-body lymph nodes, spleen, liver, and bone marrow (Figure 3). Flow cytometric analysis revealed that the peripheral unclassified cells were positive for CD3, CD4, CD5, CD8, and T-cell receptor (TCR) αβ, and negative for CD20 and CD56. We performed skin, lymph node, and bone marrow biopsies. Flow cytometry analysis of each specimen showed CD3-, CD4-, CD5-, and TCRαβ-positive abnormal cells. Soon after these examinations, her peripheral eosinophils increased from 644/µL to 2949/µL after hospitalization, and her respiratory condition deteriorated rapidly. Chest radiography and CT scan (Figure 4a and 4b) revealed diffuse infiltrative shadows in the bilateral lungs. She developed acute respiratory failure and needed ventilator management in the intensive care unit. Bronchoscopy was performed and bronchoalveolar lavage fluid (BALF) revealed total cells of 16.9 × 105/mL, with differential counts of alveolar macrophages (27.2%), abnormal lymphocytes (28.6%), neutrophils (1.1%), and eosinophils (43.1%). The infiltrated lymphocytes in the lung were positive for CD3, CD4, CD5, and TCRαβ, similar to the abnormal peripheral blood cells. Based on the radiological findings and the increased eosinophils in the plasma and BALF, we speculated that the respiratory involvement might indicate eosinophilic pneumonia and infiltration of unclassified malignant lymphocytes. Corticosteroid pulse therapy was initiated under respirator management. Her respiration improved and she no longer required respirator assistance after 9 days of corticosteroid therapy (Figure 4c). While managing her respiratory impairment, the biopsy specimens (skin, lymph node, bone marrow) demonstrated CCR4-positive PTCL-NOS (Figure 5).

Reddish papules and nodules were observed on the whole skin in the present case.
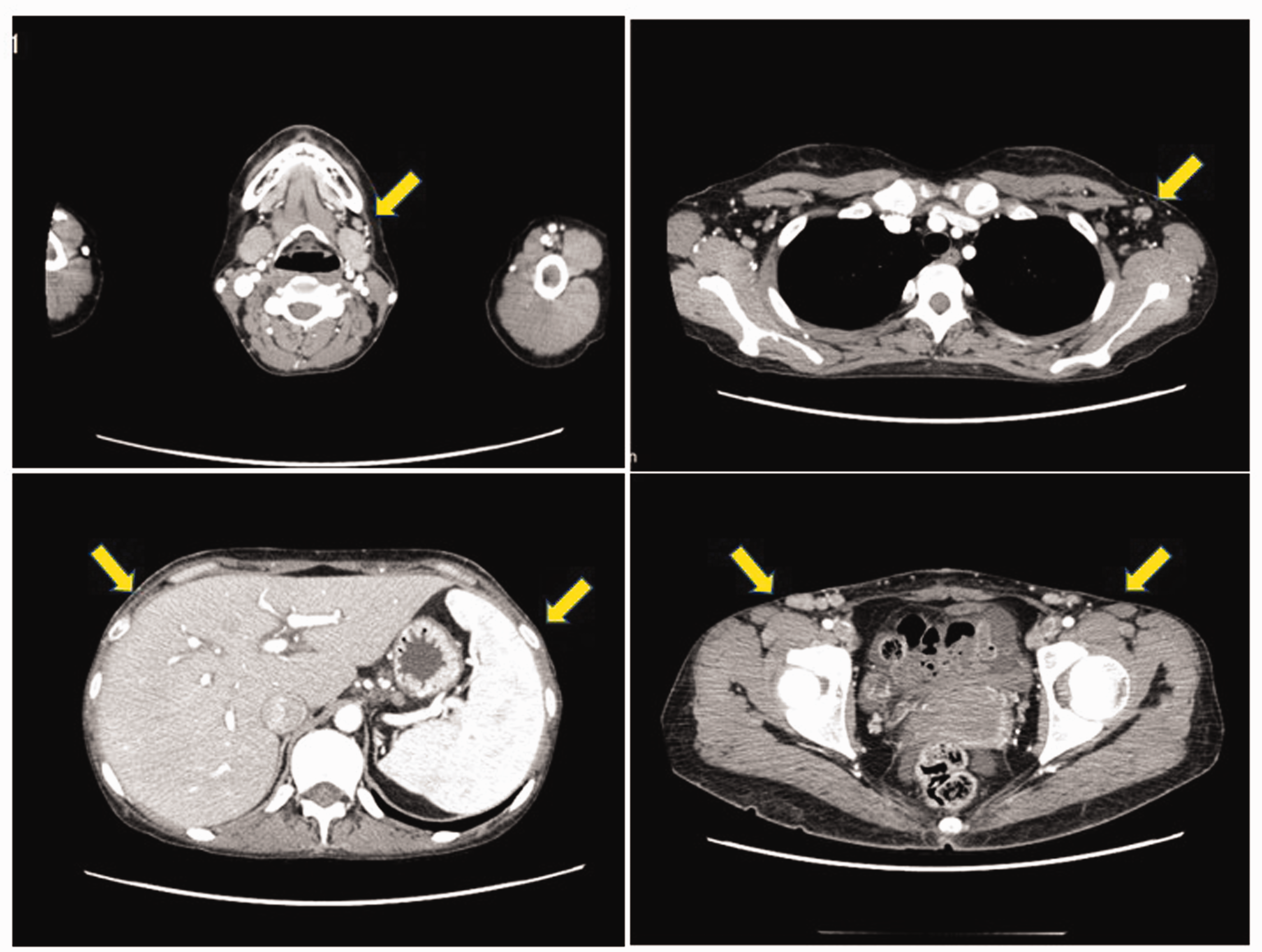
Computed tomography identified hepatosplenomegaly and superficial lymphadenopathy (yellow arrows).

Positron emission tomography with fluorodeoxyglucose (FDG)-computed tomography revealed increased FDG uptake in the lymph nodes, spleen, liver, and bone marrow.

Imaging before and after steroid therapy. Chest radiograph (a) and computed tomography scan (b) revealed diffuse infiltrative shadows in bilateral lungs. The infiltrative shadows disappeared after steroid therapy (c).
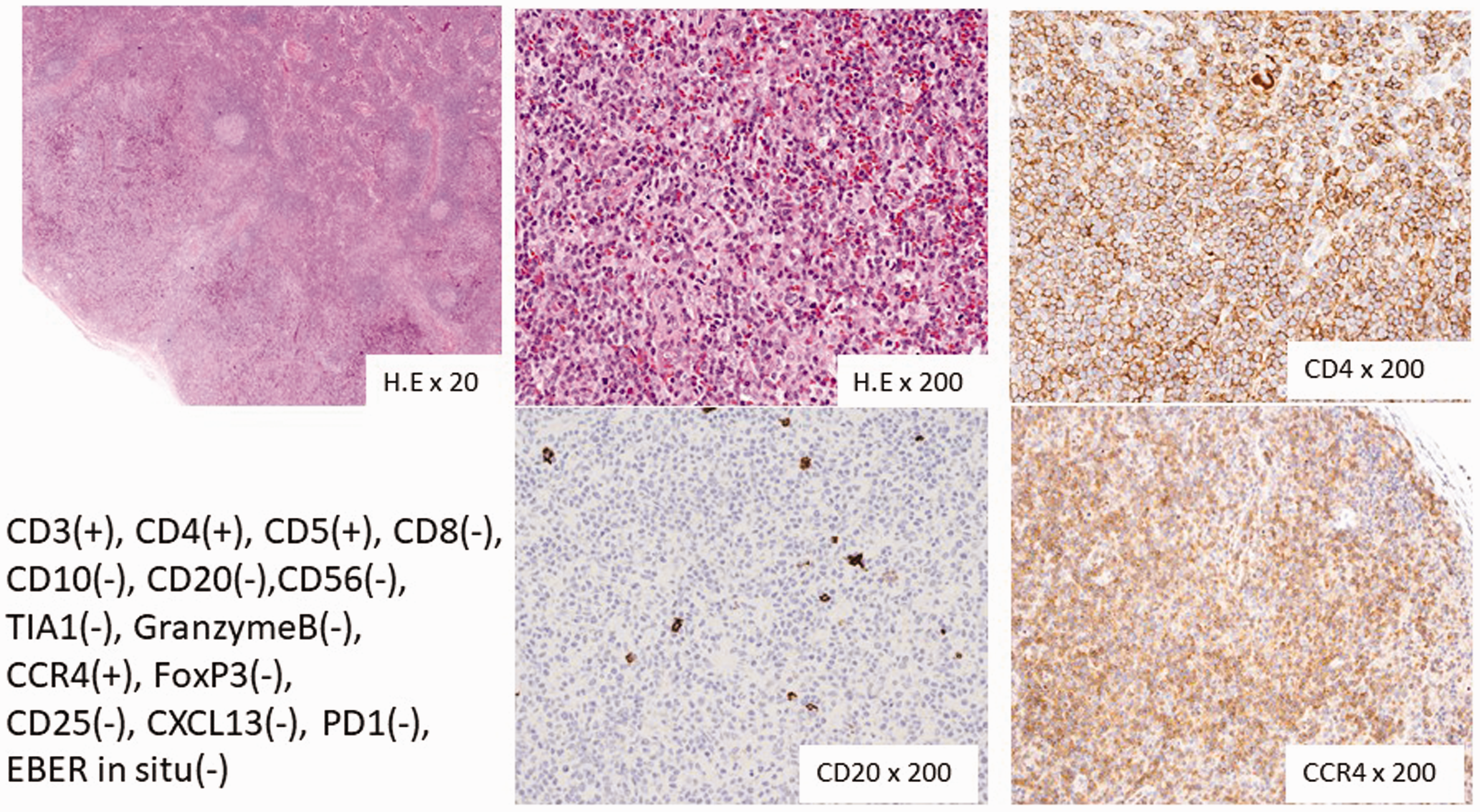
Histopathological findings in resected inguinal lymph nodes upon diagnosis of peripheral T-cell lymphoma not otherwise specified, showing diffuse proliferations of lymphoid cells positive for CD4 and CC chemokine 4 (CCR4), and negative for CD20.
After the diagnosis, the patient received six cycles of CHOEP-14 chemotherapy (cyclophosphamide, doxorubicin, vincristine, prednisolone, etoposide), with no specific toxicities, resulting in no abnormal cells in the peripheral blood and bone marrow. She then underwent myeloablative conditioning and allogeneic stem cell transplantation, with peripheral blood stem cells from an HLA fully matched donor. Tacrolimus and methotrexate were used as immunosuppressants to prevent graft-versus-host disease, followed by prednisolone. Bone marrow examination after stem cell transplantation confirmed complete remission. She was discharged from hospital 54 days after her stem cell transplant. She developed acute pancreatitis during hospitalization and viral meningitis after hospitalization, but these complications improved following appropriate treatments. Her skin involvement due to pustular psoriasis was mild during prednisolone therapy (5–10 mg/day), and she remained alive 6 years after her stem cell transplant.
We retrospectively measured IL-5 levels in the plasma and BALF at the onset of acute respiratory failure. Her BALF and plasma IL-5 levels were 469 pg/mL and 164 pg/mL, respectively, just before steroid treatment. The plasma IL-5 was higher than 2 weeks before (<3.9 pg/mL) and 3 weeks after (<3.9 pg/mL) the onset of acute eosinophilic pneumonia.
Discussion
We report on a young woman who developed PTCL-NOS during prolonged treatment with prednisolone and cyclosporine A for pustular psoriasis. There is a relationship between the use of immunosuppressive therapies and the development of lymphoproliferative malignancies, including PTCL.1–3,8,9 However, most cases involve classical immunosuppressive treatments including cyclosporine A and methotrexate, 8 and the resulting lymphomas are mainly B-cell non-Hodgkin's lymphomas associated with EBV infection. 9
The median age at presentation of PTCL-NOS is approximately 60 years, with a male predominance. 1 Unusually therefore, the current case was a young female with undetectable EBV. We speculated that the development of PTCL-NOS in this case was associated with prolonged cyclosporine A therapy for pustular psoriasis. We searched the PubMed database using the keywords “pustular psoriasis” and “PTCL”, but found no similar reports. To the best of our knowledge therefore, this case provides the first report of PTCL-NOS in a patient with a history of pustular psoriasis. Notably, the current patient also showed expression of CCR4. CCR4 is generally highly expressed in mature T-cell lymphomas, such as adult T-cell leukemia/lymphoma, and cutaneous T-cell lymphoma. 10 Although the mechanism or interaction was unknown, the expression of CCR4 in the present case of PTLC-NOS might also have been related to the prolonged therapy with immunosuppressants and/or clinical inflammation associated with pustular psoriasis.
Most patients with PTCL-NOS present with advanced disease and have a poor prognosis, with a 5-year overall survival of only 20% to 30%.1–3,11 In addition, patients with CCR4-positive lymphomas have a significantly shorter survival time compared with CCR4-negative patients6,7 The present patient had a good clinical outcome after CHOEP chemotherapy and allogeneic stem cell transplantation. The addition of etoposide to CHOP previously improved the treatment-free interval compared with CHOP therapy in a subset of young patients with PTCL-NOS. 3 Although the heterogeneity and rarity of PTCL-NOS mean there is currently no standard treatment, allogeneic stem cell transplant therapy is considered to be a novel therapeutic option for PTCL-NOS.12,13 The current case provides important additional information, but further case series are needed.
Eosinophilic pneumonia involves pulmonary infiltration of the lung by eosinophils. The etiology of this disease has been related to helminth infections, drugs, environmental exposure, and collagen disease. 14 Although eosinophilic pulmonary infiltration in patients with malignant lymphoma is extremely rare, several cases of eosinophilic pneumonia have been reported in patients with T-cell lymphoma.5,15,16 Eosinophilia is probably secondary to the production of cytokines including IL 4–6 and 10 by YH2 cells. 14 IL-5 levels in the BALF were found to be increased in several patients with eosinophilic pneumonia15–17; however, the level in the current case was higher than in other reports.15–17 The proportion of CCR4-expressing T cells among CD4+ T cells was higher in BALF than in the peripheral blood in patients with eosinophilic pneumonia, 15 and there was a significant correlation between the number of CCR4+ CD4+ T cells and IL-5 levels in the BALF in patients with eosinophilic pneumonia. 15 Tumor-cell derived IL-5 in the present case might thus be related to the development of eosinophilic pneumonia, while CCR4 might play an important role in the onset of eosinophilic pneumonia in patients with T cell lymphoma. Further studies are needed to determine the role of CCR4 in eosinophilic pneumonia.
Conclusion
We report a case of CCR4-positive PTCL-NOS that developed during the clinical course of pustular psoriasis. Although the diagnosis and management of PTCL-NOS are difficult owing to the complexity of the disease, this case highlights eosinophilic pneumonia as a possible complication, and pustular psoriasis as an underlying disease of PTCL-NOS. In addition, appropriate treatment, including bone marrow transplantation, could improve the clinical outcome. It is therefore necessary to be aware of the presence and clinical features of PTCL-NOS.
Footnotes
Ethics statement
The patient provided written informed consent for all the treatment procedures and for publication of this case report and any accompanying images. Ethical approval by the institutional research committee was not relevant or applicable for this case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
