Abstract
Objective
Increasing evidence from randomized controlled trials shows the anti-inflammatory and haemodynamic effects of levosimendan in advanced heart failure (AdHF), however, conflicting results have been reported in some studies. The aim of this study was to estimate the anti-inflammatory and haemodynamic effects of levosimendan on AdHF (registration number: INPLASY202250097).
Methods
The MEDLINE, PubMed, ClinicalTrials.com and Cochrane Library databases were systematically searched for studies published in English up to April 2019. Data were extracted from applicable articles. Meta-analyses were performed to assess interleukin (IL)-6, cardiac index, pulmonary artery pressure (PAP) and New York Heart Association (NYHA) functional class efficacy outcomes, following PRISMA 2020 guidelines.
Results
A total of 11 studies were included (211 patients who received levosimendan and 193 controls). Meta-analyses showed that the levosimendan group displayed significantly reduced IL-6 (standardized mean difference [SMD] −1.05; 95% confidence interval [CI] −1.44, −0.66; I
Conclusions
Levosimendan infusion was beneficial in patients with AdHF, displayed by anti-inflammatory and improved haemodynamic effects, and improved NYHA functional class.
Keywords
Introduction
Heart failure is a disorder of progressive functional decline characterized by limitation of function, worsening of symptoms and deterioration of quality of life, and is a relatively important public health problem due to its increasing incidence and poor prognosis. 1 Advanced heart failure (AdHF) is associated with high rates of morbidity and mortality. Health-related quality of life in patients with AdHF is substantially undermined due to reduced strength and capacity for physical activity.2,3 Symptom severity in AdHF leads to frequent heart failure-related hospitalization. 4 Moreover, the prognosis of patients with AdHF is relatively poor, with nearly 40% of patients with severe heart failure reported to die within a year of acute exacerbation. 5
In the treatment of AdHF caused by systolic dysfunction, intravenous positive inotropic agents play an important role in improving symptoms and haemodynamic abnormalities. 5 Levosimendan, a calcium-sensitizer with positive inotropic properties, increases myocardial contractility without promoting intracellular calcium accumulation, thereby increasing the effects of calcium on cardiac myofilaments during systole and improving contraction. 6 The clinical efficacy of levosimendan in patients with acute decompensated heart failure has been described in a previous meta-analysis by the present authors. 7
The beneficial effects of levosimendan on haemodynamics in the context of inflammation have also been reported, 8 and there is an increasing prevalence of studies that demonstrate the additional properties of levosimendan, such as anti-inflammatory effects during AdHF, endotoxemia or ischaemia reperfusion.9,10 However, small randomized controlled trials on the anti-inflammatory and haemodynamic effects of levosimendan in patients with heart failure have yielded inconsistent results. Therefore, the aim of the present meta-analysis of published trials was to assess the anti-inflammatory and haemodynamic effects of levosimendan in patients with AdHF.
Materials and methods
Search strategy
The MEDLINE, PubMed, ClinicalTrials.gov and Cochrane Library databases were searched for studies published in English language up to April 2019. The search terms and strategy comprised the following: (“levosimendan”) AND (“acute”) AND (“heart failure” OR “HF”). Additionally, the reference lists of review papers and every publication retrieved were manually searched for any additional published articles.
Selection criteria and exclusion criteria
For selection, studies were required to include the following: (1) patients with AdHF (New York Heart Association II−IV); (2) studies that included levosimendan; (3) baseline (pretreatment) and post-treatment data for levosimendan, with or without comparators (placebo or active treatment controls); and (4) extractible data for efficacy outcomes, such as: interleukin (IL)-6, cardiac index, pulmonary artery pressure (PAP) and New York Heart Association (NYHA) functional class. Exclusion criteria comprised: (1) animal experimental studies; (2) paediatric studies; (3) oral administration of levosimendan; (4) lack of required data; (5) unpublished studies; (6) patients with heart failure due to restrictive or hypertrophic cardiomyopathy, or use of immunosuppressive drugs; (7) reviews or case reports; and (8) no abstract available for initial screening.
Data extraction
The titles and abstracts of studies retrieved by the electronic search were independently screened by two researchers, and any disagreement was resolved by a third researcher. Articles that remained following title and abstract screening, and articles for which necessary information was unavailable in the titles and abstracts, underwent comprehensive full-text review. The following data were extracted from each study: the first author’s name, publication year, regimes for intervention and control groups (levosimendan versus active comparators or baseline pretreatment versus posttreatment), general condition of patients (NYHA functional class and left ventricular ejection fraction [LVEF]), number of patients in each group, and drug dose and duration of administration. Outcome data extracted for efficacy analysis mainly included: IL-6, cardiac index, PAP and NYHA functional class. Internal validity and publication bias were assessed by Cochrane Collection methods (Cochrane.org). Publication bias was assessed by visually inspecting funnel plots.
Statistical analyses
The meta-analyses of efficacy outcomes were performed using STATA software, version 12.0 (Stata Corporation, College Station, TX, USA). Outcomes from individual studies were extracted and pooled to calculate estimate risk ratios (RR) and corresponding 95% confidence intervals (CIs). Study heterogeneity was measured by the χ2-based Q test and I2. A fixed-effects model was used if I2 was ≤50% and P ≥ 0.1, which indicated no statistically significant heterogeneity. A primary analysis was conducted with a random-effects model if P < 0.1 or I2 > 50%, respectively, considered to indicate significant heterogeneity. The statistical significance of pooled estimates was assessed by Z-test, and P < 0.05 was considered to be statistically significant. 11 Publication bias was assessed by constructing a funnel plot of the effect RR of each study on the x-axis and the standard error of the variance of the log RR on the y-axis, and then by both Begg’s and Egger’s tests.11,12
Results
The process for selecting studies for inclusion in the meta-analysis is shown in Figure 1. The initial search resulted in 386 records, of which, 336 articles were excluded following removal of duplicates and screening of titles and abstracts. A further 39 studies were excluded following full-text review. Finally, 11 articles, involving 404 patients in total (211 who received levosimendan and 193 controls) were selected according to the inclusion/exclusion criteria in this meta-analysis (Figure 1). The characteristics of the 11 included studies are summarized in Table 1.13–23 In six of the studies, patients were administered a bolus dose of levosimendan followed by continuous infusion,13–18 while in five studies, patients were administered a continuous infusion of levosimendan without the bolus dose.19–23 Intravenous bolus doses ranged between 6 and 12 µg/kg, and continuous infusion doses ranged from 0.1 to 0.4 µg/kg/min. In some studies, posttreatment values were compared with baseline pretreatment values (designated ‘blank’ group; Table 1)

Flow diagram of the research strategy and selection of studies for the meta-analysis.
Characteristics of 11 studies that investigated the effects of levosimendan in patients with advanced heart failure.a
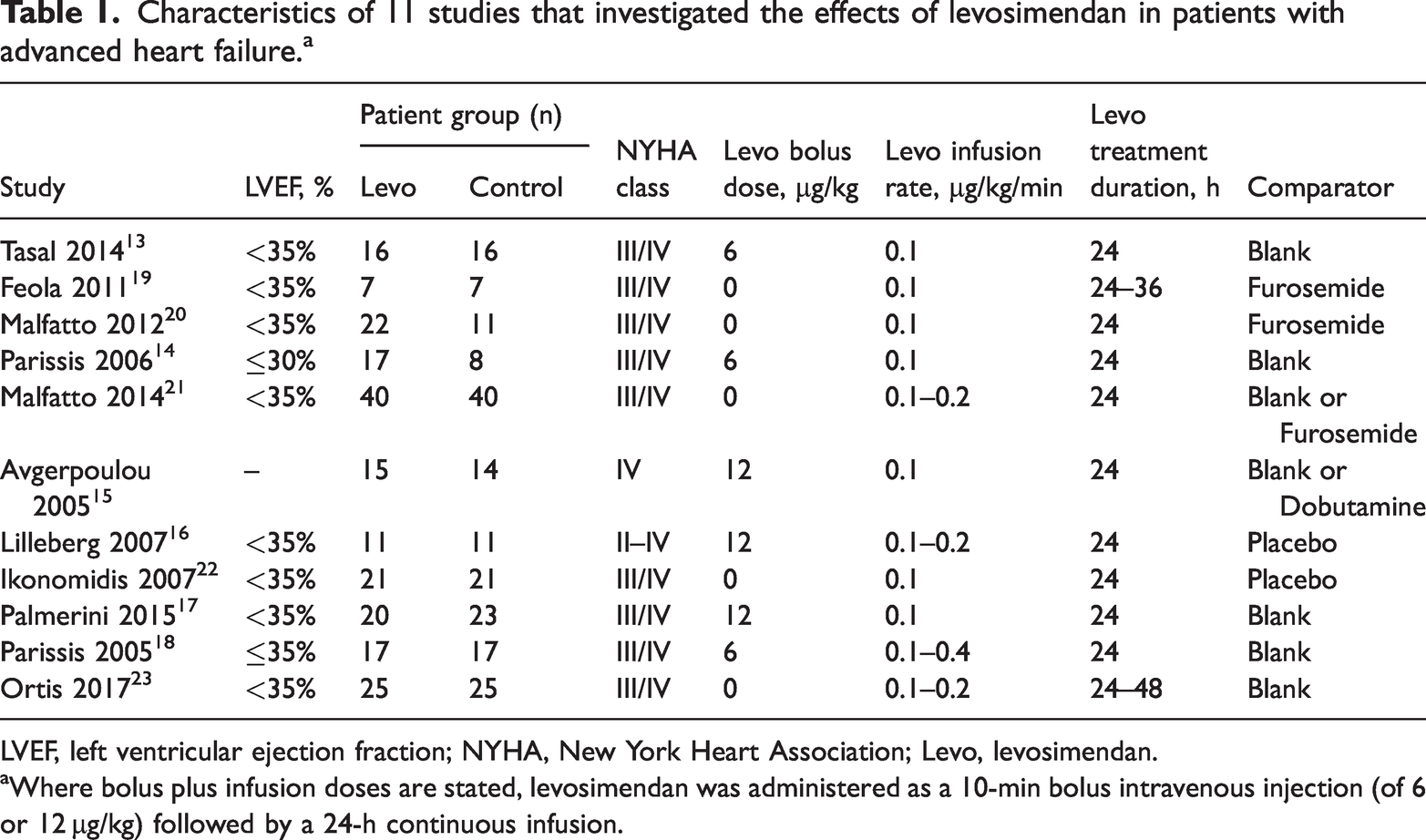
LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; Levo, levosimendan.
aWhere bolus plus infusion doses are stated, levosimendan was administered as a 10-min bolus intravenous injection (of 6 or 12 µg/kg) followed by a 24-h continuous infusion.
Interleukin-6 reduction
Five studies reported IL-6 data that were pooled for analyses (Figure 2). A random-effects model was used because of significant heterogeneity observed (I2 = 50.9%). Pooled results showed that the level of IL-6 was significantly reduced versus comparator groups (standardized mean difference [SMD] −1.05; 95% CI −1.44, −0.66; P < 0.001; I2 = 50.9%; Figure 2). Subgroup analysis showed that the level of IL-6 was significantly reduced in the levosimendan-treated group versus blank (pretreatment), placebo or dobutamine subgroups (P < 0.001, P = 0.001 and P < 0.001, respectively; Figure 2). However, only one study was included in the subgroup analysis of levosimendan versus dobutamine. Funnel plot analysis showed that there was no obvious publication bias (Figure 3).

Forest plot showing the effect of levosimendan versus controls on interleukin-6 levels in patients with advanced heart failure. Blank group, baseline (pretreatment values); SMD, standardized mean difference; CI, confidence interval.

Funnel plot analysis of interleukin-6 outcomes. SMD, standardized mean difference; s.e., standard error.
Cardiac index improvement
Three studies reported data on improvement in cardiac index in patients who received levosimendan. Due to no between-trial heterogeneity (I2 = 0.0%), a fixed-effects model was used for analysis. Pooled results showed that use of levosimendan contributed to significantly improved cardiac index (SMD 0.59; 95% CI 0.29, 0.88; P < 0.001; I2 = 0.0%; Figure 4). Subgroup analysis showed that improvement in cardiac index was significantly enhanced in levosimendan versus blank (pretreatment) subgroups (SMD 0.59; 95% CI 0.25, 0.93; P = 0.001; I2 = 0.0%; Figure 4). However, no statistically significant difference was observed between levosimendan and furosemide subgroups (P = 0.06; Figure 4).

Forest plot showing the effect of levosimendan versus controls on cardiac index in patients with advanced heart failure. Blank group, baseline (pretreatment values); SMD, standardized mean difference; CI, confidence interval.
Pulmonary artery pressure reduction
The PAP data reported by eight studies were pooled for analyses (Figure 5). Due to significant heterogeneity (I2 = 89.7%), a random-effects model was used. Overall, the use of levosimendan contributed to significantly reduced PAP (SMD −1.22; 95% CI −1.91, −0.53; P = 0.001; I2 = 89.7%; Figure 5). Subgroup analysis revealed a significantly enhanced reduction in PAP in the levosimendan versus blank (pretreatment) subgroups (SMD −1.91; 95% CI −2.94, −0.88; P < 0.001; I2 = 93%; Figure 5). However, no statistically significant differences were observed in the levosimendan versus furosemide or placebo subgroups (P = 0.651 and P = 0.214, respectively; Figure 5).

Forest plot showing the effect of levosimendan versus controls on pulmonary artery pressure in patients with advanced heart failure. Blank group, baseline (pretreatment values); SMD, standardized mean difference; CI, confidence interval.
New York Heart Association (NYHA) functional class improvement
Four studies reported NYHA functional class. Due to significant heterogeneity (I2 = 74.6%), a random-effects model was used for analyses. Pooled results showed that improvement in NYHA functional class was significantly enhanced in the levosimendan group versus controls (SMD −1.66; 95% CI −2.27, −1.04; P < 0.001; I2 = 74.6%; Figure 6). Subgroup analysis also showed that improvement in NYHA functional class was significantly enhanced with levosimendan versus furosemide, blank (pretreatment) or placebo subgroups (P = 0.017, P < 0.001 and P < 0.001, respectively; Figure 6). Only one study was included in the levosimendan versus placebo subgroup analysis.

Forest plot showing the effect of levosimendan versus controls on New York Heart Association functional class in patients with advanced heart failure. Blank group, baseline (pretreatment values); SMD, standardized mean difference; CI, confidence interval.
Discussion
The present meta-analysis of 11 published studies evaluated the anti-inflammatory and haemodynamic effects of levosimendan compared with controls in patients with AdHF. The result of this meta-analysis showed that levosimendan was associated with anti-inflammatory effects, displayed by decreased IL-6, and improved haemodynamic effects (increased cardiac index, reduced PAP, and improved NYHA functional class), and the effects were enhanced versus controls.
Interleukin-6 has long been recognised as a key cytokine in inflammation. Furthermore, chronic inflammation is associated with prolonged exposure to IL-6 signalling. As part of the inflammatory secretome, IL-6 is secreted by immune cells in response to infection. 24 Tasal et al. 13 reported that concentrations of IL-6 and tumour necrosis factor (TNF)-α were decreased after levosimendan infusion in patients with AdHF, and Parissis et al. 25 showed that levosimendan administration caused a significant reduction of circulating pro-inflammatory cytokines (IL-6 and high-sensitivity C-reactive protein [hsCRP]) in patients with AdHF. Similar results were reported by Kasikcioglu et al. 26 In addition, Krychtiuk et al. 27 found that levosimendan exerted anti-inflammatory effects on cardiac myocytes and endothelial cells in vitro. The present meta-analysis showed that levosimendan was associated with a significant reduction of IL-6 in patients with AdHF. Levosimendan infusion displays anti-inflammatory and antiapoptotic properties in patients with heart failure, reducing circulating pro-inflammatory cytokines (IL-6 and hsCRP) and soluble apoptosis mediators, 25 however, levosimendan also has the effect of improved haemodynamics. Avgeropoulou et al. 15 observed that levosimendan not only displayed beneficial anti-inflammatory effects (by reducing IL-6 and TNF-α) but also a haemodynamic effect.
A meta-analysis focusing on patients with heart failure complicating acute coronary syndrome reported haemodynamic improvements in the levosimendan group compared with control group. 28 Haemodynamic improvements after levosimendan infusion were reported in patients with AdHF in two meta-analyses.29,30 The studies by Feola et al. 19 and Ikonomidis et al. 22 showed an obvious tendency for levosimendan infusion to reduce PAP, however, the populations in these studies were small. The Levosimendan Infusion versus Dobutamine (LIDO) study revealed that levosimendan improved haemodynamic performance more effectively than dobutamine in patients with severe low-output heart failure, 31 and a study by Nieminen et al. 32 highlighted the effect of levosimendan on haemodynamics, increased cardiac index, reduced PAP and improved NYHA functional class in patients with AdHF. In the current study, stratified analysis was performed to produce an overall estimate of the included studies, and to compare the therapeutic effect with control groups at the same time. The results provided updated evidence on the effect of levosimendan in patients, showing increased cardiac index, reduced PAP, and improved NYHA functional class, supporting the results of previous studies.
The results of the present meta-analysis may be limited by several factors. Only 11 small studies were included in the analyses (including prospective, retrospective and real-world cohort studies), with a relatively small number of patients in each study. Thus, the statistical power of the present findings is weakened, due to the limited number of original studies, the small number of patients in each subgroup, and the significant variability in study designs.
Conclusion
The current evidence suggests that treatment with levosimendan appears beneficial in patients with AdHF, via anti-inflammatory and improved haemodynamic effects (increased cardiac index, reduced PAP, and improved NYHA functional class). Thus, levosimendan should be recommended for clinical application in patients with AdHF. Large, multicentre randomized controlled trials are warranted to validate these meta-analysis findings.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221148402 - Supplemental material for The anti-inflammatory and haemodynamic effects of levosimendan on advanced heart failure patients: a meta-analysis of published studies
Supplemental material, sj-pdf-1-imr-10.1177_03000605221148402 for The anti-inflammatory and haemodynamic effects of levosimendan on advanced heart failure patients: a meta-analysis of published studies by Suzhen Zhou, Denghua Li, Lieliang Zhang and Juxiang Li in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221148402 - Supplemental material for The anti-inflammatory and haemodynamic effects of levosimendan on advanced heart failure patients: a meta-analysis of published studies
Supplemental material, sj-pdf-2-imr-10.1177_03000605221148402 for The anti-inflammatory and haemodynamic effects of levosimendan on advanced heart failure patients: a meta-analysis of published studies by Suzhen Zhou, Denghua Li, Lieliang Zhang and Juxiang Li in Journal of International Medical Research
Footnotes
Acknowledgements
The authors would like to express their gratitude to all who helped with the article.
Author contributions
Suzhen Zhou and Juxiang Li conceived and designed the research. Lieliang Zhang contributed analytical tools and analysed the data. Suzhen Zhou wrote the manuscript. Denghua Li revised the manuscript. Suzhen Zhou and Denghua Li are co first authors. All authors read and approved the manuscript.
Data availability statement
Data included in this research are openly available in a public repository.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
