Abstract
To date, there is no clinical scoring system or laboratory indicator that can rule out cerebral venous thrombosis (CVT) or provide diagnostic proof for evaluating post-treatment thrombosis recanalization during follow-up. We therefore explored an imaging method for quantitative assessment of CVT and assessed thrombotic changes during follow-up. A patient presented with severe posterior occipital distension extending to the top of the forehead and an elevated plasma D-dimer (DD2) level. Computed tomography and pre-contrast-enhanced magnetic resonance imaging revealed only a small amount of cerebral hemorrhage. Three-dimensional (3D) T1-weighted (T1W) BrainVIEW pre-contrast-enhanced magnetic resonance scanning showed subacute thrombosis in the venous sinus, and the post-contrast-enhanced scan combined with volume rendering reconstruction showed cerebral thrombosis of the venous sinus and allowed for measurement of the thrombus volume. On days 30 and 60 of post-treatment follow-up, post-contrast-enhanced scans showed progressive reduction of the thrombus volume as well as recanalization and fibrotic flow voids in the chronic thrombosis. 3D T1W BrainVIEW was helpful to observe the size of the thrombi and the situation of venous sinus recanalization during the follow-up after clinical treatment of CVT. This technique can reflect the imaging manifestations of CVT throughout the whole process to guide clinical treatment decisions.
Keywords
Background
Cerebral venous thrombosis (CVT) refers to thrombosis of cerebral veins or venous sinuses. CVT has various etiologies and results in obstruction of blood backflow or cerebrospinal fluid circulation, leading to cerebral hypertension and focal brain damage. The incidence of CVT in patients with cerebrovascular disease is approximately 1%, but the mortality rate is as high as 10% to 13%.1,2 Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-related CVT has attracted widespread attention in recent years.2–4 The main methods for diagnosing cerebral CVT are imaging techniques combined with detection of the plasma D-dimer level and cerebrospinal fluid manometry by lumbar puncture; however, diagnosing CVT in the emergency setting is still a significant challenge. 5 We herein describe a patient with cerebral CVT who underwent three-dimensional (3D) T1-weighted (T1W) BrainVIEW pre- and post-contrast-enhanced magnetic resonance scanning. The clinical application value of this imaging technique is discussed in combination with a literature review.
Case presentation
Clinical data
The reporting of this study conforms to the CARE guidelines. 6 A man in his early 50s presented with a 6-day history of aggravated pain in the posterior occipital region that had gradually expanded from the posterior occipital region to the top of the forehead. One day before admission, he had developed diplopia and right limb weakness. The patient denied a history of coronary heart disease, hyperlipidemia, hypertension, and diseases; he also denied a history of SARS-CoV-2 infection. He had no special medical history or relevant cerebrovascular disease risk factors, and he had not taken any special medications before the onset of illness. He had received the SARS-CoV-2 vaccine 6 months before admission.
Specialist examinations revealed the following: normal consciousness, bilaterally centered eyeballs, slightly limited left eye abduction, and papilledema in both eyes. The muscle strength of the right extremity was grade 5− (i.e., active movement against strong resistance over the feasible range of motion, but distinctly weaker than the contralateral side), and that of the left extremity was grade 5; the muscle tone was normal. The bilateral tendon reflexes were active. There was no obvious abnormality in the patient’s sense of depth and lightness of the whole body. No pathological reflexes were elicited. Laboratory tests showed the following results: plasma D-dimer (DD2) level of 1.25 mg/L, antithrombin III activity of 82.10%, prothrombin activity of 84.60%, and international normalized ratio of 0.98. Cerebrospinal fluid examination showed increased pressure at 420 mmH2O. Routine and biochemical indicators were normal. On the day of admission, pre-contrast-enhanced head computed tomography (CT) revealed a small amount of cerebral hemorrhage in the bilateral frontal lobes. On the third day after admission, pre-contrast-enhanced head magnetic resonance imaging (MRI) showed bilateral frontal lobe hemorrhage and other lesions, susceptibility-weighted imaging (SWI) and diffusion-weighted imaging (DWI) revealed bilateral frontal lobe venous malformation with cerebral hemorrhage, and magnetic resonance venography (MRV) showed the superior sagittal sinus and inferior sagittal sinus but not the right transverse sinus, suggesting the possibility of thrombosis (Figure 1). On the seventh day after admission, digital subtraction angiography (DSA) revealed thrombosis in the superior sagittal sinus, inferior sagittal sinus, right transverse sinus, and sigmoid sinus. Further imaging of the posterior segment of the superior sagittal sinus showed that drainage of the left transverse sinus and sigmoid sinus was unobstructed, the cortical veins and deep veins were poorly visualized, the right scalp veins and emissary veins were significantly dilated, and the arteriovenous circulation time was prolonged (<10 s) (Figure 1).
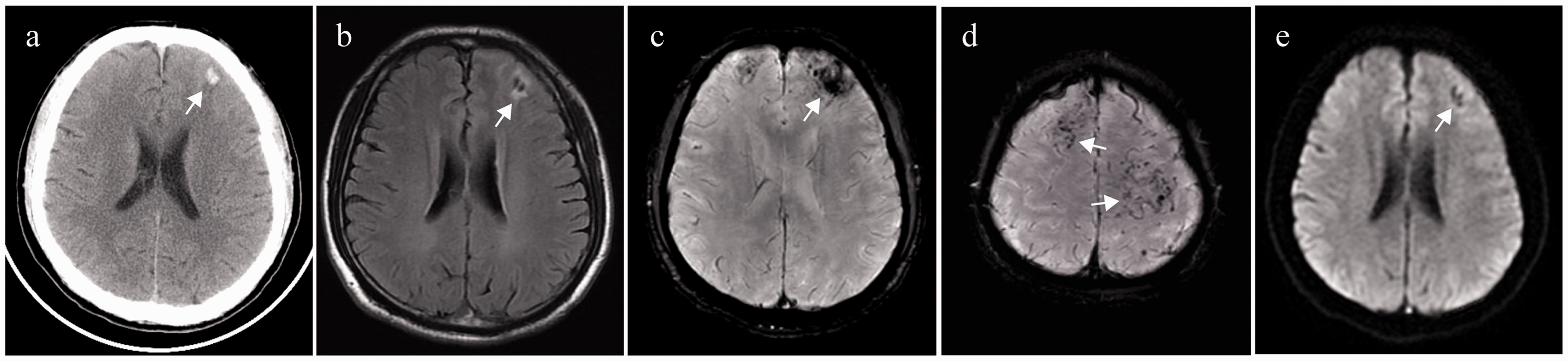
(a) CT scan shows a small amount of cerebral hemorrhage in the left frontal lobe (indicated by arrows). (b) T2W FLAIR shows a patchy short T2 signal in the left frontal lobe surrounded by a patchy long T2 signal (arrows). (c, d) SWI shows bilateral frontal venous malformations with cerebral hemorrhage (arrows) and (e) DWI shows thickened vermiform, small speckle, and nodular hypointense shadows in the gray and white matter areas of the bilateral frontal lobes (arrows).
Based on the above-described findings, the patient’s diagnoses were (1) CVT (thrombosis in the superior sagittal sinus, inferior sagittal sinus, torcular Herophili, right transverse sinus, and sigmoid sinus) and (2) intracerebral hemorrhage (bilateral frontal lobe and bilateral centrum semiovale malformation).
Clinical treatment
In the early stage, we used an intravenous drip of mannitol to reduce the patient’s intracranial pressure and administered subcutaneous injection of low-molecular-weight heparin. However, only oral warfarin was administered for anticoagulation in the late stage. All the above treatments were administered with the patient’s consent.
Follow-up assessment by imaging methods
Inspection method
The 3D T1W BrainVIEW pre- and post-contrast-enhanced scan was performed using a Philips Ingenia 3.0 T magnetic resonance scanner. Using the head standard 15-channel phased array coil, the sagittal scanning range covered the whole brain. The scanning parameters were as follows: repetition time, 600 ms; echo time, 32 ms; field of view, 220 × 220 ×220 mm; acquisition mode, Cartesian; ACQ voxel MPS, 0.70 × 0.70 × 0.70 mm; Act slice gap, −0.35 mm; slices, 480; fat-suppression spectral presaturation with inversion recovery (SPIR); turbo spin echo (TSE) factor, 20; TSE-es, 8.1; refocusing control angle, 40°; 3D VIEW, Brain T1; and acquisition time, 09:56. Gadolinium-diethylenetriamine pentaacetic acid (Gd-DTPA) was intravenously administered at a dose of 0.1 mmol/kg for a total of 14 mL. The Gd-DTPA injection was performed via the median cubital vein at a flow rate of 2 mL/s, and scanning was started 5 minutes after the injection. After scanning, Philips IntelliSpace Portal (ver. 7.0.4.20175) was used for semiautomatic quantitative measurement of the thrombus volume, and volume rendering (VR) reconstruction of the thrombus was performed.
Results
The first 3D T1W BrainVIEW pre-contrast-enhanced magnetic resonance scan performed on the 23rd day of admission showed that the subacute thrombosis in the posterior segment of the superior sagittal sinus had a clear outline. However, extensive chronic thrombosis formation was not clearly depicted in the superior sagittal sinus, inferior sagittal sinus, torcular Herophili, right transverse sinus, and sigmoid sinus. The post-contrast-enhanced images showed that the shape and size of the thrombi were more clearly displayed at the above-mentioned venous sinuses, and very little blood flow in these sinuses could be observed. In addition, the thrombus volume was approximately 16.4 mL (Figure 2). Post-contrast-enhanced images of the thrombi were better visualized with VR reconstruction (Figure 3).

(a1, b1) The first 3D T1W BrainVIEW sagittal/axial pre-contrast-enhanced images show the iso-T1W signal of the anterior segment of the superior sagittal sinus, inferior sagittal sinus, right transverse sinus, right sigmoid sinus, straight sinus, and torcular Herophili. However, the contour is not clear, and a long strip of high signal and clear contour can be observed in the posterior segment of the superior sagittal sinus (arrow) (c1, d1) Sagittal/axial post-contrast-enhanced images show that the superior sagittal sinus, inferior sagittal sinus, sinus confluence, right transverse sinus, and right sigmoid sinus are enhanced with a high T1W signal intensity, and the thrombus is clearly visualized. The range is more defined, with a mixed streak-like low signal (arrow) within it. (e1) The measured thrombus volume is 16.4 mL. (a2) On the 3D T1W BrainVIEW sequence conducted after 30 days, sagittal pre-contrast-enhanced images show that the original high signal in the posterior segment of the superior sagittal sinus had changed to an iso-T1W signal (arrow). (b2) The transverse axial pre-contrast-enhanced images show other T1W signals in the right sigmoid sinus and transverse sinus, but the range is not clear. (c2, d2) The post-contrast-enhanced images show high T1W signals with precise contours, and (e2) the thrombus volume is slightly smaller than before, approximately 13.8 mL. The range of mixed cord-like hypointensity in the superior sagittal sinus had increased (arrow). (a3, b3) On the 3D T1W BrainVIEW sequence conducted after 60 days, the pre-contrast-enhanced images are roughly the same as those achieved 30 days later, in which (c3, d3) the post-contrast-enhanced images clearly show venous sinus prothrombosis. (e3) The measurement volume is approximately 12.0 mL, and (c3) the cord-like low signal range in the venous sinus is higher than that 30 days earlier (arrows).

(a, b) MRV images show the superior sagittal sinus and inferior sagittal sinus, but the right transverse sinus is not shown. (c–f) The DSA image shows partial occlusion of the superior sagittal sinus, inferior sagittal sinus, and right transverse sinus and (g, h) 3D T1W BrainVIEW post-contrast-enhanced VR reconstruction images show thrombus enhancement (triangular arrows), while the normal venous sinus is depicted as a black area (long arrows).
The second 3D T1W BrainVIEW inspection was performed 30 days later, and the scanning parameters and post-processing steps were the same as in the previous examination. Pre-contrast-enhanced imaging showed that the original high signal in the posterior segment of the superior sagittal sinus had turned into an iso-T1W signal, indicating that the thrombus in this segment had changed from subacute to chronic thrombosis. In the post-contrast-enhanced images, the outline of the thrombus was clearly displayed and the thrombus volume had decreased in the superior sagittal sinus, inferior sagittal sinus, torcular Herophili, and right transverse sinus (approximately 13.8 mL). Partial recanalization of the superior sagittal sinus was also evident (Figure 2).
The third 3D T1W BrainVIEW examination was conducted 60 days later, and the scanning parameters and post-processing steps were the same as in the previous examination. The pre-contrast-enhanced images were roughly the same as those obtained 30 days earlier. The post-contrast-enhanced scan clearly showed venous sinus thrombosis, and the cord-like low signal range in the venous sinus was greater than that 30 days earlier, indicating that the thrombus volume had progressively decreased. The thrombus volume was approximately 12.0 mL (Figure 2).
Discussion
Common causes of CVT include hypercoagulable states (acquired or hereditary), infections, autoimmune diseases, and tumors. There are various clinical manifestations of CVT, and if CVT is not diagnosed in a timely manner, serious consequences and even death may occur. 7 No clinical scoring system or laboratory index has been established to determine the occurrence of CVT, making the definitive diagnosis of CVT an important clinical challenge.8,9 Brain CT and MRI combined with MRV are commonly used to diagnose CVT. However, these imaging techniques have potential drawbacks such as anatomical variations, significant variabilities in thrombus signal intensity, and imaging artifacts. Although DSA is the gold standard for diagnosing CVT, it may cause complications with poor reproducibility, and complications can also occur during percutaneous puncture and catheter procedures. Moreover, DSA cannot distinguish different stages of thrombus evolution, making it difficult to evaluate recanalization after treatment. 9
Our patient’s head CT examination did not show the three typical direct signs of CVT: the dense triangle sign, empty triangle sign, and spinal cord sign.10,11 However, because of the elevated plasma DD2 level in the patient’s laboratory examination and because CVT could not be ruled out in combination with the clinical symptoms, we performed routine head MRI, DWI, SWI, MRV, and other examinations again after admission. Among these, the MRV scan showed the partial superior sagittal sinus, inferior sagittal sinus, right transverse sinus, and sigmoid sinus, suggesting thrombosis (MRV results are indirect imaging manifestations of CVT).12–15 MRV uses the blood flow signal in the venous lumen to indirectly reflect whether an abnormality is present (e.g., stenosis or occlusion) in the blood vessels. It cannot directly display the thrombus in the venous sinus lumen or the shape of the lumen. Moreover, MRV imaging relies on inflow effects and is sensitive to slow blood flow associated with anatomical variation, and it overlaps with a dysplastic or incomplete venous lumen. On imaging, both thrombosis and a dysplastic venous sinus can show blood flow interruption or sinus stenosis on MRV imaging; thus, there is a possibility of misdiagnosis of thrombosis. 9
The 3D TSE magnetic resonance sequence with a variable flip angle16–21 is an emerging MRI technology. The 3D T1W BrainVIEW sequence, an advanced 3D TSE black-blood technology produced by Philips MRI, has been improved to visualize the brain tissue structure. It uses optimized refocusing pulses and slice selection gradients to reduce the scan time by 20%, providing high-contrast, high-signal-to-noise images of gray–white matter and other tissues. The sequence also applies slight refocusing angle (40°) technology. We believe that the use of the 3D T1W BrainVIEW sequence is characterized by its ultralow (specific absorption rate) features. In the present case, the whole-body specific absorption rate was <0.4 W/kg, far lower than the international safety standard (International Electrotechnical Commission 2 W/kg). 22 This avoids thermal damage caused by high-field MRI scans, making the use of this sequence theoretically safer than conventional scan sequences for patients with low fever and pregnancy.
Although the 3D T1W BrainVIEW sequence has reportedly been used to observe arterial lesions,16,17 it has not been used to study CVT. Based on the experience of applying magnetic resonance black-blood thrombus imaging technology (a variable flip-angle 3D TSE sequence similar to 3D T1W BrainVIEW) to CVT MRI on Siemens 3.0 T MRI equipment,23–25 we used a 3D T1W BrainVIEW sequence to perform a whole-brain venous scan. In the examination of our patient with suspected CVT, we used the 3D T1W BrainVIEW sequence to perform the whole-brain venous scan. The imaging results confirmed that 3D T1W BrainVIEW had a promising “black-blood” effect in displaying the cerebral venous sinuses. The diagnosis of CVT was in agreement with the cerebral angiography findings; at the same time, the anatomical structures of the venous sinus, such as the sinus wall, arachnoid granules, and surrounding tissues, could also be clearly observed (Figure 2). The clinical history and imaging features can also be combined to distinguish subacute thrombosis from chronic thrombosis14,26,27 (Figure 2(a1) and (a2)). Although pre-contrast-enhanced imaging has advantages in diagnosing subacute thrombus, it still has some shortcomings in the accurate assessment of chronic thrombus size and thrombus recanalization.
In reviewing the examination results of this patient, we found that the chronic thrombosis showed an equal or slightly lower T1W signal on the pre-contrast-enhanced 3D T1W BrainVIEW sequence. A 3D T1W BrainVIEW post-contrast-enhanced magnetic resonance scan can reportedly overcome the underestimation of thrombus size in angiography. 17 After administration of the gadolinium agent for post-contrast-enhanced scanning, we found that the 3D T1W BrainVIEW sequence directly showed the boundary of the chronic thrombosis and could distinguish the enhanced images of vascular wall inflammation caused by the thrombosis. It was easier to accurately evaluate the thrombus volume through post-processing software, which was consistent with the thrombus range displayed by DSA (Figure 3(c)–(h)). We also found that the gadolinium agent, combined with the black-blood properties of the 3D T1W BrainVIEW sequences, showed clearer recanalization and fibrotic flow voids in the hypointense chronic thrombosis (Figure 2(c1)–(c3)). The reason for this finding may be that the red blood cells in the thrombus rupture over time, free diluted methemoglobin is formed, and the thrombus evolves into a high signal on T1W images over time. In the chronic phase of vascular recanalization, a flow void signal appears in the thrombus, and the T1 signal in the venous sinus is also significantly reduced. 21
During the treatment of CTV, the changes in the cerebral venous vessels and thrombi were dynamically evaluated, and the treatment plan was adjusted. We also performed enhanced examination with the 3D T1W BrainVIEW sequence at the 30-day and 60-day follow-ups and accurately measured the thrombus volume. We found that the thrombus had gradually decreased in size and that some vessels had recanalized, suggesting that the treatment was effective. Additionally, enhanced scanning with the 3D T1W BrainVIEW sequence combined with VR volume reconstruction not only displayed high-signal thrombi and low-signal normal sinus cavities but also provided a clearer spatial description. Our experience suggests that a 3D T1W BrainVIEW post-contrast-enhanced magnetic resonance scan is of great value in observing the thrombus size and venous sinus recalculation during follow-up after clinical treatment of CVT, and it is expected to be a reliable technique for monitoring thrombus changes.
The main limitation of this study is that we did not obtain images of the thrombus in the acute stage of CVT, and the diagnostic value of 3D T1W BrainVIEW enhanced MRI for acute venous sinus thrombosis remains to be further demonstrated. In addition, the accuracy of the clinical application of this imaging technique in the diagnosis and treatment of CVT requires observations of large samples.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231166277 - Supplemental material for Clinical application of three-dimensional T1-weighted BrainVIEW in magnetic resonance imaging of cerebral venous thrombosis: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605231166277 for Clinical application of three-dimensional T1-weighted BrainVIEW in magnetic resonance imaging of cerebral venous thrombosis: a case report and literature review by Cheng Yang, Huang Fang, Jun Hu, Mei Li, Chunxia Wei, Jinfei Miao and Wencai Huang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231166277 - Supplemental material for Clinical application of three-dimensional T1-weighted BrainVIEW in magnetic resonance imaging of cerebral venous thrombosis: a case report and literature review
Supplemental material, sj-pdf-2-imr-10.1177_03000605231166277 for Clinical application of three-dimensional T1-weighted BrainVIEW in magnetic resonance imaging of cerebral venous thrombosis: a case report and literature review by Cheng Yang, Huang Fang, Jun Hu, Mei Li, Chunxia Wei, Jinfei Miao and Wencai Huang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605231166277 - Supplemental material for Clinical application of three-dimensional T1-weighted BrainVIEW in magnetic resonance imaging of cerebral venous thrombosis: a case report and literature review
Supplemental material, sj-pdf-3-imr-10.1177_03000605231166277 for Clinical application of three-dimensional T1-weighted BrainVIEW in magnetic resonance imaging of cerebral venous thrombosis: a case report and literature review by Cheng Yang, Huang Fang, Jun Hu, Mei Li, Chunxia Wei, Jinfei Miao and Wencai Huang in Journal of International Medical Research
Footnotes
Author contributions
Conception and design: CY and WH. Administrative support: HF and WH. Provision of study materials or patients: CY and HF. Collection and assembly of data: CY, JH, ML, CW, and JM. Data analysis and interpretation: CY and HF. Manuscript writing: all authors. Final approval of manuscript: all authors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Cheng Yang reports the receipt of research support from Philips Medical Solutions. Zhen Zhao, Peng Sun, Ying Yu, and Bai Jun Wang are employees of Philips AG Healthcare (not authors of the present study) and provided guidance on the MRI technology used in this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics statement
This study was approved by the Medical Ethics Committee of the General Hospital of Central Theater Command (2022-015-01). Written informed consent for the publication of the patient’s clinical details and images was obtained from the patient’s guardians prior to collection of the data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
