Abstract
Objectives
The exact etiology of Parsonage–Turner syndrome is unknown, but it is known to be preceded by infection, vaccination, or surgical intervention. In this review, we describe associations of Parsonage–Turner syndrome with COVID-19 infection and vaccination.
Methods
A systematic literature search was conducted using PubMed/MEDLINE, ScienceDirect, and Google Scholar. Microsoft Excel was used for data extraction and statistical analysis. The quality of case reports and case series was assessed using the Joanna Briggs Institute Critical Appraisal Tool.
Results
We selected 44 case reports and 10 case series, including 68 patients (32 post-vaccination and 36 with post-COVID-19 infection Parsonage–Turner syndrome). Middle-aged males were predominantly affected in both groups. The most frequently administered vaccine was Comirnaty (Pfizer) (53%). The mean latency was 11.7 days in the post-vaccination group and 20.3 days in the post-infection group. The most affected nerves in both groups were the axillary, suprascapular, and musculocutaneous nerves; and 78.1% and 38.9% of patients showed partial amelioration of their symptoms in the post-vaccination and post-infection groups, respectively.
Conclusion
Post-vaccination Parsonage–Turner syndrome presents earlier than post-infection disease. Pain and sensorimotor deficits of the upper limb are common in both situations. Complete or partial recovery occurs in most cases.
Keywords
Introduction
Parsonage–Turner syndrome (PTS) is a lesser-known neurologic condition that was first described by Parsonage and Turner 1 in 1948. It has been reported in the literature many times since then under different names, including amyotrophic neuralgia, 2 brachial neuritis, 3 and acute brachial neuropathy. 4 PTS commonly affects adult men and typically presents unilaterally, with sudden onset of severe shoulder pain followed by motor weakness and sometimes patchy sensory deficit of the upper limb. 5 Many atypical presentations, including diaphragm and lower limb defects, have been reported in addition to the characteristic shoulder girdle defects.6,7 The exact etiology is currently unknown, but the onset of the disease is preceded by upper respiratory tract viral infection, vaccination, or surgery. Genetic determinants may also affect the threshold for the development of disease.8,9 Magnetic resonance imaging (MRI) and electromyographic and nerve conduction studies are helpful in confirming the diagnosis. The prognosis of most cases is excellent, with complete recovery. 5
Coronavirus disease (COVID)-19 was declared to be a pandemic in January 2020 by the World Health Organization (WHO) and >630 million cases had been reported by November 2022. 10 In early 2021, a robust vaccination drive, using a number of mRNA-based and vector-based vaccines, was launched worldwide to curb the pandemic, and >4.97 billion people had been fully vaccinated by November 2022. 10 Two processes, viral infection and vaccination, the principal suspected triggers of PTS, were present during the period of the pandemic. In July 2020, Siepmann et al. 11 reported the first case of PTS in a patient with confirmed COVID-19. The rapidly increasing literature regarding the topic merits a systematic review to explore the possible triggers, pathophysiology, clinical presentation, and the outcomes of this association to familiarize clinicians with this disease as a possible sequela of COVID-19 infection or vaccination and to provide a foundation for further work aimed at increasing knowledge of the potential complications of COVID-19.
Methods
This article is fully compliant with the PRISMA (Preferred Reporting Items for Systematic Reviews) 2020 statement. 12 The review was registered with the international prospective register of systematic reviews PROSPERO (registration number: CRD42022372719). Institutional ethics approval was not required for this retrospective data analysis. The authors ensured that consent had been obtained from the patients for the original publication of their cases.
Search strategy
A systematic literature search was conducted up to 1 November 2022 of the following four databases: PubMed/MEDLINE, Cochrane, ScienceDirect, and Google Scholar. The search string consisted of a combination of the following keywords and MeSH terms: “COVID-19” [MeSH], “Covid*”, “SARS-CoV-2”, “COVID Vaccination” [MeSH], “Vaccination”, “Vaccine”, “Parsonage–Turner syndrome”, “Brachial plexopathy”, “Neuralgic amyotrophy”, “Amyotrophic neuralgia”, and "Brachial plexus neuritis” [MeSH].
The complete search string used in each database is provided in the supplementary files. No filters regarding time, study design, language, or country of publication were used in the retrieval of the relevant publications.
Eligibility criteria
Inclusion criteria
All articles that described PTS, confirmed by electrodiagnostic or radiological studies, in association with COVID-19 infection or vaccination, were included.
Exclusion criteria
Articles describing patients in whom a diagnosis of PTS was not confirmed by electrodiagnostic or radiological studies, or in which a diagnosis was not associated with COVID-19 infection or vaccination, were excluded.
Study selection
Relevant articles were selected and screened according to the PRISMA flowchart presented in Figure 1. The records identified through a preliminary search were downloaded into Mendeley (Elsevier, Amsterdam, Netherlands) and duplicates were removed. Two authors (MZA and FA) independently performed the screening and identified case reports, case series, abstracts, and letters to the editor reporting cases of PTS in association with COVID-19. The bibliographies of these articles were also screened to identify any omitted cases.
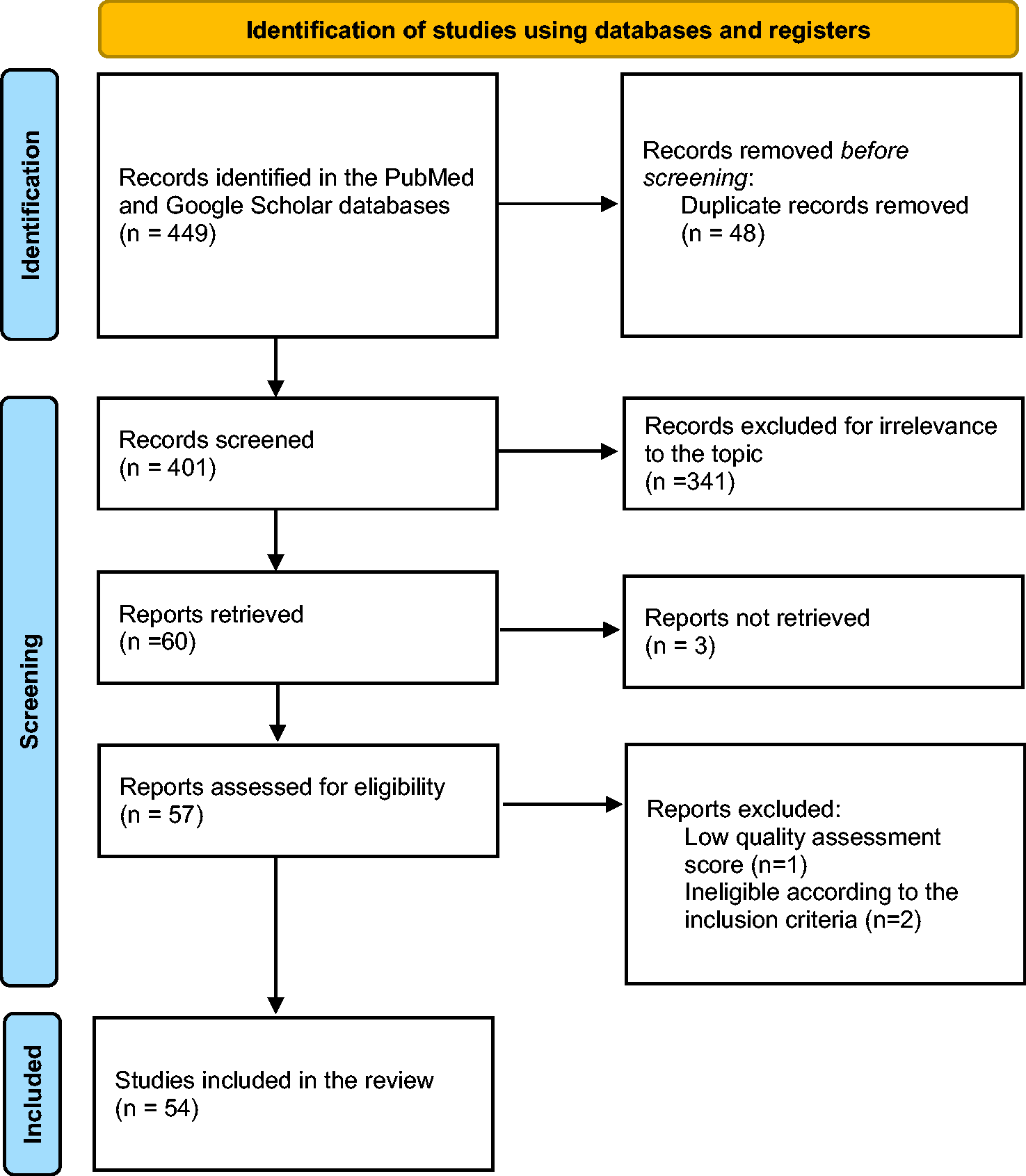
PRISMA flow diagram for study selection.
Data extraction
Data were extracted and collated in the form of two tables, one summarizing the demographics, clinical features, imaging, electrodiagnostic investigations, and outcomes of COVID-19-associated PTS (Table 1); and the other for summarizing the same parameters for COVID vaccination-associated PTS (Table 2). Continuous variables are presented as means ± standard deviations and categorical variables are presented as absolute values and percentages. Microsoft Excel (Redmond, WA, USA) was used for the data extraction and statistical analysis. Datasets were compared using the unpaired t-test and chi-square test, as appropriate. Articles in languages other than English were translated using Google Translate (translate.google.com). The references were added using Zotero (zotero.org).
Demographics and clinical characteristics of patients with COVID-associated Parsonage–Turner syndrome

F, female; M, male; n/r, not reported; EMG, electromyography; NCS, nerve conduction studies; MUAP, motor unit action potential; SNAP, sensory nerve action potential; MRC, Medical Research Council muscle power scale; STIR, short tau inversion recovery; MRI, magnetic resonance imaging; USG, ultrasonography; NSAIDs, non-steroidal anti-inflammatory drugs; APB, abductor pollicis brevis; ECR, extensor carpi radialis; ED, extensor digitorum; EDC, extensor digitorum communis; EI, extensor indicis; FCU, flexor carpi ulnaris; FPL, flexor pollicis longus; OP, opponens policis.
Demographics and clinical characteristics of patients with COVID vaccination-associated Parsonage–Turner syndrome

F, female; M, male; n/r, not reported; EMG, electromyography; NCS, nerve conduction studies; MUAP, motor unit action potential; SNAP, sensory nerve action potential; MRC, Medical Research Council muscle power scale; MRI, magnetic resonance imaging; STIR, short tau inversion recovery; USG, ultrasonography; CXR, chest X-ray; NSAIDs, non-steroidal anti-inflammatory drugs; ADM, abductor digiti minimi; APB, abductor pollicis brevis; ECR, extensor carpi radialis; ED, extensor digitorum; EI, extensor indicis; EPL, extensor pollicis longus; FCU, flexor carpi ulnaris; FDP, flexor digitorum profundus; FDS, flexor digitorum superficialis.
Quality assessment
The quality of the case reports and case series was assessed using the Joanna Briggs Institute Critical Appraisal Tool. 13 Three authors (MHB, AUH, and HA) first independently scored each article and then decided on a score for each. The scores are provided in the supplementary files.
Results
The search of the four databases identified 449 articles, of which 48 were excluded because of duplication, 341 were not relevant, 3 were not eligible, and the full text of 3 could not be retrieved. The remaining 54 articles, comprising 44 case reports and 10 case series, were included in the final quantitative synthesis.11,14–66 The systematic review of these articles included individual data from 68 patients, who were categorized according to whether the likely trigger was COVID-19 infection or vaccination. The demographics and clinical characteristics of the patients are summarized in Table 1 and Table 2. A comparison of the findings in the two groups is shown in Table 3.
Comparison of findings in patients with COVID-associated and COVID vaccination-associated Parsonage–Turner syndrome
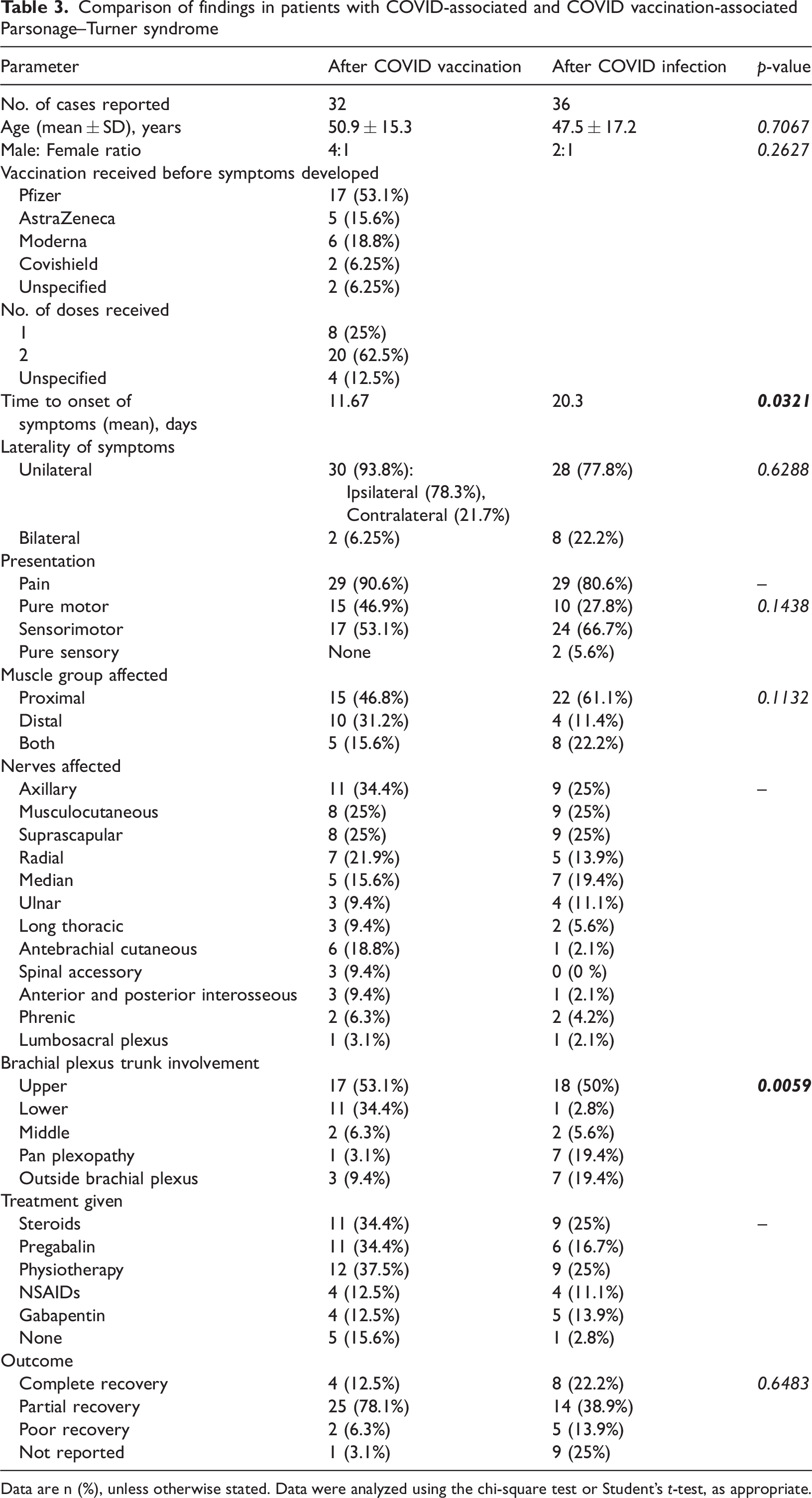
Data are n (%), unless otherwise stated. Data were analyzed using the chi-square test or Student’s t-test, as appropriate.
The mean age of the 32 patients who developed PTS after vaccination was 50.6 ± 15.3 years, and 25 (78%) were male. Most patients (n = 17, 53%) had received Comirnaty (Pfizer, New York, NY, USA). The mean interval between vaccination and the onset of symptoms was 11.7 days. The patients presented with the following symptoms: pain (n = 29, 90.6%), motor weakness (n = 15, 46.9%), and sensorimotor deficits (n = 17, 53.1%). The symptoms were unilateral in 30 (93.8%) and bilateral in 2 (6.3%) patients. The axillary nerve was affected in 11 (34.4%) patients. Thirty patients (83.3%) had MRI findings and 32 patients (88.9%) had electrodiagnostic findings suggestive of PTS. Twelve (37.5%) patients were treated with physiotherapy and 11 (34.4%) with steroids or pregabalin. Twenty-nine (93.5%) patients for whom outcomes were reported had achieved complete or partial recovery at follow-up.
The mean age of the 36 patients who developed PTS after COVID-19 infection was 47.5 ± 17.1 years, and 25 (69.4%) were male. Eleven of these patients (30.6%) had undergone prone ventilation. The mean interval between the onset of infection and the onset of symptoms was 20.3 days. The patients presented with pain (n = 29, 80.6%), motor weakness (n = 10, 27.8%), sensorimotor deficits (n = 24, 66.7%), and/or pure sensory deficits (n = 2, 5.6%). Twenty-eight (77.8%) patients had unilateral symptoms and 8 (22.2%) had bilateral symptoms. The suprascapular, musculocutaneous, and axillary nerves were affected in nine (25%) patients each. Proximal weakness was reported in 22 patients (61.1%) and distal weakness in 4 (11.4%). Seventeen patients (53%) had MRI findings and 31 patients (96.8%) had electrodiagnostic findings suggestive of PTS. Nine (25%) patients were treated with steroids and nine (25%) patients with physiotherapy. Twenty-two (81.5%) patients for whom outcomes were reported had achieved complete or partial recovery at follow-up.
Discussion
PTS is a rare neurological phenomenon that was first described by Parsonage and Turner in 1948. 1 Although its exact etiology is unknown, according to recent studies, viral infection and immunization are the most common risk factors. 5 The development of this rare disease has been associated with a number of etiologies, including hepatitis E, 67 cytomegalovirus, 68 varicella zoster virus, 69 and Borrelia burgdorferi 70 infections. PTS has also been shown to be associated with immunization, with clinical evidence suggesting that vaccinations against influenza, 71 typhoid, 72 human papillomavirus, 73 and DPT 74 in children are possible etiologic factors. Because cases of PTS have been increasingly reported following COVID-19 vaccination, Kim et al. 75 performed a study using disproportionality analysis, and concluded that the risk of neuralgic amyotrophy associated with COVID-19 vaccines does not exceed that associated with influenza vaccines. However, this weak association is still crucial in the context of both vaccination hesitancy among the general population and other COVID-19 vaccination-related neuropathic symptoms and syndromes. 76
During the COVID-19 pandemic, cases of PTS were reported after both vaccination and infection. In this review, we have summarized the findings associated with 32 cases that developed following vaccination and 36 that developed following infection. Under both circumstances, middle-aged men were the most commonly affected group. The characteristics of the two etiologic groups were similar, except that the mean latency period for PTS following COVID-19 infection was almost double that following vaccination (p = 0.0321). Vaccinated patients also showed a higher prevalence of the involvement of the lower trunk of the brachial plexus than patients who had COVID-19 infection (p = 0.0059).
The majority of patients in the post-vaccination group had received the Comirnaty vaccine, followed by Spikevax (Moderna) and the AstraZeneca vaccine/Vaxzevria (Astra Zeneca, Cambridge, UK). While the exact pathogenesis of vaccination-associated PTS remains unclear, molecular mimicry and bystander activation of immune-mediated mechanisms affecting the brachial plexus are the likely culprits in certain individuals who are susceptible to the condition. 6 The delay in the onset of symptoms following the administration of vaccines, calculated to be a mean of 11.7 days in the present review, is consistent with this pathogenetic mechanism. This latency period is similar to the time required to achieve a maximal immune response following vaccine administration. 77 In view of the above findings, it can be theorized that mRNA vaccines are more likely to be associated with the subsequent development of PTS, and this hypothesis has been previously been tested and confirmed by Kim et al. 75 This finding is especially important in the context of the COVID-19 pandemic, during which clinicians should have a high index of suspicion of PTS in patients who present with compatible symptoms who have received Comirnaty in the recent past. It is noteworthy that, in the present review, we found that most (78.3%) patients experienced symptoms of PTS ipsilateral to the injection site, which suggests that the pathogenesis may involve the local spread of the inoculum. We also found that most patients who developed PTS following vaccination were treated with corticosteroids and non-steroidal anti-inflammatory drugs, and most showed partial or complete recovery within a year. This finding is indicative of a pathogenetic mechanism involving inflammation in such cases.
It is also important to discuss the pathogenesis of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-induced PTS. SARS-CoV-2 binds to angiotensin-converting enzyme-2 receptors in various tissues, including the lungs, olfactory epithelium, intestinal epithelium, and endothelial cells in the kidneys and blood vessels. This accounts for the multi-organ defects associated with “long COVID”. SARS-CoV-2 also elicits an aberrant immune response, causing an overproduction of proinflammatory cytokines. 78 Both these models are supported by the findings of edema in patients by MRI, indicative of inflammation in the nerves and muscles involved.
Cheung et al. 17 reported a case of PTS following COVID-19 infection in a 55-year-old man, who presented with pain that extended from his neck and right interscapular region to his shoulder and along his arm and forearm; a weak hand grip, and disturbed sleep. His sister had also developed similar neuropathic symptoms. Pivaral et al. 28 also reported two post-infection cases of PTS in two siblings. The clinical presentations of the two siblings were similar, and nerve conduction studies demonstrated abnormal postganglionic sensory nerve action potentials in the left brachial plexus at the level of the trunk. This suggests that genetics may be involved in the etiology of post-infection PTS. Kuhlenbäumer et al. 9 have identified three mutations in the gene encoding septin 9 (SEPT9) in six families with hereditary neuralgic amyotrophy (HNA) that are linked to chromosome 17q25. PTS is a more common, sporadic form of painful brachial plexus neuropathy that is clinically indistinguishable from HNA, and the genes involved may be closely linked to or even at the same locus. However, this possibility must be evaluated in future studies.
Of the 36 patients who had been diagnosed with COVID-19 prior to the development of PTS, 11 had received prone ventilation as part of their treatment. Prone positioning improves the distribution of blood flow and ameliorate the ventilation-perfusion mismatch, in addition to helping with the drainage of secretions, and hence is used as an adjunct in patients requiring mechanical ventilation. 79 Surgical interventions involving over-abduction of the arms in a prone position can damage the brachial plexus, 80 possibly by causing injury to the blood-nerve barrier of the brachial plexus. 81 Although there is mounting evidence that PTS also develops following surgical procedures that involve little-to-no traction or compression injury of the brachial plexus, 82 mechanical trauma during the ventilation of patients with COVID-19 must be considered when investigating the etiology of post-infection PTS.
Given that the exact etiology of PTS is not known, and some of the patients had concomitant factors that could have contributed to its development in various ways, it is critical to consider the possible interplay between multiple risk factors. However, we believe that the well-known neuropathic potential of SARS-CoV-2 is consistent with such an association. Andalib et al. 83 studied nerve pain and skeletal muscle injury, Guillain–Barré syndrome, cranial polyneuritis, and neuromuscular junction disorders as peripheral nervous system manifestations in patients with COVID-19. We believe that PTS can be added to this list of the possible neurologic manifestations of COVID-19. Similarly, SARS-CoV-2 vaccination has been reported to be associated with neuropathic side effects. In their study, Safavi et al. 76 reported new neuropathic symptoms that developed within the first month following SARS-CoV-2 vaccination, including paresthesia, a burning sensation, numbness, and autonomic symptoms.
Although most of the reported patients presented with similar symptoms, some atypical clinical findings were also observed. Kang and Cho 52 reported a case of PTS that followed COVID-19 vaccination that was characterized by a restrictive pattern on pulmonary function testing. Kim et al. 65 also reported a case of post-vaccination PTS that featured lower extremity involvement, instead of the more typical upper extremity involvement; and lumbosacral plexopathy (L2–L4) that was confirmed by electromyography. There have been previous reports of lumbosacral plexus involvement, but this is commonly associated with an underlying medical illness. 84
The clinical presentations of patients in the present study are comparable to those reported in earlier studies of PTS. Van Alfen et al. 6 analyzed 246 cases and concluded that most patients are middle-aged men (mean age, 41.3 years) who initially present with pain, followed by unilateral weakness of the upper limbs. The upper trunk of the brachial plexus is most frequently involved, and most patients show partial recovery, with some residual symptoms.
To the best of our knowledge, this is the first systematic review of all the evidence available regarding the incidence of PTS in association with COVID-19 infection or vaccination (Figure 2). An awareness of such an association may aid in the early diagnosis, reduction of morbidity, and a better prognosis for patients with PTS.

Summary of Parsonage–Turner syndrome associated with COVID vaccination or infection
The authors would also like to acknowledge a few limitations in the reporting of their findings. Owing to the rarity of the disease and its association with COVID-19, some cases of post-vaccination or post-infection PTS may have been misdiagnosed or missed completely. This issue of unreported cases brings into question the generalizability of the present findings to the general population. Moreover, there was also an inherent heterogeneity in the results, owing to inter-individual variations in the reported patients and subjectivity in reporting of the cases by the physicians. However, given the rigorous methodology used for literature searching and data extraction, we believe that we have provided a comprehensive overview of all the available literature regarding the clinical and pathophysiological aspects of PTS.
Conclusions
The incidence of PTS has increased considerably during the rollout of the COVID-19 vaccines. Post-vaccination PTS presents earlier and involves the upper or lower trunks of brachial plexus, in contrast to the post-infection disease, which presents later and principally involves the upper trunk. A presentation of pain, followed by proximal weakness, and sensory deficits in the upper limb following COVID infection or vaccination, should arouse clinical suspicion of PTS. A diagnosis can be confirmed by either electrodiagnostic studies or MRI. Complete or partial recovery has been reported for most patients, irrespective of the etiology.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231187939 - Supplemental material for Association of Parsonage–Turner syndrome with COVID-19 infection and vaccination: a systematic review
Supplemental material, sj-pdf-1-imr-10.1177_03000605231187939 for Association of Parsonage–Turner syndrome with COVID-19 infection and vaccination: a systematic review by Muhammad Zain Ameer, Ata Ul Haiy, Muhammad Hassan Bajwa, Huzaifa Abeer, Biah Mustafa, Fatima Ameer, Zunaira Amjad and Aqeeb Ur Rehman in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231187939 - Supplemental material for Association of Parsonage–Turner syndrome with COVID-19 infection and vaccination: a systematic review
Supplemental material, sj-pdf-2-imr-10.1177_03000605231187939 for Association of Parsonage–Turner syndrome with COVID-19 infection and vaccination: a systematic review by Muhammad Zain Ameer, Ata Ul Haiy, Muhammad Hassan Bajwa, Huzaifa Abeer, Biah Mustafa, Fatima Ameer, Zunaira Amjad and Aqeeb Ur Rehman in Journal of International Medical Research
Footnotes
Author Contributions
MZA and AUR conceived the study. AUH and FA established the search strategy. MZA and FA retrieved the articles and screened them for relevance. AUH and ZA assisted with full text screening. MHB, AUH and HA performed the quality assessment of the selected articles. Data were extracted by AUH, MHB, and HA. FA and ZA proofread the extracted data. MZA and BM performed the statistical analysis. AUH and BM wrote the article. AUR critically revised the article. All the authors read and approved the final version of the article.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
