Abstract
Skin and soft tissue diffusion metastasis (also known as occult cancer) is rare in renal cell carcinoma (RCC). Here, we report an extremely rare case of a 67-year-old male patient with occult primary RCC who developed metastases to the gums, skin, and diffuse soft tissue. The primary renal lesion was missed by computed tomography (CT), ultrasound, and 18F-fluorodeoxyglucose positron emission tomography (PET)/CT, and the diagnosis was confirmed by biopsy of gums and subcutaneous nodules. Subsequent enhanced CT revealed a lesion in the left kidney. The patient had progression-free survival of 16 months after treatment with axitinib and pembrolizumab. Pseudoprogression and tumor heterogeneity pose major challenges in the evaluation of immunotherapy. PET/CT is indispensable especially for cases with multiple metastases, widespread distribution of lesions, and major heterogeneity. In this case, the total lesion glycolysis was calculated by PET/CT and was used to evaluate systemic tumor load before and after immunotherapy, which was calculated as the product of the metabolic tumor volume and the mean standardized uptake value of the target lesion, which increased the accuracy of assessing diffuse lesions. Total lesion glycolysis can be used as a new method to quantitatively evaluate the efficacy of immunotherapy.
Keywords
Introduction
Renal cell carcinoma (RCC) is the most common malignant tumor of the kidney and originates from the proximal tubule. Clear cell RCC is the major pathological subtype, with other pathological subtypes making approximately 12% of diagnoses.1–3 With the rapid development of tumor immunology, immune checkpoint inhibitors (ICIs) have been approved for the treatment of various malignant tumors. The Keynote-426 trial confirmed that axitinib plus pembrolizumab is superior to sunitinib monotherapy in advanced RCC, and since that study, this immunotherapy combination has been considered standard-of-care.4–7 Lungs, bone, and the adrenal gland are routine metastatic sites of RCC, while soft tissue metastasis of RCC (including gum and skin) is rare. 8 Furthermore, there have been few reports of the use of 18F-fluorodeoxyglucose positron-emission tomography/computed tomography (18F-FDG PET/CT) to confirm the heterogeneity and efficacy in RCC patients receiving immunotherapy. Total lesion glycolysis (TLG) is a common PET/CT parameter that is a three-dimensional measurement index, which quantitatively reflects the overall metabolic burden of lesions. Most studies on TLG have been limited to lymphoma, 9 and this is the first time that TLG has been used to evaluate immunotherapy in kidney cancer. Herein, we report a rare case of occult RCC with extensive soft tissue metastasis, in which 18F-FDG PET/CT was used for disease diagnosis and evaluate treatment efficacy. To overcome the difficulties in such evaluations caused by diffuse metastases and tumor heterogeneity, we added TLG parameters to quantitatively evaluate systemic tumor burden on the basis of the immune PET Response Criteria in Solid Tumors (iPERCIST) guidelines.
Case presentation
A 67-year-old male patient presented with a progressively increasing mass on the palatal side of the right maxillary anterior teeth in August 2020. Subsequently, superficial masses were found in the neck, abdominal wall, back, and buttocks. Plain abdominal CT showed multiple nodules on the abdominal wall, buttocks, right psoas major, and soft tissue surrounding the left kidney, while no abnormalities were observed in the kidneys. Ultrasound imaging also showed no occupying lesions in bilateral kidneys. The clinical diagnosis was occult primary cancer (OPC). Subsequently, on 12 August 2020, 18F-FDG PET/CT was performed, which showed increased 18F-FDG uptake by soft tissues in the right palate, right adrenal gland, subcutaneous tissues, muscular nodules, multiple lymph nodes, and bones (Figure 2a). The patient was pathologically diagnosed with metastatic undifferentiated carcinoma by puncture biopsy of the chest wall mass. Immunohistochemistry results showed that Pax-8 was positive, which suggested primary renal undifferentiated cancer. Furthermore, contrast-enhanced abdominal CT showed an isodensity lesion in the left kidney that showed moderate enhancement after contrast administration (Figure 1). Finally, the patient was diagnosed as occult RCC with diffusion soft metastasis. On 26 August 2020, the patient was treated with intravenous pembrolizumab (200 mg, every 3 weeks) plus oral acitinib (5 mg, twice daily).
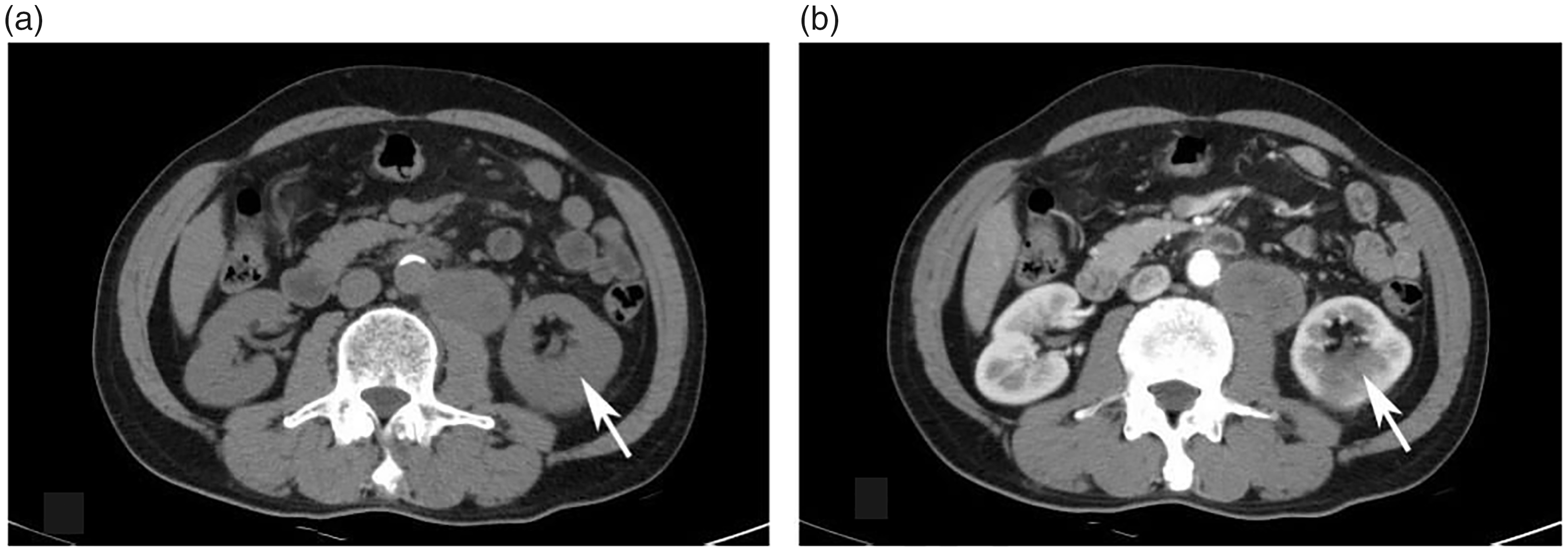
Contrast-enhanced abdominal computed tomography showing an isodensity lesion in the left kidney with moderate enhancement; (a) the white arrow indicates the iso-density shadow; the density is 35 Hounsfield units (HU) and (b) the white arrow indicates the space occupied by the left kidney, which is moderately enhanced after enhancement, with a density of 72 HU. The malignant tumor of the left kidney is suspicious.
On 3 March 2021, the patient underwent a second PET/CT, which showed partial response (PR). Most metastases were smaller with lower standard uptake values (SUVs), but some were larger, and there were even new metastatic lesions (Figure 2b). On 24 September 2021, the patient underwent a third PET/CT, which showed progressive disease (PD). Most metastases were larger with higher SUVs, while a few were smaller with lower SUVs ( Figures 2c, 3c, and 4c). Progress-free survival (PFS) from the first-line treatment was 16 months. Then second-line treatment of pembrolizumab plus pazolpa was initiated. On 31 March 2022, the patient underwent a fourth PET/CT, which showed PR. Most metastases were smaller with lower SUVs, while some were larger, and there were again some new metastases ( Figures 2d, 3d, and 4d).
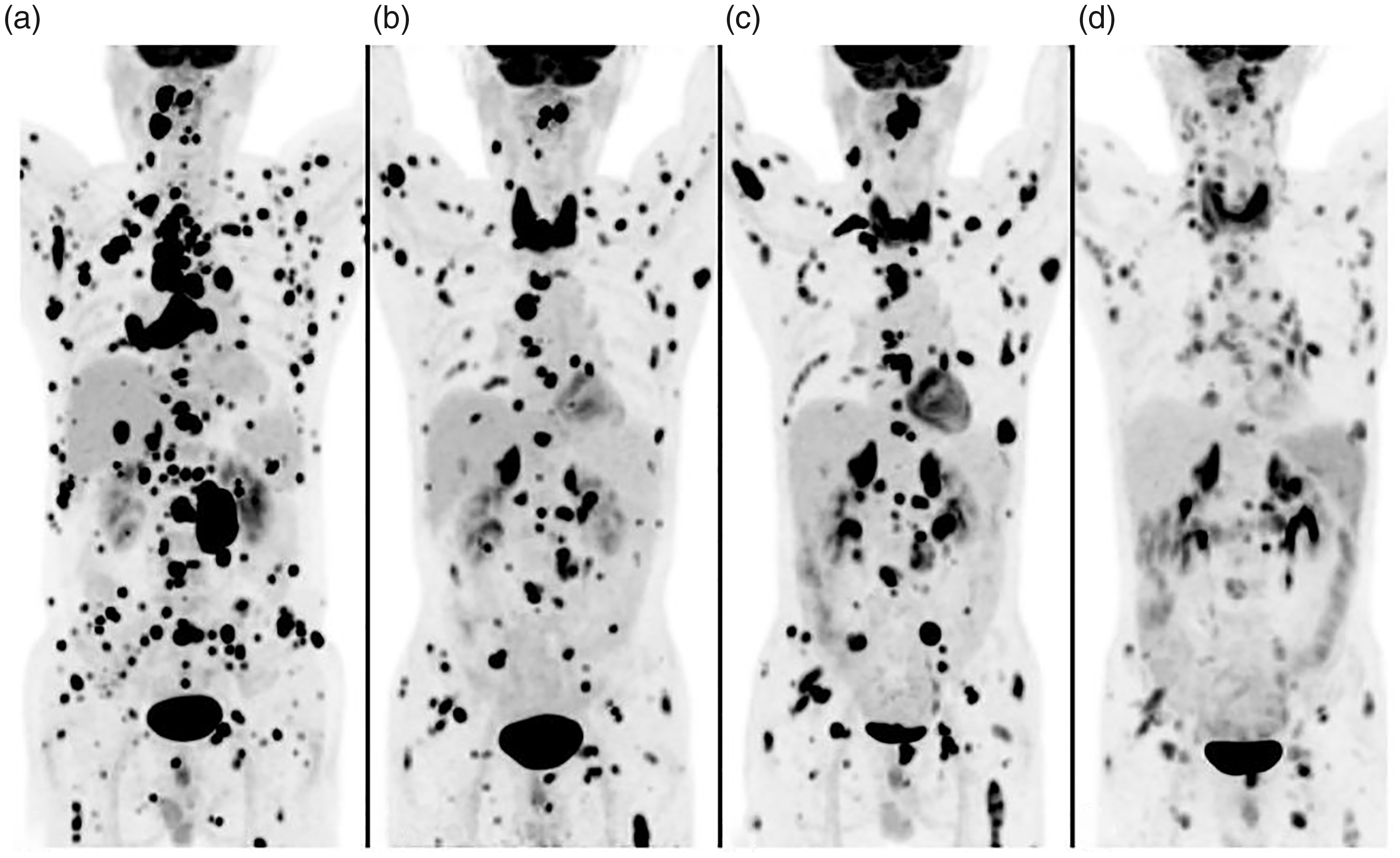
Evaluation of tumors by 18F-fluorodeoxyglucose (FDG) positron emission tomography/computed tomography; (a) there were multiple nodular and flaky lesions with increased FDG uptake throughout the body; (b) most lesions were smaller with the decreased standard uptake value, while some were larger, and there were even new lesions; (c) most lesions in the whole body were enlarged, accompanied by increased FDG uptake, while a few were still shrinking with reduced FDG uptake and (d) most lesions in the whole body shrank with decreased FDG uptake or even disappeared, while a few had grown with increased FDG uptake, and there were very few new lesions.
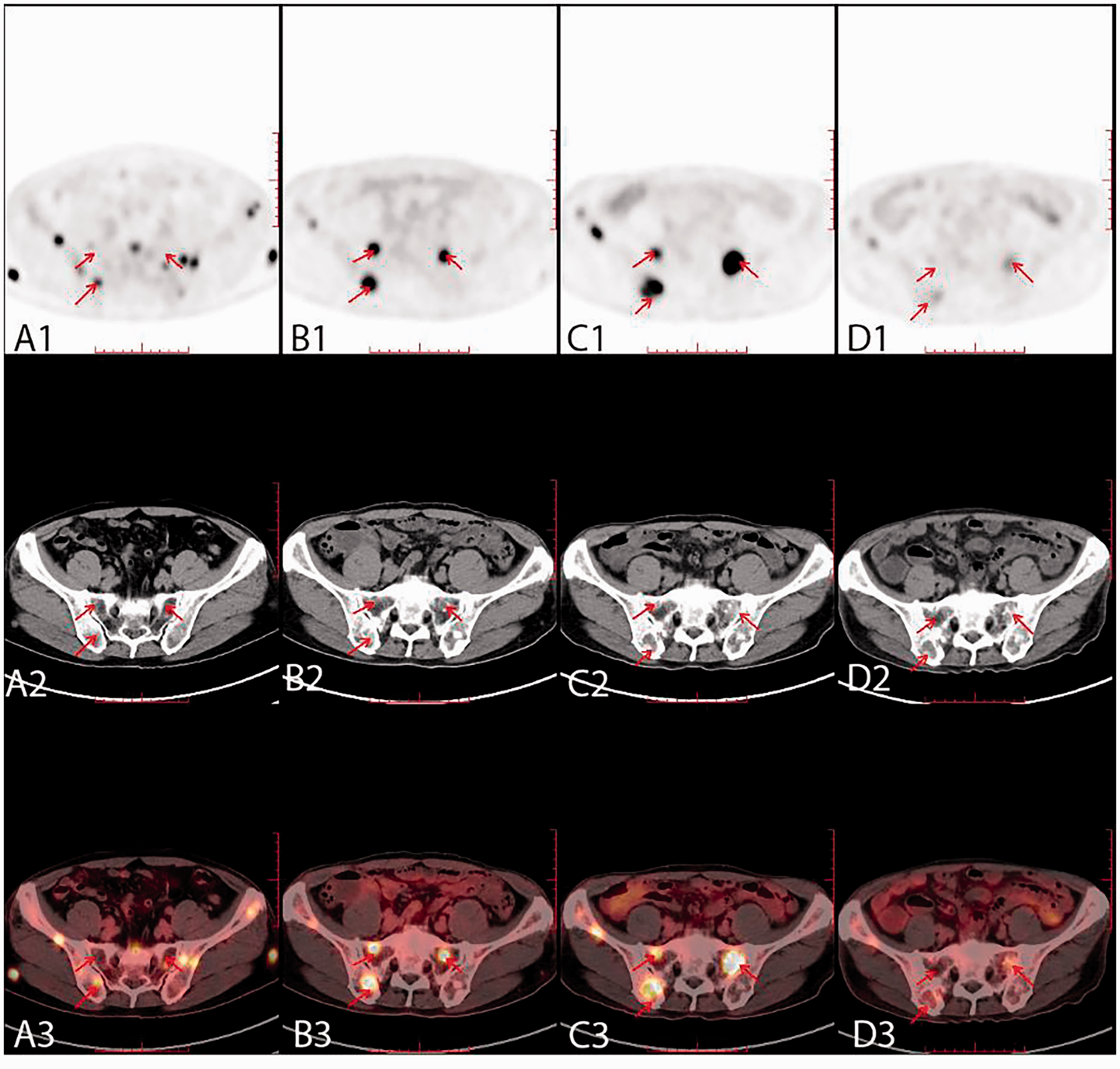
Comparisons of 18F-fluorodeoxyglucose (FDG) uptake; a1, multiple foci with increased FDG uptake; a2, Multiple foci with abnormal density; a3, multiple foci with abnormal density and increased FDG uptake; b1, FDG metabolism was higher than before; b2, the density of abnormal foci was greater than before; b3, the density of abnormal foci was greater and FDG uptake was increased; c1, FDG metabolism was higher than before; c2, the density of abnormal foci was greater than before; c3, the density of abnormal foci was greater and FDG uptake was increased; d1, decreased FDG uptake compared with before; d2, the density of abnormal foci was less than before; d3, the density of abnormal foci was less and FDG uptake was decreased.

Evaluations of 18F-fluorodeoxyglucose (FDG) uptake; a1, multiple lesions with increased FDG uptake; a2, multiple density abnormalities; a3, multiple density abnormalities and increased FDG uptake; b1, FDG uptake disappeared; b2, the density abnormality disappeared; b3, abnormal density and FDG metabolic focus disappeared; c1, no abnormal FDG uptake; c2, no foci with abnormal density; c3, no obvious abnormality was found on positron emission tomography/computed tomography (PET/CT); d1, no abnormal FDG uptake; d2, no foci with abnormal density; d3, no obvious abnormality was found on PET/CT.
Quantitative evaluation of tumor burden by TLG suggested that after treatment with pembrolizumab plus acitinib, tumor burden was lower than baseline. The third PET/CT examination indicated tumor progression, after which treatment was adjusted to pembrolizumab plus pazolpa. The fourth PET/CT examination showed decreased TLG values of the lesions, suggesting that second-line treatment was effective (Figure 5).

Total lesion glycolysis (TLG) values from 18F-fluorodeoxyglucose positron emission tomography/computed tomography scans performed on the indicated dates; 2020.08.12: TLG = 31011.5 (g); 2021.03.03: TLG = 12836.2 (g); 2021.09.24: TLG = 20030.6 (g); 2022.03.31: TLG = 7353.3 (g).
The patient provided written informed consent for all treatments, the PET/CT examinations to evaluate treatment efficacy and monitor tumor heterogeneity, and publication of this case report. The patient repeatedly expressed his gratitude for our care and treatments. The reporting of this study conforms to the CARE guidelines. 10
Discussion
Metastases to skin and soft tissues by RCC is relatively rare. This case showed diffusion soft tissue metastases as the primary symptom and was clinically diagnosed as OPC. The patient was eventually diagnosed with occult primary RCC by contrast-enhanced abdominal CT and biopsy. Systematic imaging evaluation is indispensable when the primary tumor is unclear. In this patient, the primary site was occult on plain CT scan because the left kidney lesion showed iso-density and the tumor did not protrude from the renal cortex. Therefore, when PET/CT shows multiple metastases throughout the body, but the primary site is unknown, it is important to further examine whether there is any disease in the kidneys.
18F-FDG PET/CT can accurately identify distant diffusion soft tissue metastases, which is useful for the evaluation of tumor burden. 18F-FDG PET/CT is indispensable for monitoring tumor heterogeneity. The PET/CT image from 3 March 2021 suggested local progression, but most metastases showed PR. The PET/CT image from 24 September 2021 indicated progression of most metastases, while a few showed PR. Overall, the patient's disease was progressive, and the treatment was changed to second-line pembrolizumab plus pazolpa. The fourth PET/CT on 31 March 2022 indicated that most metastases were smaller, while a few lesions were new and/or had advanced. Conventional Response Evaluation Criteria in Solid Tumors (RECIST), which primarily relies on tumor size and morphology, has been universally accepted as the standard response assessment criteria for conventional chemotherapies. However, RECIST cannot accurately assess overall efficacy in patients with metastases to more than two organs, more than five target lesions, diffuse systemic multiple metastases, or obvious tumor heterogeneity. The imaging manifestations in patients receiving immunotherapies appear to be distinct from those receiving conventional cytotoxic therapies. Thus, new patterns of response to ICIs have challenged traditional efficacy evaluation criteria. 11 Function imaging at the molecular level and anatomical imaging are integrated in PET/CT to visualize the biological process of tumors so that early changes in tumor metabolism can be recognized.12,13 18F-FDG is an analogue of glucose, which is the main carbon source for the synthesis of amino acids and nucleic acids. Therefore, 18F-FDG uptake intensity is closely related to the proliferation of living cells. Understandably, decreased 18F-FDG uptake can be observed when treatment is effective. 14 Under certain circumstances, RCC enters into dormancy when tyrosine kinase inhibitors and ICIs are administered. Hence, there would be no increase in tumor volume or new metastases. 15 Moreover, external morphological changes are not obvious due to liquefaction and necrosis inside the tumor when there is response to targeted therapy. 16 Compared with PET/CT imaging at baseline in this patient, most of the metastases were reduced to varying degrees despite a few new lesions appearing. Decreased maximum SUV (SUVmax) in a few metastases that did not change in size also indicated that the treatment was effective. Heterogeneity of RCC may partly explain such imaging features. On the basis of RECIST and non-specific clinical responses in immunotherapy, iPERCIST was proposed. According to iPERCIST, objective response rates were significantly higher than according to immune RECIST (iRECIST). Moreover, overall survival between responders and non-responders showed statistically significant differences (19.9 vs. 3.6 months), indicating potential superiority of iPERCIST. 17 There are operational difficulties to using iPERCIST for the evaluation of each individual metastatic lesion in patients with multiple metastases. However, immunotherapy is usually administered to patients with advanced tumors, at which time patients often exhibit widespread metastases involving multiple organs. Therefore, clinicians cannot accurately assess each metastatic lesion through local imaging and subjective judgments, let alone directly assess the overall tumor burden of patients. Hence, we propose that the overall evaluation of multiple metastases in immunotherapy is superior to evaluations of each individual metastatic lesion.
We propose a new and better immunotherapy evaluation method that utilizes the characteristics of whole-body PET/CT evaluation, as well as TLG parameters, which could be used to assess systemic tumor burden. By comparing TLG before and after immunotherapy, decreased TLG indicates decreased tumor burden, suggesting that immunotherapy was effective, while increased TLG indicates tumor progression. We look forward to further validation of large samples based on the results of this case study.
Conclusion
This is the first report of RCC with gingival metastasis. Plain CT and PET/CT have disadvantages in the diagnosis of primary RCC, and enhanced CT should be used when necessary. During immunotherapy for RCC, PET/CT can better evaluate response and monitor tumor heterogeneity in patients with diffuse multiple metastases, which maximizes benefit to the patient. TLG is more comprehensive for the assessment of diffuse soft tissue metastasis, and PET/CT is indispensable in such cases.
Footnotes
Author contributions
TJ and HJZ made contributions to the case analysis and were major contributors in writing the manuscript. MJD and JFF made important contributions to the conception and design of the research. KD and GHW were responsible for data collection and analysis. SSZ and CHL drafted the work. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics statement
This study was approved by the Ethics Committee of Affiliated Jinhua Hospital, Zhejiang University School of Medicine (Zhejiang, China).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
