Abstract
Surgical resection is rarely employed for the treatment of metastatic gastric cancer, especially in patients with adrenal metastases, which usually indicate advanced systemic dissemination. Few published case reports have thus described the use of adrenalectomy for adrenal metastases from gastric cancer. In addition, most primary gastric malignancies are gastric adenocarcinomas, and gastric large cell neuroendocrine carcinoma (GLCNEC) is less common and has a poor prognosis. We report the case of a 71-year-old man who was diagnosed with solitary adrenal metastases 10 months after radical resection for GLCNEC and who was treated by adrenalectomy. He was followed-up for 9 months after adrenalectomy, with no further evidence of disease at his last follow-up examination. This case indicates that elective surgical resection may be feasible, even in rare cases of GLCNEC metastases to the adrenal glands, provided that the patient meets certain criteria, including solitary, metachronous tumors less than 4 cm.
Keywords
Introduction
Common metastases after radical resection of gastric cancer include peritoneal dissemination and hematogenous metastases, with liver metastasis being the most common (48%), followed by peritoneal metastasis (32%). 1 The rich blood supply to the adrenal gland also makes it a common site for metastasis, with an incidence of adrenal metastasis from gastric cancer of 16% to 18% in autopsy reports; 2 however, because adrenal metastases are often indicative of hematogenous systemic disease, surgical excision is rarely used in patients with gastric cancer-related adrenal metastases. 3 As a result, few cases of adrenalectomy for gastric cancer metastases have been reported, mostly involving gastric adenocarcinomas. In contrast, gastric large cell neuroendocrine carcinoma (GLCNEC) is a very rare histopathological form of gastric cancer, usually associated with a highly aggressive nature and poor prognosis compared with gastric adenocarcinomas. 4 We report a rare case of an elderly man who developed solitary metachronous metastasis to the unilateral adrenal gland 8 months after undergoing gastrectomy for GLCNEC. He received laparoscopic adrenalectomy and was clear of metastatic recurrence 9 months after the procedure.
Case presentation
A man in his early 70 s presented with worsening postprandial epigastric pain with melena, as well as a 3-kg unintentional weight loss over 1 month. His epigastric pain continued to worsen, with more-frequent episodes of postprandial nausea. He denied fatigue, dysphagia, vomiting, hypertension, and diabetes mellitus. He was allergic to sulfonamides, but had no family history of cancer. He underwent abdominal computed tomography (CT), upper gastrointestinal tract endoscopy, and tumor marker screening. The main tumor was located by abdominal CT scan, which revealed an active focus in the stomach. Tumor abnormal protein (TAP) was 244.50 (normal range 0–121 µm2), carcinoembryonic antigen was 0.12 (normal range 0–5 µg/L), and carbohydrate antigen 19-9 was 5.23 (normal range 0–37 U/mL). Other indicators were within normal ranges, but the elevated TAP levels indicated a possible rapidly proliferating malignancy. The patient provided consent for treatment. Upper gastrointestinal tract endoscopy confirmed the presence of a mass measuring approximately 10 × 9 × 7 cm on the lesser curvature of the stomach, which was biopsied. Pathologic examination revealed nests and trabeculae of high-grade tumor cells with neuroendocrine differentiation within a desmoplastic stroma. The patient underwent total gastrectomy with an esophagojejunal Roux-en-Y anastomosis in January 2021. Tumor extravasation occurred intraoperatively, and a round-like lymph node with a diameter of about 5 cm was seen adjacent to the left gastric artery, which was closely adherent to the pancreas. Groups 7, 8a, 8p, 9, 11p, and 12a lymph nodes were removed intraoperatively. Pathological evaluation of the tumor and lymph nodes revealed diffuse synaptophysin (Syn) positivity and patchy chromogranin positivity (Figure 1). Immunohistochemistry indicated CDX-2 positivity and a Ki-67 proliferation index of 90%+, indicating a rapidly growing lesion (Table 1). The tumor was determined to be GLCNEC, with a few localized adenocarcinomas (<30%) and no lymph node malignancy. The patient recovered well and was discharged about 1 week after surgery, receiving cisplatin in combination with 5-fluorouracil chemotherapy.

Patient’s pathological histological tissue specimens. Histological specimen after resection for gastric cancer (hematoxylin and eosin (H&E) staining; original magnification ×100).
Immunohistochemical results.

Syn, synaptophysin; SSTR2, somatostatin receptor 2; NA, not available; Pt, partial.
The patient had regular follow-up abdominal CT and gastroscopy appointments at the outpatient clinic, but there were no symptoms or abnormal findings that suggested recurrence. However, an abdominal CT scan in November 2021 revealed a solitary right adrenal mass measuring 2.7 cm, with inhomogeneous enhancement (Figure 2), which was suspected to indicate disease relapse or an adrenal incidentaloma, with no abnormalities in the liver, kidneys, spleen or pancreas. Duodenal bulbitis was suspected based on the gastroscopy findings. The patient was admitted for further tests and underwent a chest CT scan, as well as hematology, biochemistry, and hormonal tests. The patient declined preoperative puncture biopsy and positron emission tomography (PET)/CT because of the low diagnostic value of adrenal biopsy, the absence of local or metastatic recurrence on chest CT, and the patient's financial reasons. Hormone testing for hypercortisolism and aldosteronism and to rule out pheochromocytoma (PCC) revealed a nonfunctional adrenal mass (Table 2). After obtaining sufficient clinical data, surgical resection was planned with the patient's informed consent, and he underwent right laparoscopic adrenalectomy under general anesthesia in the left side position in November 2021, during which an adrenal tumor was excised. The pathological results suggested that the tumor cells were poorly differentiated, partly arranged in nested sheets and partly located in mucin lakes (Figure 3a). The immunohistochemical findings in line with gastric cancer were positive for Syn (Figure 3b) and partially positive for AE1/AE3, vimentin, and CD56 (Figure 3c), and the Ki-67 proliferation index was 70%+ (Table 1), all consistent with large cell neuroendocrine carcinoma (LCNEC). The adrenal serological results showed significantly decreased adrenocorticotropic hormone levels (Table 2) indicating that the patient had developed adrenal insufficiency following adrenalectomy, and he therefore received routine hydrocortisone replacement therapy.

Follow-up computed tomography scan of the patient in November 2021 showing a 2.7-cm right adrenal mass (arrow).
Hormone test results.

ACTH, adrenocorticotropic hormone; NMN, normetadrenaline; VMA, vanillylmandelic acid.
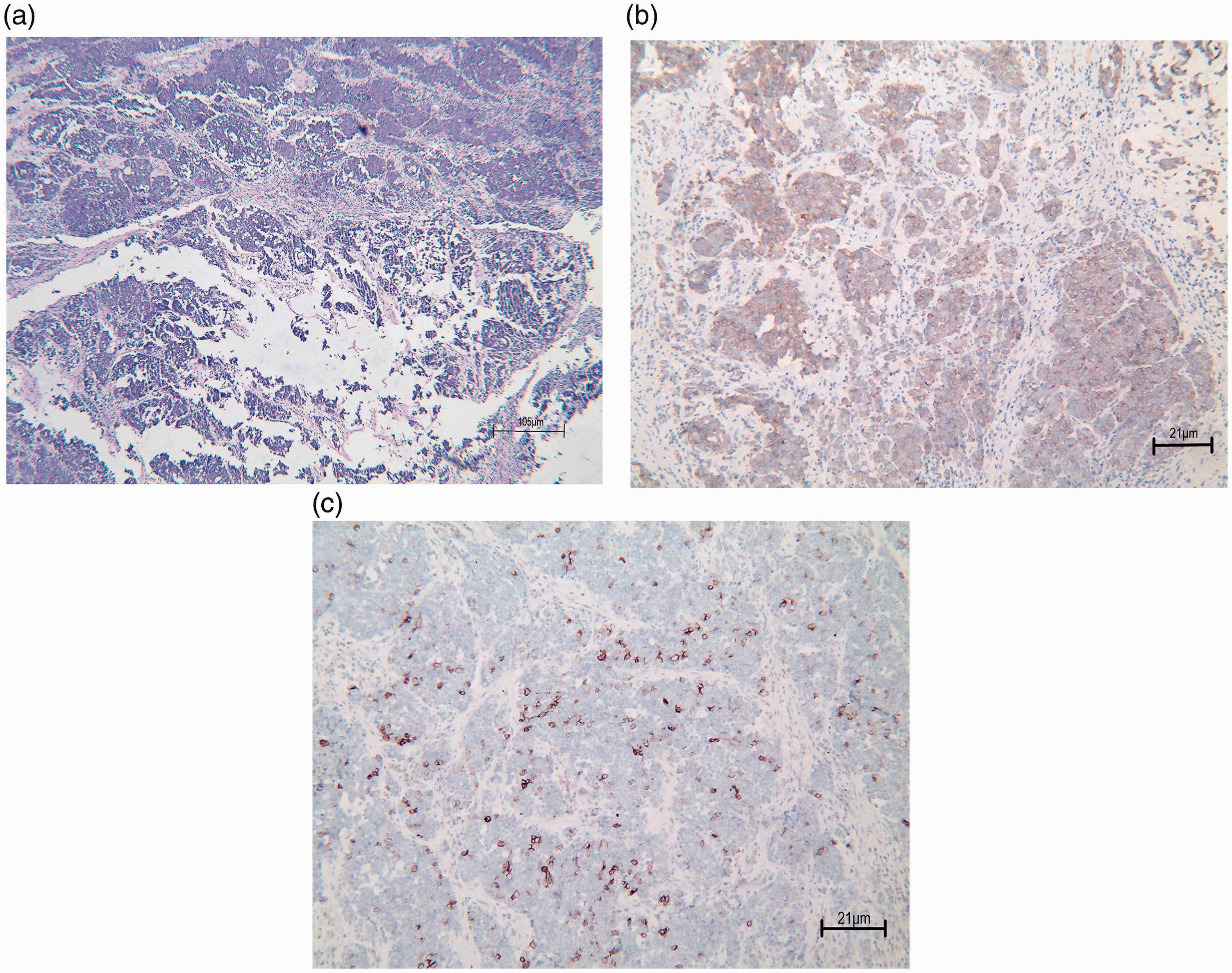
Adrenal pathology determined by histology and immunohistochemistry. (a) Histological specimen after resection of adrenal tumors (H&E staining; original magnification × 40). (b) Immunohistochemical evaluation of post-adrenalectomy specimens showed synaptophysin positivity (H&E focal staining; original magnification ×100) and (c) Immunohistochemical evaluation of post-adrenalectomy specimens showed CD56 positivity (H&E focal staining; original magnification × 100).
The patient was safely discharged 1 week following surgery. He continued to be followed up as an outpatient, and abdominal CT in August 2022 revealed no new metastases or tumor recurrence. We will continue to monitor the patient because of the poor prognosis of LCNEC; however, the patient remained alive for at least 9 months after resection of the right adrenal metastasis.
The reporting of this study conforms to CARE guidelines 5 and all patient details have been de-identified. The study was approved by the ethics committee of the Affiliated Hospital of Shanxi Medical University, and all procedures involving human participants were in accordance with the Declaration of Helsinki (as revised in 2013). Informed consent was obtained from the patient for the anonymous use of the clinical, imaging, and histologic data for publication. The patient’s son gave written informed consent for the publication of this report.
Discussion
Lung cancer is the most common primary tumor to metastasize to the adrenal glands, followed by kidney cancer, breast cancer, colon cancer, and melanoma. 6 Autopsy data show that adrenal metastasis also occurs in 16% to 18% of patients with gastric cancer, with the hematologic or lymphatic pathway being the most common route; however, it is usually detected as part of multi-organ metastases, with solitary adrenal metastases being relatively uncommon. 7 This suggests that adrenal metastasis in these patients represents part of advanced systemic spread with a poor prognosis, and is thus usually not suitable for surgical treatment; surgical resection of adrenal metastases from stomach cancer is therefore uncommon. Gastric neuroendocrine carcinoma is an extremely aggressive malignancy with a substantially worse prognosis than gastric adenocarcinoma, which has already spread to lymph nodes or distant organs at diagnosis. 8 Metastases discovered less than 6 months following the primary tumor's diagnosis are referred to as synchronous metastases, while those discovered more than 6 months after are referred to as metachronous metastases. 9 The current patient had a unique clinical path; he was diagnosed with GLCNEC but acquired solitary metachronous metastases, which is a rare occurrence in patients with metastatic gastric cancer. Although adrenal metastases of stomach cancer are not generally removed, the procedure has been carried out in patients with primary lung cancer or primary renal cell carcinoma.10,11 Patients with adrenal metastases who underwent surgery had better survival than those who received non-surgical treatment, particularly in patients with a disease-free interval (DFI) of more than 6 months and solitary metastases. 12 Hatano et al. concluded that adrenal metastases with a diameter of less than 4 cm were good candidates for surgical resection 13 ; however, few metastases satisfy these criteria and prospective randomized trials are lacking. Syn metastases or a short DFI should thus not be used as exclusion criteria for surgical treatment in patients who might achieve long-term disease-free survival.
Imaging is reliable for differentiating between benign and malignant adrenal masses; however, the differential diagnosis of adrenal metastases from the primary tumor remains challenging because adrenal metastases are usually functionally and anatomically asymptomatic. 14 Preoperative hormonal testing is required to rule out other adrenal diseases, particularly PCC, and one study concluded that solitary adrenal masses in patients with a history of cancer also had a 24% chance of being a PCC, and carried a risk of hypertensive crisis. 15 Fine needle aspiration biopsy is associated with risks of tumor cell metastasis and puncture hemorrhage, which can cause adhesions between the adrenal gland and surrounding tissue, and should thus only be used for adrenal metastases that are not surgically treatable. 12 Although PET/CT is useful for identifying adrenal metastases, 14 the current patient declined this option prior to surgery because of cost concerns. For individuals with probable adrenal metastases, we recommend hormone screening and PET/CT scans. Augmented radiological uptake combined with normal hormone examination results suggests metastatic lesions and needle biopsy should thus only be considered in extreme circumstances.
Most previously published pathological results of adrenal metastases of gastric cancer have involved adenocarcinomas, with only one case of signet ring cell carcinoma (Table 3). Adrenal metastases caused by GLCNEC are poorly understood and the therapeutic options are limited. GLCNEC differs from gastric adenocarcinoma morphologically, having bigger cells with more cytoplasm and chromatin, a high mitotic count (>10/2 mm2), tumor necrosis, and positive immunostaining for at least one neuroendocrine marker (chromogranin, Syn, or CD56). 21 Although these criteria are well-established, it might still be difficult to distinguish LCNEC from poorly differentiated adenocarcinoma in practice. Because LCNEC can arise from adenocarcinoma, it usually contains an adenocarcinoma component and thus has a high risk of being misdiagnosed as adenocarcinoma, which can hinder the pathological diagnosis. 22 The pathology in the current patient accordingly revealed an adenocarcinoma component, highlighting the need to understand the morphological and pathological characteristics of GLCNEC. Our patient had a solitary, metachronous (8 months after gastrectomy) tumor less than 4 cm in diameter and underwent successful radical resection. We intend to examine the patient on a regular basis because of the rarity of this instance.
Cases of postoperative adrenal metastasis from gastric cancer reported in the literature.
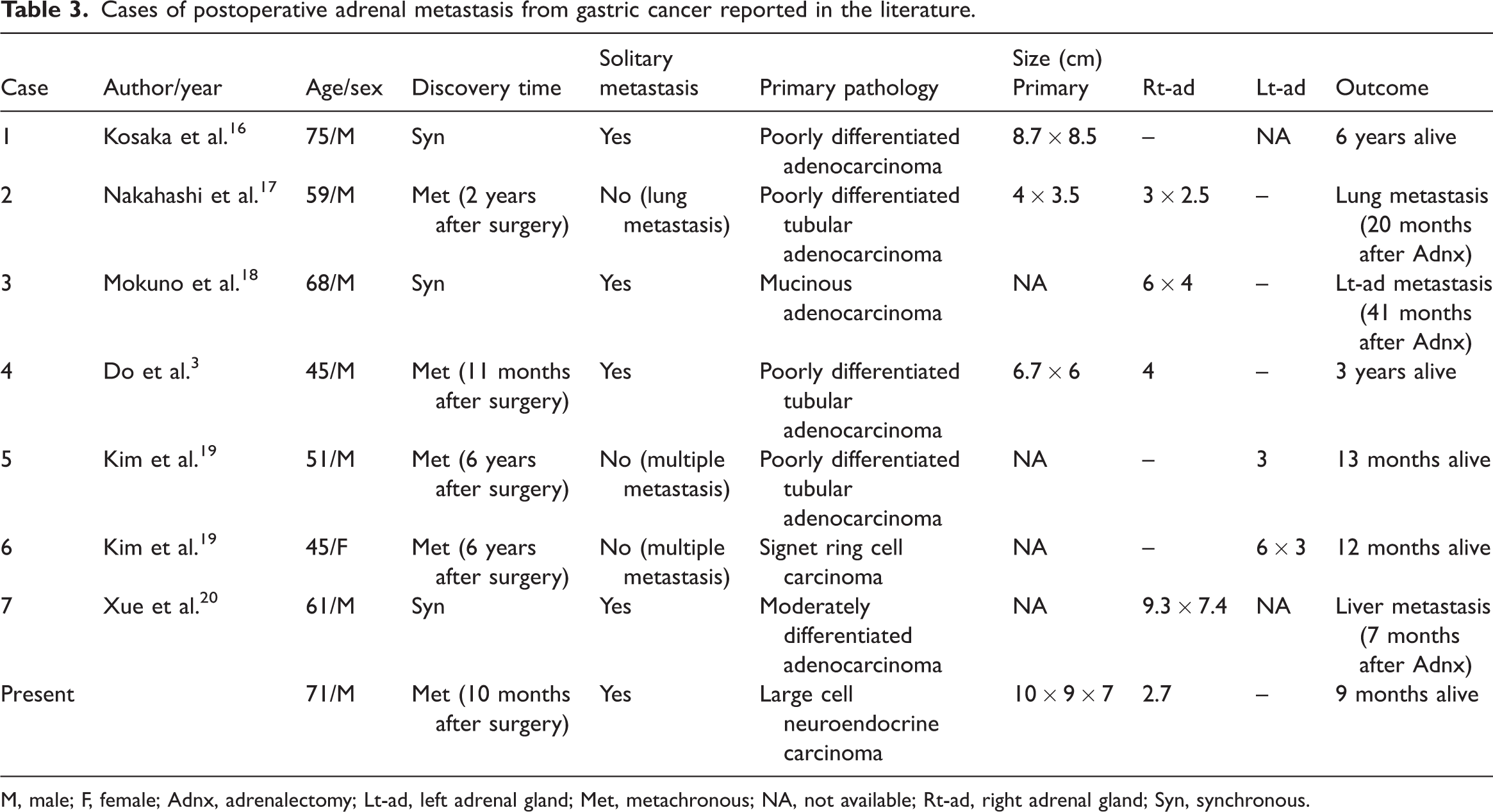
M, male; F, female; Adnx, adrenalectomy; Lt-ad, left adrenal gland; Met, metachronous; NA, not available; Rt-ad, right adrenal gland; Syn, synchronous.
The absence of local invasion of neighboring structures and a DFI of more than 6 months after the original tumor diagnosis suggests excellent tumor biology and a better prognosis after adrenal metastasectomy. 2 The size of the adrenal metastasis (4 cm) and the initial tumor type were also predictive factors for the postoperative outcome. 23 Goujon et al. examined prognostic markers for adrenal metastases after adrenalectomy in 106 patients, comprising the largest sample size to date, and found that a longer DFI and initial tumor's renal origin may prevent recurrence and improve prognosis. 24 However, despite the large sample size, none of the primary tumors were of gastric origin, and further studies and cases are therefore needed to examine potential treatment options and prognosis.
In conclusion, GLCNEC metastasis to the adrenal gland is extremely rare and can present challenges to the clinician in terms of surgical therapy and preoperative diagnosis. However, radiological and hormonal testing can aid the detection of adrenal metastases. Although GLCNEC is more aggressive than gastric adenocarcinoma and has a worse prognosis, certain patients may still benefit from surgical treatment, particularly those with metastases less than 4 cm and solitary metachronous metastases, giving surgeons more options for treating refractory patients.
Footnotes
Author contributions
WQ made contributions to the acquisition of the patient’s history and images, and wrote the manuscript. JS performed the treatment, and reviewed the manuscript to give clinical opinions. SY, ZC, and HC reviewed the manuscript and proposed changes. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work were appropriately investigated and resolved. This study was approved by the ethics committee of the Affiliated Hospital of Shanxi Medical University, and informed consent was obtained from the patient for the anonymous use of the clinical, imaging, and histologic data for publication. All procedures performed in this study involving human participants were in accordance with the Declaration of Helsinki (as revised in 2013). The patient’s son gave written informed consent for the publication of this report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shanxi Province Science and Technology Fund (Award No. 202203021211072), which provided financial support for the manuscript revision and data collection.
