Abstract
A 50-year-old man with a 20-year history of left-sided ulcerative colitis (UC) presented to our hospital with sudden onset of watery diarrhea. To this point, he had been treated with mesalazine 2.0 g/day for UC and had maintained remission. We considered that the UC had worsened. We immediately performed surveillance colonoscopy, which revealed a normal mucous membrane. The results of blood laboratory examinations were normal. Histopathology of colonic biopsies revealed new-onset collagenous colitis (CC), with a thickened subepithelial collagen band (SECB) and inactive UC. We herein report the importance of random colonic biopsies to diagnose CC even when the endoscopic appearance of the colon is normal in patients with inflammatory bowel disease with worsened diarrhea.
Keywords
Introduction
Collagenous colitis (CC) very rarely develops in patients with pre-existing inflammatory bowel disease (IBD). CC is a rare disease characterized by chronic watery diarrhea and thickening of the collagenous fibrous zone just below the large intestine, that is, the subepithelial collagen band (SECB).1,2 The severity of diarrhea is independent of SECB thickness and the number of intraepithelial lymphocytes but is related to the degree of infiltration of inflammatory cells in the lamina propria. Patients without symptoms may be diagnosed by endoscopic biopsy; endoscopic findings are usually normal. 3 An erythematous colonic mucosa, edema, blood vessel changes, and minimal ulcers called mucosal tears, are sometimes observed. 4
The etiology of CC is unknown; however, genetic and environmental factors have been suggested as causes.5,6 A close relationship between CC and dysfunctional mucosal immune responses has been considered but not confirmed. Investigations of whether there is a significant association between CC and IBD have not yielded clear results.7–9 Cases of simultaneous-onset CC and IBD have been reported.10,11
Here, we report a patient with ulcerative colitis (UC) who was in remission and who developed CC. We also present a review of the literature.
Case report
The patient was a 50-year-old man with a 20-year history of left-sided UC that had been treated with 2.0 g/day of mesalazine. He was in clinical remission during the 20-year period. There was no family history of autoimmune diseases, and the patient had no history of medical conditions other than UC. In May 2019, he suddenly developed increased bowel movements with loose stools but no bloody stools. He then presented to our hospital.
At presentation, his height was 178 cm, and his weight was 72 kg. His body temperature was 35.7°C, blood pressure was 103/63 mmHg, and heart rate was 64 beats/min. He had mild abdominal pain and discomfort. He denied food contamination, medication changes, or the recent use of antibiotics. Additionally, he had no extra-intestinal manifestations, such as oral lesions. Laboratory tests (Table 1) showed normal blood cell counts and no evidence of coagulopathy. No significant bacteria were detected in the stool culture, and Clostridium difficile toxin testing was negative. Urinalysis findings were normal, and abdominal ultrasonography showed no acute abnormalities.
Laboratory data for the patient.
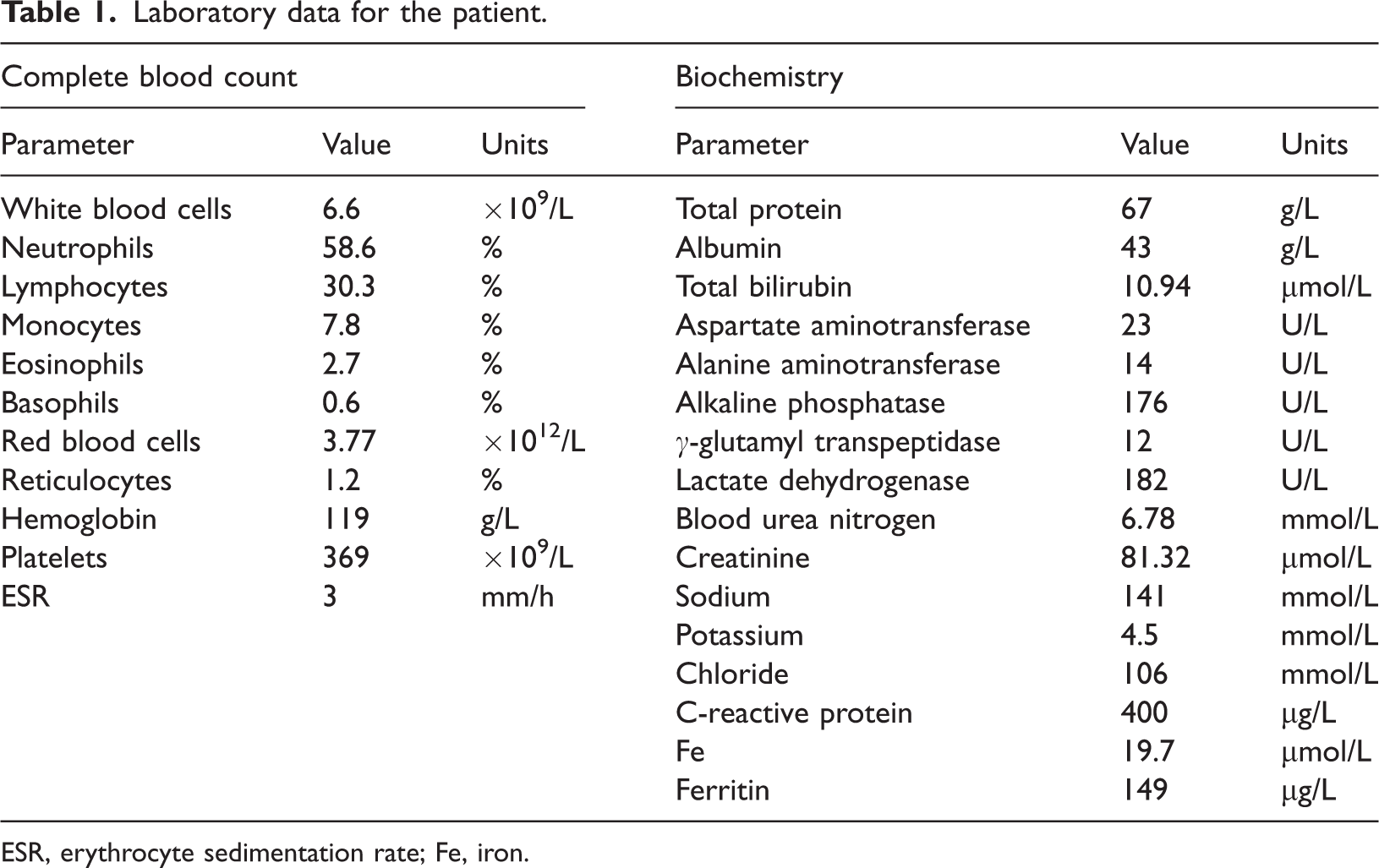
ESR, erythrocyte sedimentation rate; Fe, iron.
First, we suspected exacerbation of UC and increased the dose of mesalazine from 2 g per day to 4 g per day. Within 1 month, the patient underwent colonoscopy; however, there was no evidence of the progression of UC (Figure 1a). Histological examination of colonic biopsies showed a sloughed surface epithelium, increased lymphocyte and plasma cell counts, and a marked deposition of collagen fiber bundles under the superficial layer, which was revealed by Masson’s trichrome staining (Figure 1b and c). There were also morphological alterations in Paneth cells, which are specific for IBD. Crypt abscess or cryptitis was not detected. We performed colonoscopy annually for UC follow-up, with surveillance biopsies showing normal results. The most recent colonoscopy also showed a normal mucosa, and histology did not reveal collagen fibers (Figure 2a and b).

(a) Colonoscopic image obtained July 2019 showing a normal mucosa. (b) Biopsy specimen stained with hematoxylin and eosin (×200) showing that the surface epithelium has sloughed, and the numbers of lymphocytes and plasma cells are increased compared with normal epithelium. Morphological changes in Paneth cells, which are specific for inflammatory bowel disease (IBD), are visible. Crypt abscess or cryptitis is not present and (c) Masson’s trichrome staining of the same specimen as that seen in (b) (×200). Marked deposition of collagen fibers with an increased thickness of >10 μm are visible under the superficial layer.

(a) Colonoscopic image obtained 1 year before the patient presented with CC showing a normal mucosa and (b) Histological findings for a biopsy specimen obtained 1 year before the patient presented with CC, stained with hematoxylin and eosin (×200) and showing a normal mucosa and a small number of inflammatory cells.
Oral administration of ramosetron hydrochloride 5 μg per day was started after obtaining pathology findings suggestive of CC. Because no improvement was observed, the treatment was discontinued after approximately 4 weeks. This was followed by oral administration of colestimide 3 g per day. Mesalazine (4 g/day) for UC had been administered continuously. In October 2019, the number of loose bowel movements had decreased, and the fecal properties had improved. In August 2020, follow-up colonoscopy showed that the collagenous fiber bundles suspected to be due to CC had disappeared (Figure 3a and b), and oral administration of colestimide was stopped. The dosage of 4 g per day of mesalazine was then decreased to the original 2 g per day. Clinical remission of UC was maintained thereafter.

(a) Colonoscopic image obtained 1 year after the diagnosis of CC showing a normal mucosa and (b) Colonic biopsy specimen obtained 1 year after the diagnosis of CC stained with hematoxylin and eosin (×200). The collagen band has disappeared, and the surface epithelium is normal. Additionally, the numbers of inflammatory cells has decreased compared with previous biopsies.
Discussion
In the current case, although the UC was in remission, diarrhea had worsened owing to CC. Although cases of CC in IBD patients have been reported, such reports are extremely rare. CC is a chronic IBD characterized by watery, non-bloody diarrhea.1,7 The diagnosis of CC is based on histological findings, including collagen fiber bundles with a thickness of ≥10 μm, which are found just below the basement membrane of the colonic epithelium and are designated as SECB. 2 Additional features of CC are the infiltration of lymphocytes and plasma cells into the lamina propria, normal arrangement of crypts, detachment and flattening of the superficial epithelium, and increased intraepithelial lymphocytes. 12 CC shows no obvious abnormal findings on endoscopy. 3 However, in recent studies, some endoscopic features have been reported, such as elevated capillaries, crowded tortuous vasculature, granular surface changes in the mucosa, and elongated longitudinal ulcers described as “linear mucosal defects”. These mucosal ulcers were reported to be significantly more frequent in the left colon among Japanese patients compared with Western patients.4,13 Regarding the mechanism of development of CC, it is thought that the mucous membrane hardened by collagen fiber deposition is torn by increased internal pressure and mechanical stimulus associated with intestinal peristalsis and hyperextension.14,15
In Japan, most CC is related to the use of drugs, such as proton-pump inhibitors, non-steroidal anti-inflammatory drugs, such as aspirin, and ticlopidine. Discontinuation of such drugs is the first choice of treatment. However, the patient in this report had no history of the use of these drugs. UC, which is an autoimmune disease, was suspected. Why and how a patient with IBD develops CC is unknown. That IBD and CC have some relationship, especially a genetic predisposition, has been suggested; however, the exact cause remains unknown.8–10 It is often thought that active chronic inflammation induces the development of collagen bands and leads to CC; however, as in our case, most IBD patients who developed CC in previous reports had maintained remission. Saad et al. 10 reported that the temporary development of CC in IBD patients suggests an abnormal healing response resulting from an imbalance between the inflammatory and mucosal healing pathways. The collapse of this balance during the long-term healing phase would result in CC. 7 It is stated that immunodeficiency and autoimmunity are involved in the formation of the collagen bands observed in CC. 7 Additionally, anion exchange resins, such as cholestyramine, that bind to bile acids, are effective in treating CC, which indicates that intestinal factors, such as bile acids and endotoxins, are involved in the development of CC.6,16
With immune disorders such as IBD, a variety of microorganisms, such as bacteria, fungi, and viruses, help maintain homeostasis in the intestine.17,18 Patients with UC have dysbiosis because of the reduced diversity and abundance of the intestinal flora. 19 This chronic inflammation is recognized by the immune system as abnormal, and the innate immune response to the gut microbiota continues, causing excessive oxidative stress in the cells. Considering these changes, an association between UC and intestinal infectious diseases cannot be completely denied. However, in our patient, stool cultures and Clostridium difficile toxin were negative by polymerase chain reaction (PCR) testing.
Regarding the treatment of CC, if a causative drug has been identified, its discontinuation might improve symptoms. When the cause is unknown, as in the present case, dietary guidance, such as avoiding overindulgence in caffeine, alcohol, and dairy products is important. It has been reported that loperamide and cholestyramine are effective symptomatic treatments.3,6 Our case improved with the administration of colestimide, and we believe this result was affected by intestinal factors, such as bile acids and gut bacteria. There are also many reports of the usefulness of steroids in similar cases. 20
This report raises important issues in that IBD patients could develop CC despite successful UC treatment clinically and with remission shown endoscopically. Such patients are likely to be initially misdiagnosed as having diarrhea-predominant irritable bowel syndrome (IBS-D) before a biopsy is obtained and analyzed. It is well established that symptoms such as IBS can occur in up to 30% to 50% of patients with inactive IBD, and there is an overlap in symptoms between CC and functional bowel disorders.21,22 In the present case, endoscopic biopsy was reliable. However, when evaluating the results of colonoscopic biopsies, the possibility of sampling errors should be considered. The collagen layer may be thickened in specific areas and alternate with normal-sized mucosal structures. To avoid the possibility of errors due to sample selection
Conclusion
In IBD patients with increased diarrhea and exacerbation of symptoms, such as abdominal pain, despite the maintenance of endoscopically-confirmed remission, it is important to obtain random biopsies of the colon to diagnosis CC. A CC diagnosis in patients with IBD could be overlooked and misinterpreted as IBS. Because CC and IBS require different treatments, an accurate diagnosis is required. Interestingly, a few months after the diagnosis of CC, this case was successfully treated with oral colestimide, and the SECB resolved. In IBD patients with increased diarrhea and exacerbation of symptoms, such as abdominal pain, despite the maintenance of endoscopically-confirmed remission, it is important to obtain random biopsies of the colon to diagnose CC. A CC diagnosis in patients with IBD could be overlooked and misinterpreted as IBS. Because CC and IBS require different treatments, an accurate diagnosis is required. We believe that random colonic biopsies should be performed for the diagnosis of CC if patients with IBD continue to have exacerbation of diarrhea after complete mucosal healing. More research is needed to determine the mechanism underlying the development of CC in IBD patients with complete mucosal healing.
Footnotes
Author Contributions
Conceptualization, M. Haraikawa and T.S.; methodology, T.S.; clinical examination, T.S., T.K., O.N., T.T., H.F., T.M., D.I., M. Hojo, and T.Y.; investigation and data curation, M. Haraikawa, T.S., K.I., K.N., K.H., and T.K.; writing the manuscript draft and editing, M. Haraikawa; supervision, A.N.; project administration, A.N. All authors have read and agreed to the published version of the manuscript.
Data availability statement
Not applicable.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
The study was performed in accordance with the Declaration of Helsinki. The local Ethics Committee of the Juntendo University School of Medicine approved this study protocol on 10 June 2022 (Institutional Review Board no. JHS22-0033). Informed consent was obtained from the patient involved in the study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
