Abstract
In this study, we aimed to assess the efficacy of high-flow nasal oxygen (HFNO) to maintain blood peripheral oxygen saturation (SpO2) in patients undergoing suspension laryngoscopy under general anesthesia. Adult patients were included in this bicenter study. After face-mask oxygenation, HFNO at a flow rate of 70 L min−1 and fraction of inspired oxygen 100% was initiated at loss of consciousness. At the end of HFNO, blood gas analysis was performed. Of the 29 included patients, five (17.2%; 95% confidence interval [CI]: 7.6–34.5) presented SpO2 <95% during the first 15 minutes of the procedure and eight patients (27.6%; 95% CI: 14.7–45.7) presented SpO2 <95% throughout the procedure. Six patients (20.7%; 95% CI: 9.8–38.4) required rescue jet ventilation. Median apnea time before SpO2 <95% was 13.5 (interquartile range [IQR]: 10–17.7) minutes. Arterial carbon dioxide tension at the end of the procedure or at the time of study discontinuation was 9.73 (IQR: 8.8–10.9) kPa and was higher than 8 kPa in 88.9% (95% CI: 71.9–96.1) of patients. HFNO was associated with a relatively high incidence of suboptimal oxygen saturation and hypercapnia during suspension laryngoscopy under general anesthesia and may not be considered the reference technique.
Introduction
Anesthetic management of suspension laryngoscopy must meet several requirements: ensuring sufficiently deep anesthesia to avoid memory of the procedure, guaranteeing satisfactory gas exchange, and causing the least possible disturbance to the operative vision. 1 No technique can fully meet all three elements. A recent French national survey on pan-endoscopy revealed that in 2018, the top two techniques were orotracheal intubation (62%) and general anesthesia with spontaneous ventilation (37%). Other techniques therefore have anecdotal use. 2 However, it is likely that the use of high-flow nasal oxygen (HFNO) will increase for this indication, particularly because HFNO is indicated for the treatment for hypoxic patients with COVID-19. 3 HFNO consists of transnasal high-flow insufflation of heated, humidified, oxygen (O2)-enriched air. The procedure has several advantages for conscious patients compared with the other available noninvasive ventilation techniques, particularly enhanced comfort and tolerance and reduced work of breathing.4–6 Patel and Nouraei reported the use of OptiFlow™ (Fisher and Paykel Healthcare Limited, Auckland, New Zealand), a commercial device delivering HFNO in anesthetized and paralyzed patients. 7 Their technique, called transnasal humidified rapid-insufflation ventilatory exchange (THRIVE), ensured a prolonged median apnea time of 14 minutes with an interquartile range of 9 and 19 minutes and extreme values of 5 and 65 minutes, with a rate of increase in end-tidal carbon dioxide of 0.15 kPa min−1.
The aim of this open-label prospective two-center study was to assess the efficacy of HFNO to maintain blood oxygen saturation in apneic patients undergoing suspension laryngoscopy under general anesthesia.
Methods
This prospective observational, multi-center cohort study was conducted in two French hospitals (Institut Curie, Saint-Cloud and Foch Hospital, Suresnes). The study was initiated after receiving Ethics Committee approval (East Regional Research Ethics Committee, Chairperson JP Quenot, ID RCB 2018-A00396-49 on 12 March 2018) and registration at ClinicalTrials.gov (NCT03472118 on 21 March 2018). Written informed consent was obtained from patients prior to their participation in the study. This report follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational cohort studies. 8
It was decided that the sponsor of the study (i.e., the data monitoring committee) would be kept informed of the occurrence of adverse events and would make the decision whether to continue the study.
Study population
We included adult patients with American Society of Anesthesiologists physical status class I–III scheduled for short diagnostic and/or therapeutic suspension laryngoscopy (except laser surgery and cavoscopy). Exclusion criteria were pregnancy, epilepsy, arrhythmia, pulmonary hypertension, obstructive laryngeal tumors, lung disease with basal SpO2 <95%, and obesity (body mass index >30 kg/m2).
Study design
Patients were provided with information about the study and gave their written informed consent at a pre-anesthetic visit. Patients did not receive any premedication. An intravenous line was placed on arrival in the operating room. Standard monitoring included electrocardiogram, noninvasive arterial blood pressure monitoring, pulse oximetry (SpO2), capnography, bispectral index, and train-of-four monitoring. Patients were pre-oxygenated with 100% O2 using a tight-fitting face mask with end-tidal gas monitoring for 3 minutes or longer, if necessary, until an end-tidal O2 concentration ≥90% was achieved. Induction of anesthesia was performed with target controlled infusion (Alaris™ PK syringe pump, Cardinal Health, Rolle, Switzerland) of remifentanil (Ultiva®, GlaxoSmithKline AB, Solna, Sweden) and propofol (Propofol Lipuro®, B. Braun Melsungen AG, Melsungen, Germany) followed by neuromuscular blockade with 0.6 mg kg−1 rocuronium (Esmeron®, MSD, Haarlem, Netherlands). Anesthesia was maintained with target concentration infusions of propofol and remifentanil with bispectral index objectives between 40 and 60, and mean arterial pressure within 20% of baseline, with reinjection of rocuronium to maintain a null response to train-of-four monitoring. HFNO was started after the onset of apnea using a high-flow nasal cannula and the simplified Optiflow® device (Fisher and Paykel Healthcare Ltd., Auckland, New Zealand) with a flow rate of 70 L min−1 and fraction of inspired oxygen (FiO2) 100%. A transglottic jet ventilation catheter was inserted into the trachea through the mouth. HFNO was stopped and replaced by high-frequency jet ventilation when SpO2 fell to below 92% or when the procedure lasted more than 30 minutes or when the patient presented arrhythmia. Jaw thrust was applied from onset of apnea until the start of surgery and then from the end of surgery until awakening. Neuromuscular blockade was reversed using prostigmine or sugammadex (Bridion®, MSD, Hertfordshire, United Kingdom), depending on the train-of-four monitoring results. HFNO was stopped as soon as the patient regained spontaneous breathing. At the end of surgery, or earlier when SpO2 was less than 92%, an arterial blood sample was drawn for blood gas analysis. Surgeons were asked to evaluate their satisfaction with the ventilation technique using an 11-point numeric rating scale, with 0 indicating “completely dissatisfied” and 10 indicating “completely satisfied.” Patients were transferred to the post-anesthesia care unit (PACU) for routine care. All patients were routinely placed on supplemental O2 (3 L min−1) for the first 30 minutes of their PACU stay. On the first postoperative day, ambulatory patients were contacted by phone and any post-discharge complications were noted. Hospitalized patients were also screened for complications on the first postoperative day.
Outcomes
The primary outcome of the study was the incidence of peripheral oxygen saturation (SpO2) below 95% during the first 15 minutes of apnea. Secondary endpoints included the incidence of SpO2 below 95% throughout the entire procedure (maximum 30 minutes), the incidence of SpO2 below 92% throughout the procedure, the time between onset of apnea and onset of SpO2 <95%, the incidence of hypercapnia (defined as arterial carbon dioxide tension >8 kPa or 60 mmHg) at the end of the procedure (or at the end of the experiment, whichever came first), blood pH at the end of the procedure (or at the end of the experiment, whichever came first), time-course of arterial blood pressure, heart rate, intra-operative and post-operative complications, and the surgeon's satisfaction.
Statistical analysis
Because this study was purely descriptive, no power analysis was performed; we planned to include 100 patients. An interim analysis was planned after inclusion of the first 30 patients. Results are expressed as percentage (95% confidence interval, CI) and median (interquartile range, IQR). Data were analyzed using IBM SPSS Statistics, version 22 (IBM Corp., Armonk, NY, USA).
Results
The study was prematurely terminated for safety concerns owing to the development of severe hypercapnia in a number of patients (inclusion of the first patient on July 5th, 2018 and inclusion of the last one on January 14th, 2019). At the time of study discontinuation, 30 patients had been included and 29 had completed the study protocol (17 at Foch Hospital and 12 at Institut Curie) (see Figure 1). Baseline patient demographic characteristics are presented in Table 1. The median age of participants was 56 years and the male-to-female ratio was 62% to 38%.

Flow diagram.
Patient characteristics.

BMI, body mass index; IQR, interquartile range.
Median total operating time was 12 [10.00–22.00] minutes (Table 2). Five patients (17.2%; 95% CI: 7.6–34.5) presented blood oxygen saturation below 95% during the first 15 minutes of the procedure and eight patients (27.6%; 95% CI: 14.7–45.7) presented blood oxygen saturation below 95% throughout surgery. Six patients (20.7%; 95% CI: 9.8–38.4) required rescue jet ventilation for blood oxygen saturation below 92%. Median apnea time before SpO2 <95% was 13.5 (IQR: 10. 0–17.7) minutes with a minimum of 2 minutes and a maximum of 49 minutes.
Outcome results.
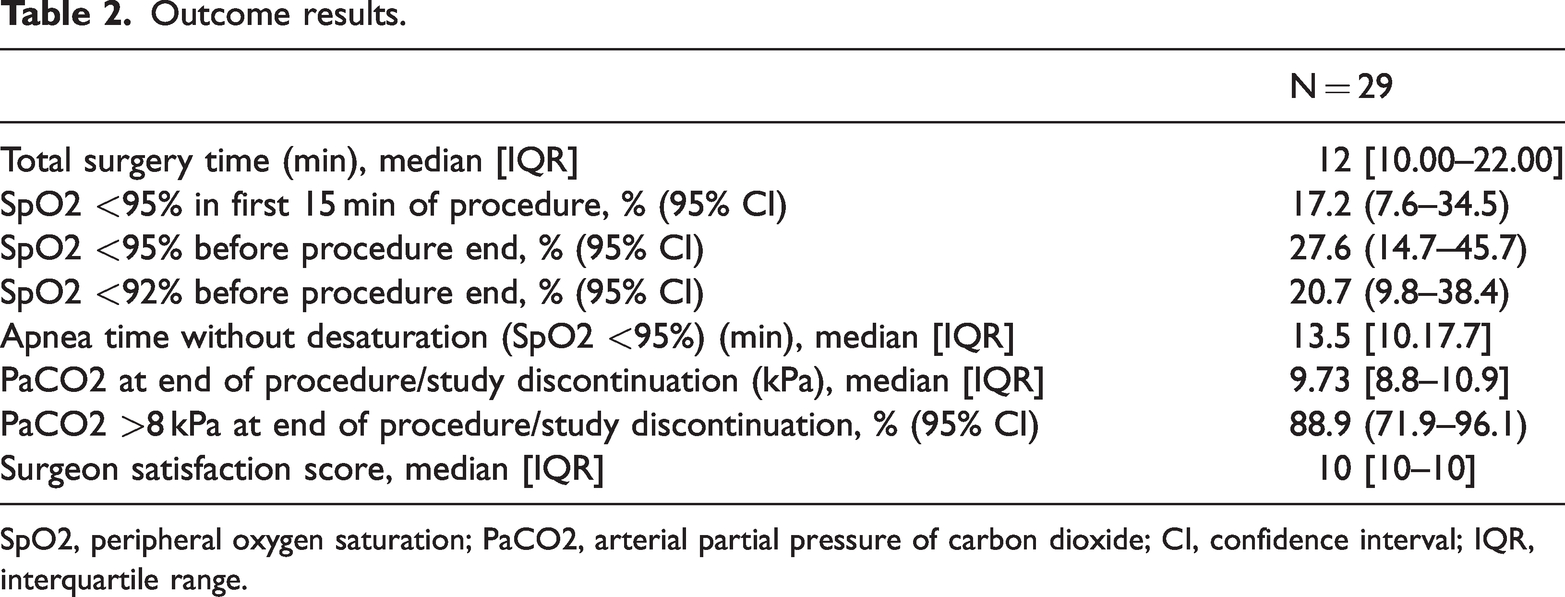
SpO2, peripheral oxygen saturation; PaCO2, arterial partial pressure of carbon dioxide; CI, confidence interval; IQR, interquartile range.
The evolution of SpO2 in all patients is shown in Figure 2, with all normoxic patients (SpO2 ≥92%) in the left panel and hypoxic patients in the right panel. Median arterial partial pressure of carbon dioxide (PaCO2) at the end of the procedure, or at the time of study discontinuation, was 9.73 (IQR: 8.8–10.9) kPa and was higher than 8 kPa (60 mmHg) in 88.9% (95% CI: 71.9–96.1) of patients. One patient had a PaCO2 of 13.45 kPa. The lowest reported pH was 7.11.

Individual evolutions of pulse oximetry. Left panel: evolution of pulse oximetry for each patient whose SpO2 value was ≥92%. Right panel: evolution of pulse oximetry for each of the 6 patients with SpO2 <92%.
No patients experienced cardiac arrhythmia. The mean time from sugammadex injection and discontinuation of propofol infusion to the patients opening their eyes was 4 minutes (95% CI: 3.0–5.5). No seizures were reported. All patients remained hemodynamically stable. One patient experienced intra-operative epistaxis. No other intra-operative complications or postoperative complications were reported.
The median surgeon's satisfaction score with laryngeal visibility was 10 (IQR: 10–10).
Discussion
In this case series, we observed that patients undergoing suspension laryngoscopy and receiving high-flow apneic nasal oxygenation could occasionally develop early oxygen desaturation and severe hypercapnia.
All anesthetic techniques used for suspension laryngoscopy and for other surgical laryngeal procedures have advantages and disadvantages. 1 Briefly, total intra-venous anesthesia and maintenance of spontaneous ventilation are widely used but the balance between sufficiently deep anesthesia and satisfactory gas exchange may be difficult or impossible to achieve. 9 In contrast, tracheal intubation avoids most ventilatory problems at the cost of discomfort for the operator, even if a small-diameter intubation probe is used. High frequency jet ventilation can be used with i) a laryngoscope with a specific channel for jet ventilation (i.e., supraglottic jet ventilation), 10 ii) a tracheal catheter placed through the mouth (i.e., subglottic jet ventilation), 11 or iii) a tracheal catheter placed percutaneously through the cricothyroid membrane. 12 However, high frequency jet ventilation remains a marginal technique owing to the requirement for a specific ventilator and the risk of a specific complication, notably pneumothorax. 13 Therefore, HFNO appears to be the ideal anesthetic technique for laryngoscopy suspension because it is non-invasive and does not interfere with operative vision to ensure satisfactory gas exchange.
Oxygen flow delivered by conventional nasal cannula is generally limited to 10 L min−1 whereas HFNO uses oxygen flows between 50 and 120 L min−1 administered via specially designed nasal cannulas. These high O2 flows mean that little ambient air is drawn into the cannula, allowing for a high FiO2 (95% to 100%).
HFNO now has a prominent role in intensive care as an alternative to noninvasive ventilation owing to several advantages, particularly clearance of the anatomical dead space, 14 improved CO2 elimination, 15 creation of positive end-expiratory pressure roughly proportional to flow (1 cm H2O for every 10 L min−1 of flow) when the patient keeps their mouth closed,16,17 reduced work of breathing, and attenuation of inspiratory resistance. 18 These characteristics of HFNO result in improvement of patient comfort as a result of humidification and heating of the delivered mixture. 19
HFNO is also used in anesthesia, especially since the publication by Patel and Nouraei reporting prolonged median desaturation-free apnea time in paralyzed patients under general anesthesia, leading to the introduction of THRIVE. 7 HFNO has also been tested for preoxygenation prior to induction of general anesthesia; 20 continuous oxygenation during intubation, especially when rapid sequence intubation is indicated; 21 oxygenation during fiberoptic intubation; 22 oxygenation during intravenous anesthesia, especially in the presence of airway obstruction; 17 and postoperative O2 therapy, 23 particularly after cardiac 24 and cardiothoracic 25 procedures.
THRIVE could be the best technique for diagnostic and surgical suspension laryngoscopy because it permits complete visualization of the glottis. Evaluation of the safety of THRIVE consists of evaluation of the risk of oxygen desaturation and hypercapnia during prolonged apnea under general anesthesia with muscle paralysis. In 2015, Patel and Nouraei 7 reported a prolonged apneic window for up to 65 minutes with HFNO, with a median apnea time of 14 minutes (IQR: 9–19) with no desaturation below 90% and an apnea time ranging from 5 minutes to 65 minutes. Other authors have reported similar apnea times: 19 (IQR: 15–24) minutes in a series of patients undergoing laryngeal or tracheal surgery performed under apneic conditions 26 and 18 (IQR: 10–27) minutes in a group of patients undergoing surgery for subglottic stenosis. 27 Of particular interest is a study by Gustafsson et al. on patients undergoing brief laryngeal surgery who were oxygenated using THRIVE with 100% O2 at 40–70 L min−1. Blood gas analyses were performed every 5 minutes during the procedure. The authors defined the apnea time as the interval between the onset of apnea and the end of the procedure, or earlier in the following situations: apnea time exceeding 40 minutes, PaCO2 >11 kPa, pH <7.15, SpO2 <90%, or the presence of malignant arrhythmia. Gustafsson et al. reported a mean apnea time of 22.5 ± 4.5 minutes, 28 which would appear to be suitable for suspension laryngoscopy performed as a diagnostic procedure or to treat phayngolaryngeal lesions as the procedure duration is only slightly longer than reported apnea times. Contrary to these data showing the safety of HFNO for these indications, we report disquieting results in our series: oxygen desaturation occurred before completion of surgery in 20.6% of cases, and we decided to introduce rescue high-frequency jet ventilation when SpO2 fell below 92% to avoid subsequent rapid desaturation because such SpO2 values are close to the steep slope of the oxyhemoglobin dissociation curve. Moreover, we think that the study of HFNO safety should not stop at the analysis of mean or median apnea times; examination of individual data shows that a relatively high percentage of patients experience oxygen desaturation, whether defined as SpO2 <95%, <92%, or <90%.7,26,28
Our protocol differs from that reported in previous publications in terms of choice of preoxygenation technique. Previous publications have reported preoxygenation using HFNO with different flows and durations: 70 L min−1 for 10 minutes, 7 40 L min−1 for 3 minutes, 28 and 80 L min−1 for 3 minutes. 26 In our study, we used face mask preoxygenation to achieve an end-tidal O2 ≥90%, which constitutes the gold standard for preoxygenation and is the only way to document lung denitrogenation, a necessary condition for ventilatory flow motion, the physiological basis of apneic oxygenation. 29
We observed one case of early desaturation (2 minutes after the onset of apnea) despite preoxygenation performed according to the protocol and application of jaw thrust as soon as apnea was obtained. A possible explanation for this case of early desaturation could be unidentified obstruction of the nasal cavity. Another hypothesis is this was owing to the presence of unidentified tracheal or bronchial collapse. To date, no published studies have identified a patient population at risk of early oxygen desaturation during apneic HFNO. In the absence of easily identifiable risk factors for HFNO failure, practitioners should always have an alternate airway management plan if they decide to use HFNO during apnea.
Several mechanisms have been proposed to explain CO2 elimination during HFNO: rapid clearance of nasal cavities, 14 positive airway pressure generation,30,31 reduced O2 dilution by nitrogen 32 and, more recently, interaction between entrained and highly turbulent supraglottic flow vortices created by HFNO and cardiogenic oscillations.33,34 Laviola et al. proposed a combination of cardiac oscillations with a high degree of gas mixing in the anatomical dead space and high-frequency glottic pressure oscillations as a mechanism of CO2 clearance with HFNO. 34 Increased blood carbon dioxide levels during THRIVE has been reported, with lower values in a laboratory model (0.15 kPa min−1) 33 and in adults under general anesthesia (from 0.17 27 to 0.24 28 kPa min−1) than in our study (0.3 kPa min−1 assuming a preoperative median value of 5 kPa). Caution must be urged when capnography is used; it has been shown that the increase in PaCO2 is twice that measured using end-tidal CO2. 28 However, no difference has been reported between transcutaneous CO2 monitoring and PaCO2. 28
As expected, surgeons reported a high level of satisfaction with THRIVE owing to the absence of interference between the ventilation technique and the surgical procedure. However, the main drawback was that THRIVE could not be used for procedures requiring the use of a laser owing to the risk of combustion. The use of laser is also contraindicated when using jet ventilation where a FiO2 of 50% is used in laser surgery, although the FiO2 actually delivered is lower owing to air entrainment. 35 However, this dilution cannot be evaluated in practice, which has led many practitioners to prefer the use of positive pressure ventilation via endotracheal tubes specifically designed for laser surgery.
This study has several limitations. This was a case series without a control group, which considerably limits the conclusions to be drawn. Furthermore, our study had to be prematurely terminated owing to patient safety concerns. Regular review of study data by the data monitoring committee revealed a high incidence of sometimes severe hypercapnia; the data monitoring committee therefore recommended termination of the study. We used an SpO2 monitor that cannot differentiate oxyhemoglobin and carboxyhemoglobin; this device may therefore have underestimated hypoxemia when patients had smoked several hours before anesthesia. 36 We used face mask preoxygenation in this study whereas all previous studies used HFNO preoxygenation before apneic oxygenation; this makes it difficult to compare our results with previously reported data and suggests that differences in the preoxygenation technique may be at least partly responsible for differences in the reported results. Further studies are needed to determine the optimal preoxygenation technique before apneic HFNO. Our study population also did not include any obese patients or patients with obstructive airway tumors, precluding generalization of our results to those clinical settings.
Conclusions
In contrast to several studies showing the safety of HFNO, our case series showed less favorable results. We observed a relatively high incidence of suboptimal oxygen saturation and occasional early desaturation during suspension laryngoscopy under general anesthesia. It is possible that the safety of HFNO can be improved if this technique is also used to provide preoxygenation. Our results indicate caution is needed if HFNO is chosen, and a reliable rescue technique must be readily available.
Research Data
Research Data for High-flow nasal oxygen for suspension laryngoscopy: a multicenter open-label study
Research Data for High-flow nasal oxygen for suspension laryngoscopy: a multicenter open-label study by Mary Saad, Aline Albi-Feldzer, Rabah Taouachi, Isabelle Wagner, Marc Fischler, Pierre Squara and Morgan Le Guen in Journal of International Medical Research
Footnotes
Acknowledgements
The authors would like to thank Cecile Naudin for her help in the management of the study. The authors would also like to thank Polly Gobin for her linguistic assistance.
Author contributions
Conceptualization: M.S., M.F., M.L-G.
Methodology: P.S.
Validation: M.S., M.F., P.S.
Formal analysis: P.S.
Investigation: M.S., A.A-F., R.T., I.W., M.L-G.
Data curation: M.S., A.A-F., R.T., I.W., M.F., M.L-G.
Visualization: M.F.
Supervision: M.F.
Project administration, Funding acquisition, Resources: P.S.
Writing—original draft preparation: M.S.
Writing—review and editing: M.F.
All authors have read and agreed to the published version of the manuscript.
Data availability statement
The dataset generated during the current study is available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt (pending publication) of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Clinique Ambroise Paré, Neuilly sur Seine, France. Paykel and Fisher provided material but did not have any involvement with the study design and conduct nor analysis of the data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
