Abstract
Objective
This study aimed to evaluate the effect of endometrial scratch injury (ESI) in infertile women undergoing
Methods
We screened MEDLINE, CENTRAL, EMBASE, Web of Science, and the Cochrane Central Register from inception to April 2023 using keywords related to endometrial scratch, implantation, infertility, and IVF. We included 41 randomized, controlled trials of ESI in IVF cycles (9084 women). The primary outcomes were the clinical pregnancy, ongoing pregnancy, and live birth rates.
Results
The clinical pregnancy rate was reported in all 41 studies. The odds ratio (OR) for the clinical pregnancy rate had an effect estimate of 1.34 with a 95% confidence interval (CI) of 1.14 to 1.58. The live birth rate was reported in 32 studies with 8129 participants. The OR for the live birth rate had an effect estimate of 1.30 with a 95% CI of 1.06 to 1.60. The multiple pregnancy rate was reported in 21 studies with 5736 participants. The OR for the multiple pregnancy rate had an effect estimate of 1.35 with a 95% CI of 1.07 to 1.71.
Conclusion
ESI increases the clinical pregnancy, ongoing pregnancy, live birth, multiple pregnancy, and implantation rates in women undergoing IVF cycles.
Introduction
The success of
Implantation starts by attachment of the growing embryo to the uterine wall and is completed by invasion of the maternal sinusoids and establishment of the maternal–fetal circulation. 1 Successful implantation requires a precise developmental synchronization of the endometrium and blastocyst. 4 Ovarian steroids are responsible for endometrial preparation for implantation, including cellular, vascular, and immunological changes. 5
Endometrial scratch injury (ESI) is intentional endometrial injury to improve its receptivity. 6 ESI delays endometrial development owing to the wound repair process and subsequently synchronizes the development of the endometrium and embryo. 7 ESI enhances endometrial gene modulation and increases the receptivity of the endometrium. 8 The ESI effect may be mediated through immunological factors. 9 The changes in growth factors and cytokine release lead to attraction of leucocytes to the implantation site and improvement in endometrial vascularity. 10 Although the first study of the effect of ESI on IVF was published in 2003, 11 its usefulness is still questionable. There is no agreement regarding the proper timing, ideal intensity, or best number of injuries to achieve the best outcome. Therefore, this meta-analysis was conducted to evaluate the usefulness of ESI before or during the stimulation cycle in women undergoing IVF/intracytoplasmic sperm injection (ICSI) cycles.
Methods
Ethics
A protocol that followed the PRISMA guidelines for a meta-analysis of randomized, controlled trials (RCTs) was prepared prospectively. The registration number is CRD42020151590. Consent was not required for this meta-analysis.
Eligibility criteria, information sources, and search strategy
The electronic databases MEDLINE, CENTRAL, EMBASE, Web of Science, and the Cochrane Central Register of Controlled Trials were searched by two authors (AM and AE) from inception to April 2023 using the keywords related to endometrial scratching, implantation, IVF, and their MeSH terms. Additionally, trial registration sites, Google Scholar, reference lists of related studies, and abstracts of international gynecology and infertility conferences were searched. Contacting authors via emails was performed for any missing information or the requirement for clarification.
Study selection
Our systematic review included all RCTs (published and unpublished) that involved ESI at different timings, preceding or at the same stimulation cycle, and of different intensities in different participants. All RCTs with single or double ESI compared with no intervention, cervical manipulations, or sham procedures were included.
Clinical pregnancy, ongoing pregnancy, and live birth rates were the primary outcome parameters. Secondary outcomes were miscarriage, multiple pregnancy, and implantation rates. The authors were contacted for any non-reported or unclear data, such as the unpublished data of Polanski and colleagues.
Data extraction
Assessment of all studies and extraction of data were performed by two investigators (AM and AE) independently, and disagreements were discussed with other investigators. The extracted data included the location, the number of participants and their characteristics, ESI (timing, intensity, and number), and primary and secondary outcomes.
Assessment of risk of bias
The Cochrane Handbook of Systematic reviews recommendations was followed in quality assessment of the included RCTs. Quality assessment was performed by two investigators (AM and YL) independently, and disagreements were discussed with other investigators. Items included random sequence generation, allocation concealment, blinding of the participants and outcome assessors, incomplete outcome data, selective reporting, and other bias. Absent and unclear data were checked by contacting the authors.
Statistical analysis
The effect estimate of outcomes was reported as the odds ratio (OR) and its 95% confidence interval (CI) for all dichotomous data. A subgroup analysis was performed according to the timing (follicular phase of the preceding cycle of stimulation, follicular phase of the same cycle of stimulation, luteal phase of the preceding cycle of stimulation, both follicular and luteal phases, or unspecified timing), the number of previous IVF cycles (first IVF cycle, one previous IVF failure, two or more previous IVF failures, or without including previous IVF failures in the selection criteria), the number of ESIs (single or double), and the intensity of ESI (mild to moderate or high). Analyses were performed with Review Manager (RevMan) version 5.1.7 (The Nordic Cochrane Centre, Copenhagen, Denmark).
Results
Our search yielded 1055 studies through databases, and 5 records were identified through other resources. A total of 681 studies were screened after the removal of duplicates, and 70 were screened for the full text (Figure 1).
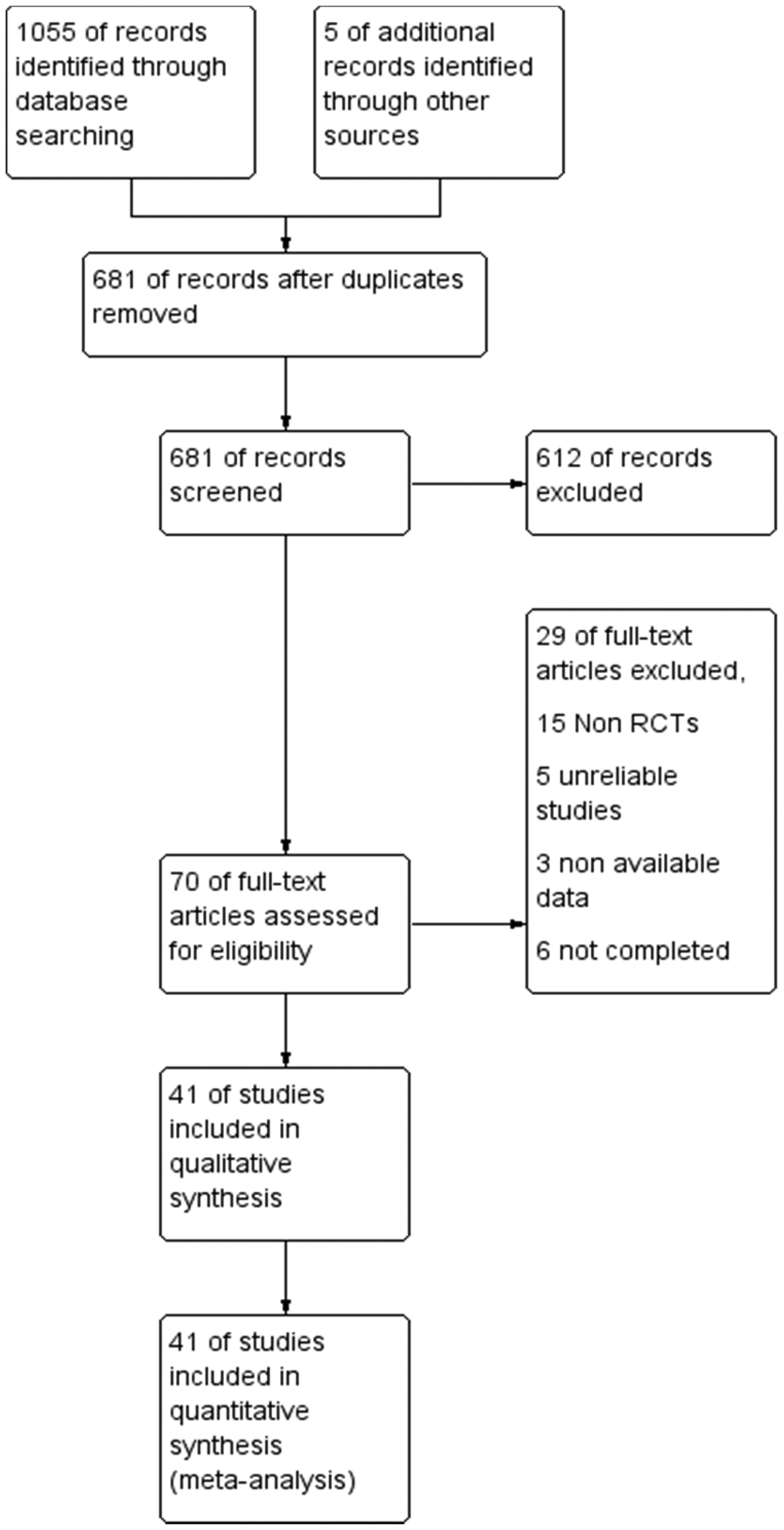
PRISMA flow chart.
Study selection and characteristics
We finally included 41 studies with 9084 women who fulfilled the inclusion criteria. All studies were in the English language. Eleven RCTs were multicenter studies (1/32 centers, 12 1/16 centers, 13 1/13 centers, 14 1/4 centers, 15 3/3 centers,16–18 and 4/2 centers19–22), while the remaining 30 studies were in a single center. One study was conducted in five countries, 14 and another study was conducted in two countries. 20 The remaining 39 studies were conducted in one country (7 in Iran,21,23–28 5 in Egypt,4,16,19,29,30 4 in China,22,31–33 3 in India,34–36 3 in Turkey,37–39 2 in the United kingdom,13,40 2 in Hong Kong,41,42 2 in Denmark,15,43 2 in the USA,44,45 and 1 each in Brazil, 46 Belgium, 47 France, 17 Israel, 48 Italy, 49 The Netherlands, 12 Poland, 50 and Spain). 51 The timing of ESI was not the same in all studies. In five studies, ESI was performed in the same follicular phase of stimulation.28,33,36,41,51 In four studies, ESI was performed in the follicular phase of the preceding cycle.20,22,38,43 In four studies, ESI was performed in both the follicular and luteal phases.33,36,45,48 In two studies, the timing of ESI was not specified.14,31 In the remaining 21 studies, ESI was performed in the luteal phase in the preceding cycles. The usefulness of ESI was tested in women undergoing their first IVF cycle in eight studies,7,18,23,24,28,37,44,46 in those with one previous IVF failure in one study, 12 and in those with two or more IVF failures in nine studies.20–22,24,26,34,42,48,49 In 23 studies, the number of previous IVF failures was not included in the selection criteria. In the included studies, ESI was performed twice in 8 studies16,23,28,35,36,39,45,48 and once in the remaining 33 studies. The intensity of ESI was high in only 6 studies20,23,26,34,38,43 and was mild to moderate in 34 studies. Among the 41 studies, only 11 were prospectively registered. Eighteen trials were retrospectively registered, and 11 trials were not registered. Women in the control group were subjected to the sham procedure in 8 studies,16,33,41,44–46,48,49 to hysteroscopy in 2 studies,20,36 to granulocyte colony-stimulating factor administration in 1 study, 32 and to no intervention in the remaining 29 studies. Table 1 summarizes the main characteristics of the included studies.
Characteristics of the included studies.

ET, embryo transfer; IR, implantation rate; CPR, clinical pregnancy rate; OPR, ongoing pregnancy rate; MR, miscarriage rate; MPR, multiple pregnancy rate; ChPR, chemical pregnancy rate; IVF, in vitro fertilization; BMI, body mass index; FSH, follicle-stimulating hormone; LBR, live birth rate; AFC, antral follicular count; OHSS, ovarian hyperstimulation syndrome; GnRH, gonadotropin-releasing hormone; PGD, preimplantation genetic diagnosis; RIF, recurrent implantation failure; ART, assisted reproductive technique; ICSI, intracytoplasmic sperm injection; SLE, systemic lupus erythematosus; HSG, hysterosalpingography; AMH, anti-Mullerian hormone.
Synthesis of results
The clinical pregnancy rate was reported in all 41 studies. The OR was 1.34 with a 95% CI of 1.14 to 1.58 and a P value of 0.0005 (Figure 2). The subgroup analysis of the clinical pregnancy rate is shown in Figure 2 and Table 2.
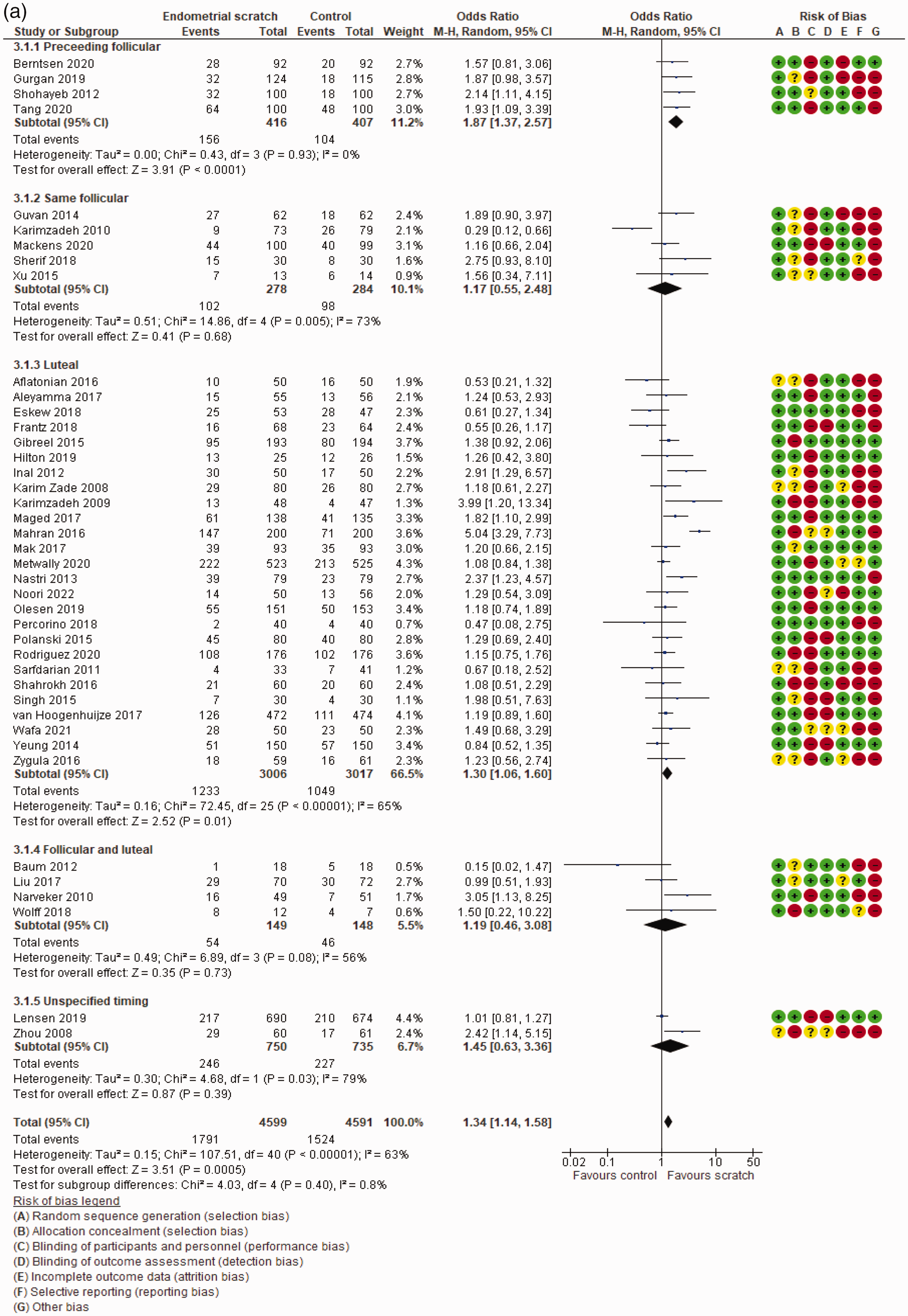
Clinical pregnancy rate according to (a) the time of endometrial scratch injury, (b) the number of previous IVF cycles, (c) the number of endometrial scratch injuries, and (d) the intensity of endometrial scratch injury.
Subgroup analysis of outcome parameters

OR, odds ratio, CI, confidence interval; IVF,
The ongoing pregnancy rate was reported in 32 studies that included 8274 participants. The OR was 1.32 with a 95% CI of 1.09 to 1.61 and a P value of 0.005. The subgroup analysis according to the timing of ESI, number of previous IVF cycles, number of ESIs, and intensity of ESI is shown in Figure 3 and Table 2.

Ongoing pregnancy rate according to (a) the time of endometrial scratch injury, (b) the number of previous IVF cycles, (c) the number of endometrial scratch injuries, and (d) the intensity of endometrial scratch injury.
The live birth rate was reported in 32 studies with 8129 participants. The OR was 1.30 with a 95% CI of 1.06 to 1.60 and a P value of 0.01. The subgroup analysis according to the timing of ESI, number of previous IVF cycles, number of ESIs, and intensity of ESI is shown in Figure 4 and Table 2.

Live birth rate according to (a) the time of endometrial scratch injury, (b) the number of previous IVF cycles, (c) the number of endometrial scratch injuries, and (d) the intensity of endometrial scratch injury.
The miscarriage rate was reported in 32 studies that included 7939 participants. The OR was 0.98 with a 95% CI of 0.81 to 1.19 and a P value of 0.86. The subgroup analysis according to the timing of ESI, number of previous IVF cycles, number of ESIs, and intensity of ESI is shown in Figure 5 and Table 2.

Miscarriage rate according to (a) the time of endometrial scratch injury, (b) the number of previous IVF cycles, (c) the number of endometrial scratch injuries, and (d) the intensity of endometrial scratch injury.
The multiple pregnancy rate was reported in 21 studies that included 5736 participants. The OR was 1.35 with a 95% CI of 1.07 to 1.71 and a P value of 0.01. The subgroup analysis according to the timing of ESI, number of previous IVF cycles, number of ESIs, and intensity of ESI is shown in Figure 6 and Table 2.
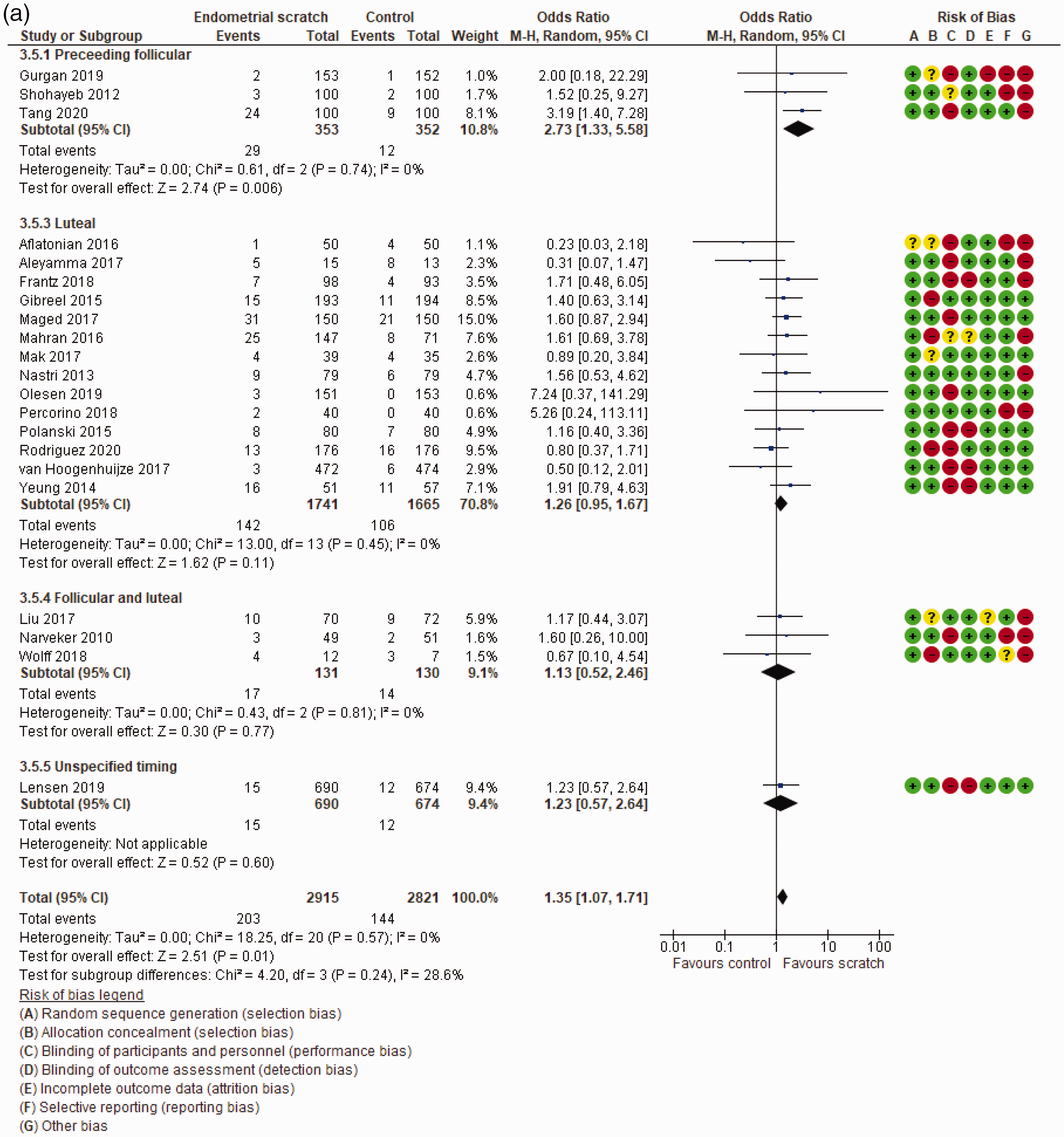
Multiple pregnancy rate according to (a) the time of endometrial scratch injury, (b) the number of previous IVF cycles, (c) the number of endometrial scratch injuries, and (d) the intensity of endometrial scratch injury.
The implantation rate was reported in 12 studies with 3391 participants. The OR was 1.38 with a 95% CI of 1.17 to 1.61 and a P value of <0.0001. The subgroup analysis according to the timing of ESI, number of previous IVF cycles, number of ESIs, and intensity of ESI is shown in Figure 7 and Table 2.

Implantation rate according to (a) the time of endometrial scratch injury, (b) the number of previous IVF cycles, (c) the number of endometrial scratch injuries, and (d) the intensity of endometrial scratch injury.
Risk of bias of included studies
Figure 8 shows the risk of bias assessment of the included studies. An adequate computer-generated random sequence was described in 35 studies, 19 had a proper allocation concealment, 8 showed appropriate blinding of both participants and personnel, 30 showed appropriate blinding of outcome assessors, 32 had no incomplete data reporting, 19 had no reporting bias, and 28 had bias related to registration.

Risk of bias summary and graph.
The quality of the evidence was evaluated using the grading of recommendations, assessment, development, and evaluations (GRADE) system and is shown in Table 3. GRADE includes publication bias, inconsistency, indirectness, imprecision, and the risk of bias in the included research. Each item reduces the evidence if there are substantial concerns about it by one level, and if there are serious issues, by two levels. The quality of evidence varied from high (if we were confident that the true effect was near to the effect estimate), moderate (if we were somewhat convinced that the true effect was close to the effect estimate, but there was the potential fora significant difference), and low (if there was no certainty that the true effect was close to the effect estimate).
Grading of recommendations, assessment, development, and evaluations quality of evidence.

N, not serious; S, serious; CI, confidence interval.
Discussion
Principal findings
In this study, ESI increased the clinical pregnancy, ongoing pregnancy, live birth, multiple pregnancy, and implantation rates in women undergoing IVF/ICSI cycles. The miscarriage rate was similar in women who underwent ESI and in those who did not undergo ESI.
There was a significant improvement in the clinical pregnancy, ongoing pregnancy, and live birth rates when ESI was performed during the follicular or luteal phase of the cycle preceding the stimulation cycle. This improvement was not significant in those who underwent ESI during the follicular phase of the same stimulation cycle or during both follicular and luteal phases. Although the clinical pregnancy rate was improved in women without specific selection criteria related to previous IVF failure, the pooled evidence of studies in women who underwent their first IVF cycle and in those with one or more IVF failures failed to show this beneficial effect. With regard to the number and intensity of ESI, the pooled evidence showed an improved clinical pregnancy rate in women who underwent a single scratch and in those who had a mild to moderate intensity scratch, but not in women with a double or high intensity scratch.
The miscarriage rate was not affected by the performance of ESI in women who underwent IVF cycles. This finding was observed in women who underwent ESI during any phase of the cycle, in all women with different numbers of previous IVF failures, and with all intensities of ESI, and regardless of whether ESI was performed once or twice.
An increase in the multiple pregnancy rate was observed when ESI was performed during the follicular phase of the cycle preceding the stimulation cycle, but this was not found if ESI was performed during the follicular phase of the same cycle of stimulation, the luteal phase, or both follicular and luteal phases. When the multiple pregnancy rate was evaluated according to the number of previous IVF failures, it was increased in women who underwent their first IVF cycle and in those with two or more previous IVF failures, but not in women with one previous IVF failure or in women with an unspecified number of IVF failures. The multiple pregnancy rate was increased in women who underwent a single ESI and mild to moderate intensity injury, but not in those with double or high intensity ESI.
There was a significant improvement in the implantation rate when injury was performed during the follicular or luteal phase of the cycle preceding the stimulation cycle. This improvement was not significant in those who underwent ESI during the follicular phase of the same stimulation cycle or during both follicular and luteal phases. The implantation rate was significantly improved in women who underwent their first IVF cycle and in those with history of two or more previous IVF failures. This beneficial effect was not observed in women without specific selection criteria related to a previous IVF failure. Data from pooled evidence confirmed an increase in the implantation rate in women who underwent single, mild to moderate intensity, or high intensity ESI, but not in those who underwent double injury.
Many theories have been suggested to explain the beneficial effects of ESI on implantation. A mechanical theory states that ESI results in a delay in endometrial maturation and subsequent enhancement of synchronicity between the endometrium and the embryo. 31 An inflammatory theory depends on the association between implantation and high levels of endometrial cytokines, chemokines, and leukocytes. 52 The wound healing process following ESI creates an optimal environment in the decidua for implantation. 53 Other theories include differential gene expression in the endometrium, recruitment of uterine immune cells to the injury site, enhancement of an angiogenic environment, and expression of a variety of proteins that co-act in the differentiation of the human endometrium. 10
Our findings suggest that ESI of mild to moderate intensity that is not performed at the time of implantation is effective in improving implantation. Mild intensity and single ESI causes a mild inflammatory reaction with ideal outcomes. However, high intensity and double scratch ESI may be associated with excessive tissue damage and an intense inflammatory reaction, which may not enhance the implantation process. Our findings also showed that ESI in the preceding cycle for implantation yielded a better implantation rate because it provided more time for starting the healing process with its beneficial effects. These effects were not evident when ESI was performed during the follicular phase in the same cycle of stimulation because it did not allow sufficient time to achieve healing of the endometrium.
Strengths and limitations
To the best of our knowledge, this is the largest systematic review to evaluate the effect of ESI in infertile couples seeking fertility through IVF cycles. We included all reliable RCTs in all available languages without restrictions. All available studies were screened and evaluated properly, and selected studies were subjected to quantitative and qualitative methodological assessment.
The main limitation is the substantial methodological heterogeneity in the inclusion and exclusion criteria among the different studies, especially regarding the previous failed IVF cycles and their numbers. We attempted to account for these effects by performing a subgroup analysis according to previous IVF failure. There was high heterogeneity regarding the timing, number, and intensity of ESI among the studies. We also attempted to control for these effects by performing a subgroup analysis according to different characteristics of ESI. We performed a meta-analysis using random effects to incorporate and examine any possible heterogenicity of the included studies. The second limitation is not reporting pain and bleeding following ESI because they were not completely reported in most studies.
Comparison of our study with existing literature
One systematic review assessed the effect of ESI on women with one or more previous failed embryo transfers 54 and another systematic review assessed the effect of ESI on women who underwent their first embryo transfer. 6 These studies focused on one category in infertile couples. We believe that studying the effect of ESI on all participants with subgroup analysis for each type of participant would provide higher evidence of the effects of ESI. These systematic reviews included 10 54 and 7 6 studies. However, we analyzed 41 studies, which provides stronger evidence of the effects of ESI than previous reviews. More conclusive evidence of the effects of ESI is required for practitioners who treat all types of patients undergoing IVF.
Metwally et al. 55 conducted a systematic review in 2022 that included the effect of scratching in women undergoing their first IVF cycle. Their review included 12 studies. A full subgroup analysis was performed because of the small number of studies.
A recent, lengthy (125 pages) Cochrane review 56 included only 37 studies. No GRADE evaluation for the quality of evidence was performed. A subgroup analysis of the timing of ESI included only the follicular and luteal phases and no recurrent, recurrent, or unclear previous failures. In our systematic review, we performed a subgroup analysis of five different timings of ESI and four groups of previous failures.
Conclusions and implications
According to our findings, we recommend the performance of ESI in all couples undergoing IVF cycles before proceeding to IVF. Practicing ESI in the preceding cycle to stimulation (at the follicular or luteal phase) leads to a better result than that in the same cycle. We recommend that mild to moderate intensity scratching should be performed once without any need to repeat it or to perform high-intensity injury. However, the evidence supporting the findings of the effects of ESI is not high, and more randomized, high-quality studies are still required.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231175365 - Supplemental material for Endometrial scratch injury in infertile women undergoing in vitro fertilization cycles: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605231175365 for Endometrial scratch injury in infertile women undergoing
Footnotes
Author contributions
AMM: Literature search, assessment, writing, and revision.
AE: Data extraction, writing, and revision.
YL: Data analysis, writing, and revision.
NE: Data extraction, writing, and revision.
All authors have read and approved the manuscript.
Synopsis
ESI increased clinical pregnancy, ongoing pregnancy, live birth, multiple pregnancy, and implantation rates in women undergoing IVF/ICSI cycles.
Summary of the study: [Database]
Why was this study conducted? To evaluate the safety and efficacy of endometrial scratching performed before or during the stimulation cycle in women undergoing IVF/ICSI cycles. What are the key findings? ESI improved the clinical pregnancy, ongoing pregnancy, live birth, multiple pregnancy, and implantation rates in women undergoing IVF/ICSI cycles. ESI had no significant effect on the miscarriage rate. What does this study add to what is already known? This study confirms the benefits of ESI in infertile women undergoing IVF/ICSI cycles. The beneficial effects of ESI are more evident when it is performed during the preceding cycle (during the follicular or luteal phase) than when it is performed in the same IVF cycle. Mild to moderate intensity injury is more beneficial than high intensity injury, and a single scratch is more effective than double injury.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
