Abstract
A man in his early 40s visited the Emergency Department because of no motor function in his lower limbs for 10 hours. Magnetic resonance imaging of his thoracic spine showed that the thoracic spinal canal (T2–T6) was occupied, and the thoracic spinal cord was compressed. In view of the severe symptoms, we quickly completed preoperative preparations and performed a thoracic laminectomy within 24 hours of paralysis of both lower limbs. Postoperatively, the patient underwent rehabilitation exercise. Four weeks later, the patient’s lower limbs had full 5/5 strength. We reviewed the related literature to summarize the clinical guidelines with spinal surgeons. Timely diagnosis of thoracic spinal epidural abscess, early surgical treatment, and anti-infection management and rehabilitation exercise are essential for the full recovery of lower limb muscle strength.
Introduction
Spinal epidural abscess (SEA) is a purulent infectious disease, which is usually spread directly through infected emboli or is blood-borne, and forms an abscess in fat and the venous plexus. SEA tends to occur in the thoracic and lumbar segments, which may be related to the rich venous plexus and fatty tissues. SEA is a clinically acute and severe disease, and has a high disability rate.1–3 The timely diagnosis of thoracic spinal epidural abscess, early surgical treatment, and anti-infection management and rehabilitation exercise are essential for the full recovery of lower limb muscle strength. We report a case of thoracic SEA. After paralysis of the lower limbs and the disappearance of sensation below the nipples in the patient, we proceeded with thoracic laminectomy and debridement. Four weeks later, both of the patient’s lower limbs had full 5/5 strength, and his feeling was completely restored. The patient had no obvious discomfort during 13 months of follow-up.
Case report
A man in his early 40 s visited the hospital for back pain for 7 days and weakness of both lower limbs for 10 hours. At onset, there was no obvious cause of his back pain, which was a dull but tolerable. At this time, the patient had a limb strength of 5/5 and did not pay attention to seeking treatment. However, 10 hours before he visited the hospital, he suddenly experienced weakness in both lower limbs, progressive aggravation, urinary incontinence, and an unresolved stool. After thoracic magnetic resonance imaging (MRI) was performed, he was transferred to our hospital. MRI showed that the thoracic spinal canal (T2–T6) was occupied, and the thoracic spinal cord was compressed. T1-weighted images showed an equal signal, and T2-weighted images showed a high signal (Figure 1).

Magnetic resonance imaging of the thoracic spine. (a) and (b) T2-weighted images in the sagittal view. (c) T1-weighted image in the sagittal view. Spinal cord compression in the T2–T6 segments (arrowheads) and (d)–(f) Spinal cord compression shown in cross-section.
The patient was previously healthy. He had a furuncle on his back a few months previously, but he recovered. A physical examination showed the following: temperature, 38.7°C; blood pressure, 84/46 mmHg; heart rate, 115 beats/minute; and respiratory rate, 28 breaths/minute. The Glasgow Coma Scale score was 15 points, he had good consciousness, good rhythm of the heart and lungs, chest and back tenderness, loss of sensation below the nipples, and loss of the abdominal wall reflex and anal reflex. He also had loss of the knee–ankle reflex, and quadriceps femoris, tibialis anterior muscle, gastrocnemius muscle, and first toe extensor and other toe extensor muscle strength of 0/5. Pathological signs in both lower limbs were not observed. Regrettably, nausea and vomiting occurred because of the excessive noise of the machine during the thoracic enhanced MRI. Therefore, we could not achieve an enhanced MRI scan. A laboratory examination showed the following: white blood cell count, 17.7 × 109/L; neutrophil count, 91.3%; calcitonin concentration, 0.20 ng/mL; C-reactive protein concentration, 311.1 mg/L, and erythrocyte sedimentation rate, 78 mm/hour. The patient met the criteria for sepsis on the basis of his clinical presentation, physical examination, and laboratory tests. Therefore, we performed emergency surgery while we attempted to deal with shock. Posterior T3–T6 laminectomy and decompression were conducted under general anesthesia and tracheal intubation. We confirmed the proper entry point of the thoracic pedicle screws and double-sided T3–T6 implant positioning pins by using a “C”-shaped arm image enhancement machine (Shanghai Siemens Co. Ltd., Shanghai, China). When one side of the lamina was incised, a large amount of yellow pus overflowed. Part of the pus was collected for bacterial culture and drug sensitivity tests, and the inflammatory tissue was sent for pathology. After complete removal of the T3–T6 lamina, a large amount of extradural pus was observed. We carefully and thoroughly removed the pus and exposed the dural sac (Figure 2). After flushing with a large amount of normal saline and iodophor solution, the positioning pins were removed, and the pedicle screws were correctly installed. After we confirmed the internal fixation device was ideal by “C”-shaped arm image enhancement using fluoroscopy, a negative pressure drainage ball was placed. The skin was then sutured layer by layer. Postoperatively, anti-infection therapy was administered (intravenous use of 1000 mg vancomycin every 8 hours, oral use of 0.6 g rifampicin once a day, and intravenous use of 0.5 g levofloxacin once a day). Bacterial culture results showed Staphylococcus aureus in the pus. Antibiotics were changed according to a drug susceptibility test. The postoperative pathological diagnosis (in the thoracic spinal canal) was inflammatory granulation tissue, inflammatory necrotic tissue, and striated muscle tissue (Figure 3).

Intraoperative images. (a) and (b) Epidural light yellow purulent secretions after intraoperative laminectomy (arrowheads) can be seen. (c) A large amount of pus attached to the ligamentum flavum (arrowheads) can be seen and (d) The dura mater was relieved of compression after the pus was removed.
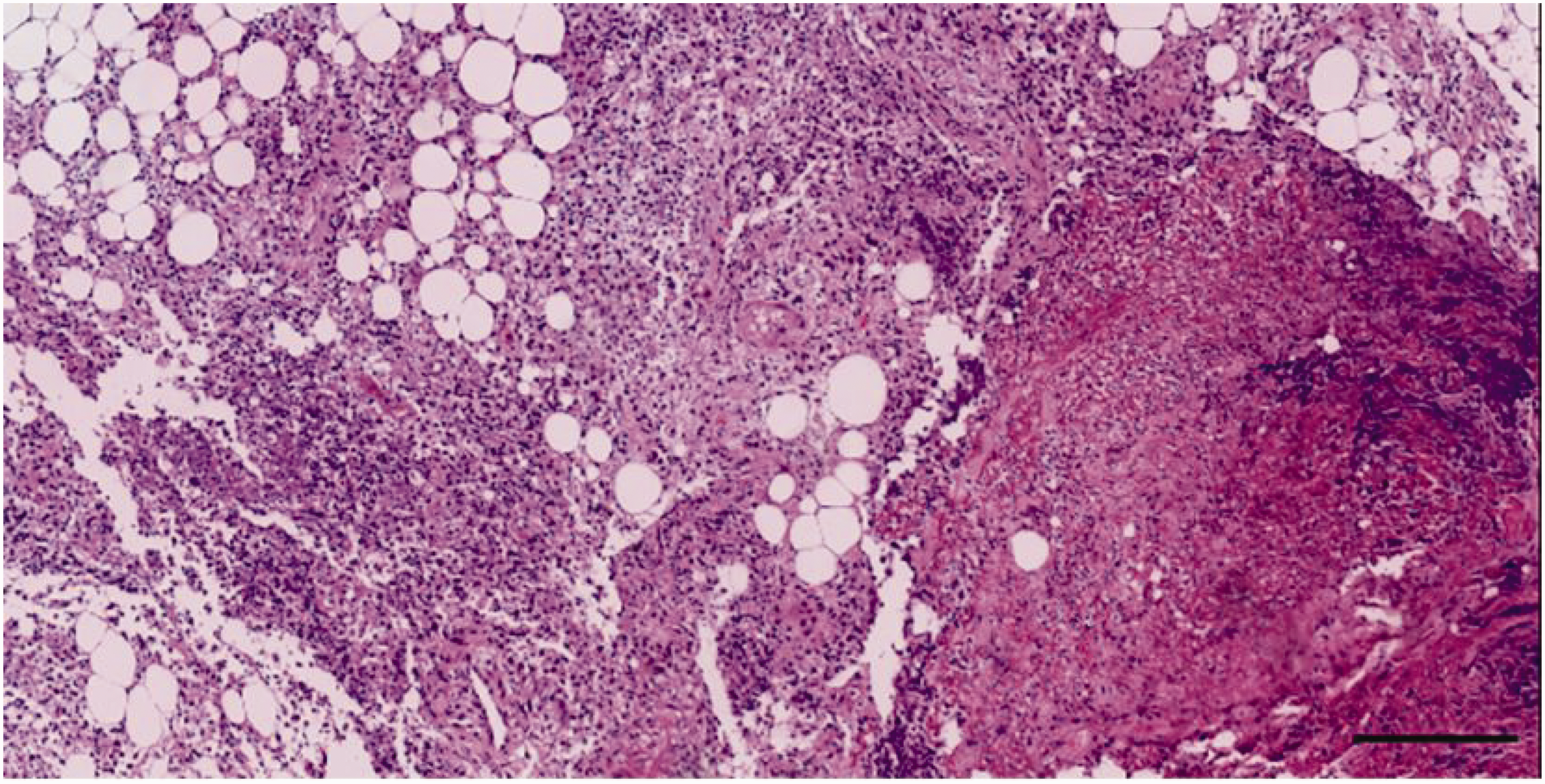
Paravertebral necrosis muscle tissue. Scale: 100 µm, hematoxylin–eosin staining.
On the first day after the operation, the patient’s left lower limb quadriceps muscle strength was 3/5, tibialis anterior muscle strength was 3/5, gastrocnemius muscle strength was 2/5, first toe extensor muscle strength was 1/5, and other toe extensor muscle strength was 0/5. The right lower limb quadriceps muscle strength was 3/5, tibialis anterior muscle strength was 3/5, gastrocnemius muscle strength was 2/5, first toe extensor muscle strength was 2/5, and other extensor toes muscle strength was 1/5. There was sensation in both of his lower limbs. The urinary catheter was removed at the third day after surgery. One week after the operation, both lower limbs had improved, and only the left back was numb. The patient could walk to the toilet with the protection of a thoracic brace. The drainage tube was removed 1 week after the operation. Inflammatory indicators were as follows: white blood cell count, 10.7 × 109/L; neutrophil count, 65%; C-reactive protein concentration, 90 mg/L, and erythrocyte sedimentation rate, 43 mm/hour. He was then discharged with rifampicin + levofloxacin tablets + linezolid tablets. The patient was then transferred to a rehabilitation hospital. Four weeks after the operation, the patient’s lower limbs had completely recovered, and his muscle strength was 5/5 (Figures 4 and 5).

Recovery time and situation of left lower limb muscle strength from preoperation to 2 weeks after the operation.

Recovery time and situation of right lower limb muscle strength from preoperation to 2 weeks after the operation.
Written informed consent for publication was obtained from the patient. This article was approved by the Ethical Review Committee of Sir Run Run Shaw Hospital, Zhejiang University School of Medicine.
Discussion
SEA is a rare spinal surgical emergency. Forty years previously, the clinical incidence rate of SEA accounted for 0.2 to 2.0/10,000 of hospitalized patients.4,5 However, the incidence rate of SEA has increased to 12.5/10,000 in recent years, 4 which may be related to intravenous drugs, diabetes, and increased immunosuppression.6,7 SEA lesions are more common in the thoracic and lumbar segments. 8 Men aged older than 60 years are more likely to have SEA, with a male to female ratio of 3:1.9,10 SEA is divided into (1) the acute type in which all lesions are pus, (2) the subacute type with coexistence of pus and granulation tissue, and (3) the chronic type with mainly granulation tissue. 11 Acute SEA has a rapid onset, and the spinal cord may be compressed and even paralyzed within a few days. Our patient developed paralysis of both lower limbs within 10 hours of acute onset. The most common pathogen in SEA is S. aureus, while Escherichia coli and pneumococcus are rare.12,13 Among them, methicillin-resistant S. aureus accounts for approximately 15%. 5 Pathogenic bacteria can be spread through blood-borne transmission, direct spread, and spinal surgery, among which blood-borne transmission is the most common. 14 In this case, a furuncle on the back occurred a few months previously, and the patient previously denied intravenous drug use, diabetes, and immunosuppressants. We considered that the patient had suffered from S. aureus by a direct spread. The bacterial culture taken during the operation showed S. aureus, which is consistent with the literature. The diagnosis of SEA is still difficult for physicians in clinics, especially when there is no typical triad of fever, back pain, and neurological deficits. However, these typical symptoms of SEA rarely occur at the same time. 15 In this case, the patient had back pain 7 days previously and did not visit a doctor. When fever and weakness of the lower limbs occurred, an MRI scan showed abnormal signals in the thoracic spinal canal and compression of the spinal cord. The diagnosis of SEA can be further confirmed by computed tomography or MRI. Although computed tomography and MRI can show space-occupying lesions in the spinal canal, computed tomography cannot clarify their nature. 10 MRI can show the location and extent of the abscess better than computed tomography. Pus based on T1-weighted imaging is a low signal. Moderate signals can also be present when the pus is thin. The pus based on T2-weighted imaging shows a high signal, and there is no radiation based on MRI.16,17 If the patient’s condition permits, an enhanced MRI scan should be performed. An enhanced scan can clearly show the location of the lesion, the local space-occupying effect, and the involvement of the spine and surrounding tissues. The wall of the abscess shows a ring-shaped enhancement during an enhanced scan. 17 In this case, T1-weighted images of thoracic MRI showed an equal signal, and the T2-weighted image showed a high signal. These imaging examinations combined with the physical and laboratory examinations suggested SEA. Our diagnosis was verified when the lamina was cut during the operation.
Early detection, early diagnosis, and early surgery are critical for thoracic epidural abscess. Incorrect or untimely treatment may lead to the permanent loss of nerve function. Studies have shown that when surgery for SEA is conducted within 2 hours, the postoperative recovery is satisfactory. However, when surgery occurs after 48 hours of the diagnosis, gaining normal postoperative nerve function is difficult.18,19 At present, the main treatment of SEA is surgical decompression combined with effective antibiotics. 20 Our patient underwent emergency surgery 16 hours after the onset of paralysis of both lower limbs. Sensitive antibiotics were administered according to a drug sensitivity test after the operation. Four weeks later, the patient’s lower limb muscle strength was 5/5. Feeling in both of his lower limbs was completely recovered.
Conclusion
The early detection and diagnosis of SEA, early surgery to relieve compression of the abscess on the spinal cord, and effective antibiotic intervention are important for treating SEA, which causes paralysis of the lower limbs.
Footnotes
Acknowledgements
The authors thank the patient for his informed consent and cooperation, and Dr. Jiahua Xu for the histopathological analysis.
Data availability statement
All data in this article are available.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
