Abstract
Objective
Mammography is the gold standard screening procedure for the early diagnosis of breast cancer. This study aimed to determine the distribution of breast density among women older than 40 years in Sulaimaniyah, Iraq, and to examine the correlations between breast density and various risk factors.
Methods
This cross-sectional study included 750 women who received routine mammographic breast screening at Sulaimaniyah Breast Center. Bilateral standard two-view mammographic images (craniocaudal and mediolateral oblique projections) were acquired and reported using a picture archiving and communication system. American College of Radiology (ACR) Breast Imaging-Reporting and Data System (BI-RADS) assessment categories C and D were considered as dense.
Results
A total of 54.3% of breasts were classified as dense, with ACR-BI-RADS categories C or D. Breast density was significantly associated with age, body mass index, a family history of breast cancer, and pre-menopause, and women with no history of breastfeeding were more likely to have dense breasts than those with partial or complete breastfeeding.
Conclusions
This study revealed that women from Sulaimaniyah with a distinct breast-density profile at mammographic screening may have a significantly increased risk of breast cancer.
Introduction
Breast cancer is the second most common neoplasm worldwide and the most frequent tumor in women. 1 Although death due to breast neoplasms has reduced in developed countries, it remains the leading cause of mortality in women in less-developed countries and the second-leading cause in developed countries. 2 The primary factor affecting breast cancer mortality is its detection in the progressive phase. Breast cancer screening regimens are thus important for aiding early cancer discovery, especially when the lesion is small and not clinically palpable. 3
Mammography is the gold-standard screening technology for the initial diagnosis of breast neoplasms. It is non-invasive, reasonably cheap, and has acceptable sensitivity (72%–88%) that increases with age. Its main benefit is its ability to detect non-palpable neoplasms <1.0 cm at an early stage, thus potentially increasing the survival rate. 4
Mammographic breast density (MBD) reflects the amount of dense (fibroglandular) compared with lucent (fatty) tissues. Increased density obscures tiny nodules, which represent a risk factor for breast cancer growth. 5 The assessment of breast density is thus a potentially useful element of mammographic screening. In addition, the sensitivity of mammography is influenced by the quantity of glandular tissue, which declines in line with increasing density because of the accumulation of radiopaque dense breast tissue overlying the primary tumor when a three-dimensional breast is imaged in a two-dimensional plane. 6
Mammographically opaque fibroglandular breast tissue is composed of epithelial glandular tissues comprising terminal ductal lobes and ducts with stromal tissues, including the supportive fibrous connective tissue inside the inter- and intralobular stroma. The American College of Radiology (ACR) Breast Imaging-Reporting and Data System (BI-RADS) lexicon mode describes mammographically opaque and dense tissue patterns as “the breasts are heterogeneously dense, which may obscure small masses” and “the breasts are extremely dense, which reduces the sensitivity of mammography”. 7 Taking account of the masking effect of breast density increases the detection rate of relapsed cancers within 1 year of a routine mammogram. 8 Women with the highest MBD thus have a four- to six-fold higher risk of breast malignancy than those with less-dense breasts. 9
Breast density is influenced by age, body mass index (BMI), parity, and menopause, but these factors only account for 20% to 30% of the differences, and studies have also demonstrated that MBD is inherited, with genetic factors accounting for 63% of the variance. 8
Breast density differs in relation to ethnicity 10 and geographical location. 11 Planning population-based screening programs thus necessitates a knowledge of the MBD characteristics of the target population. The present study aimed to document the MBD distribution among women aged over 40 years in Sulaimaniyah city, and analyze the correlations between breast density and its potential determinants or risk factors.
Methods
Study design and patients
This cross-sectional study included mammograms from women undergoing routine mammographic breast screening at the Sulaimaniyah Breast Center from November 2021 to March 2022. The inclusion criteria were consecutive women aged >40 years seeking breast cancer screening, with no signs or symptoms of breast disease or history of personal breast cancer.
Ethical approval for this study was obtained from the medical ethics committee at the College of Medicine, University of Sulaimani (198/29/08/2021/CoM/UoS). All participants provided written informed consent at the beginning of the study and were free to leave the study at any time. All patient details were de-identified.
Sample size estimation
The required sample size was calculated using Epi Info 7 software (free program issued by the US Centers for Disease Control and Prevention), 12 using the following information: number of women residing in Sulaimaniyah city (365,000), the expected prevalence of dense breasts was set at 50% to give the largest sample size, the degree of precision was established at 5%, and the confidence level was agreed at 99%. The required sample size was estimated at 662 women.
Procedure
Data were recorded using a specially designed case sheet including sociodemographic information on the participants and breast-density determinants. The study was conducted according to the relevant EQUATOR guidelines and reported in line with STROBE guidelines. 13
Bilateral standard two-view mammographic images (craniocaudal and mediolateral oblique projections) were obtained using a mammography unit (Senographe Essential, GE Healthcare, NY, USA). Two experienced radiologists in breast imaging assessed the MBD. Inter-examiner reliability was determined and calculation of the kappa value showed 96% agreement (indicating almost perfect agreement between the two examiners, with kappa between 0.81 and 1.00).
Authors K.A.S.A and S.M.F. then reviewed the mammograms and prepared the reports using a picture archiving and communication system. MBD was categorized into four densities: ACR-A, ACR-B, ACR-C, and ACR-D, based on the ACR BI-RADS atlas, 5th edition, 2013. BI-RADS assessment categories A and B were regarded as non-dense and categories C and D were regarded as dense breasts. In the case of differences between the two breasts, the BI-RADS breast density was allocated relying on the denser breast. Patient factors, including age, BMI, reproductive factors, family history of breast cancer (FHBC), oral contraceptive consumption (OCP), and hormone replacement therapy (HRT) were recorded and their correlations with breast density were analyzed.
Statistical analysis
Percentages were compared using χ2 tests and Fisher’s exact test if the expected frequency was <5% or >20% of the table's cells. All variables that were significantly correlated with breast density were entered into a binary logistic regression model with ‘presence of dense breast’ as a dependent variable. Statistical analyses were carried out using SPSS, version 25. A value of p ≤ 0.05 was considered statistically significant.
Results
Descriptive analysis
This study included four standard view mammograms for both breasts from 750 women (total 3000 mammograms). The mean (±standard deviation) participant age was 49.1 ± 7 years (median 48 years, range 40–75 years). Most women were aged 45 to 49 years (30%), followed by 40 to 44 years (29.2%), with fewer women aged >60 years (8.9%). Most (90%) women were married, 48.7% were obese, and 92% of the women were multiparous (Table 1).
Basic characteristics of the studied samples.

*The rest were single.
Factors associated with breast density
Inferential statistics showed that age and BMI were inversely associated with dense breasts (p < 0.001). Dense breasts were also significantly associated with menopausal status, a history of breastfeeding, and FHBC. However, there was no significant association between breast density and OCP or HRT (Table 2).
Correlations between breast density and participant factors
The incidence of dense breast was significantly higher among single women compared with married women. However, there was no significant association between breast density and parity, age at first delivery, or age at menarche (Table 3).
Participant factors associated with breast density.
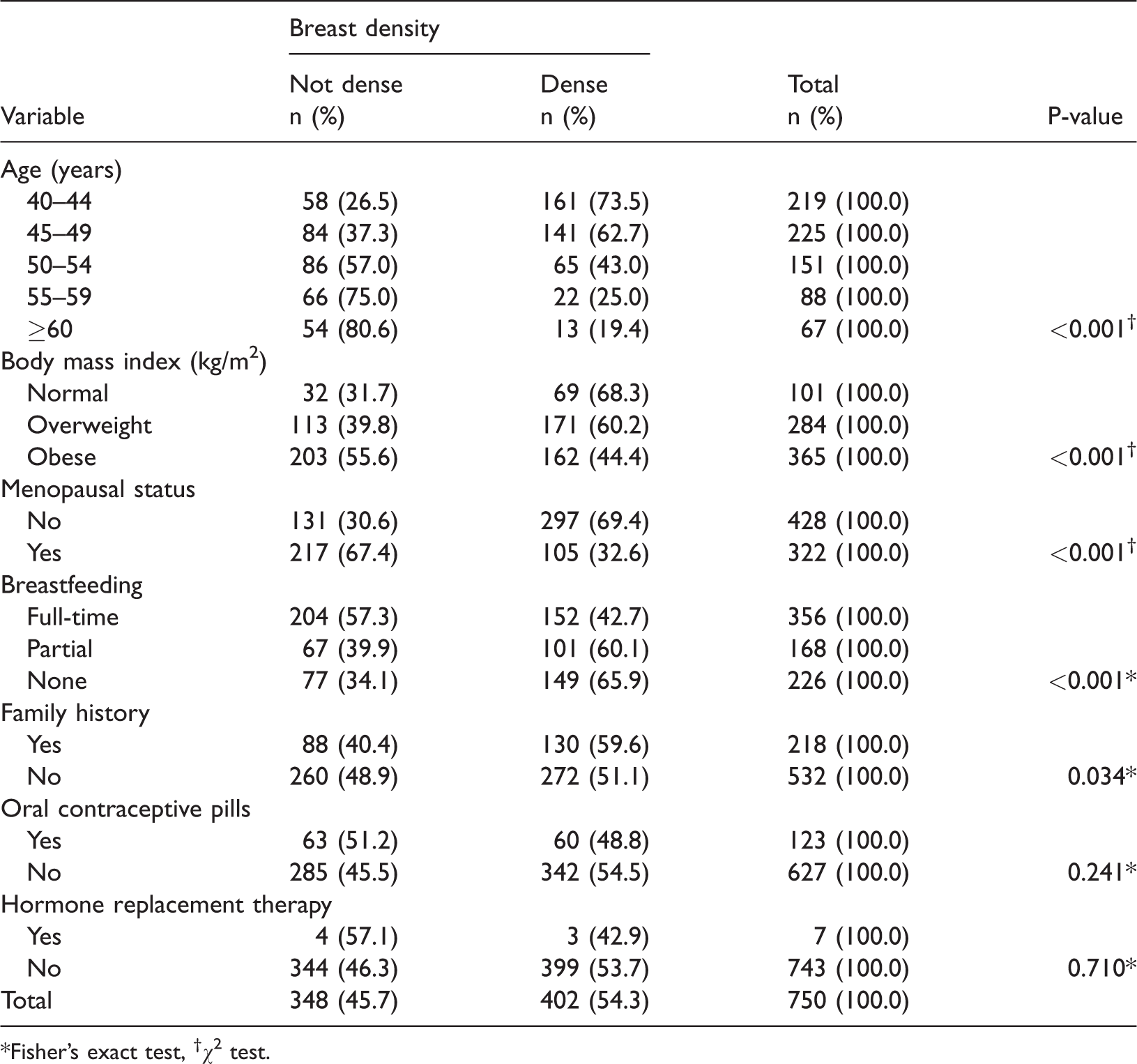
*Fisher’s exact test, †χ2 test.
Breast density according to marital status, parity, age at first delivery, and age at menarche.

*Fisher’s exact test, †χ2 test.
When the participants were analyzed according to menopausal status, there was still a significant correlation between breast density and marital status, with higher incidences of dense breasts among single women compared with married women for both non-menopausal and menopausal women (Table 4).
Association of breast density with marital status, parity, and age at first delivery among non-menopausal and menopausal women.

*Fisher’s exact test, †χ2 test.
Logistic regression analysis of prevalence of dense breasts in relation to covariants
The results of logistic regression analysis of the prevalence of dense breasts in relation to several covariants are shown in Table 5. Age <50 years was significantly correlated with dense breasts. Women with a normal BMI had a three-fold probability of having dense breasts compared with obese women, while overweight women had a two-fold higher probability. Non-menopausal women had a significantly higher likelihood of having dense breasts than menopausal women, and women with no history of breastfeeding or those who partially breastfed their babies had a higher probability of having dense breasts than women who breastfed their babies. Participants with FHBC had a significantly higher probability of having dense breasts. However, there was no significant association between marital status and dense breasts in logistic regression.
Logistic regression analysis between prevalence of dense breasts and several covariants.
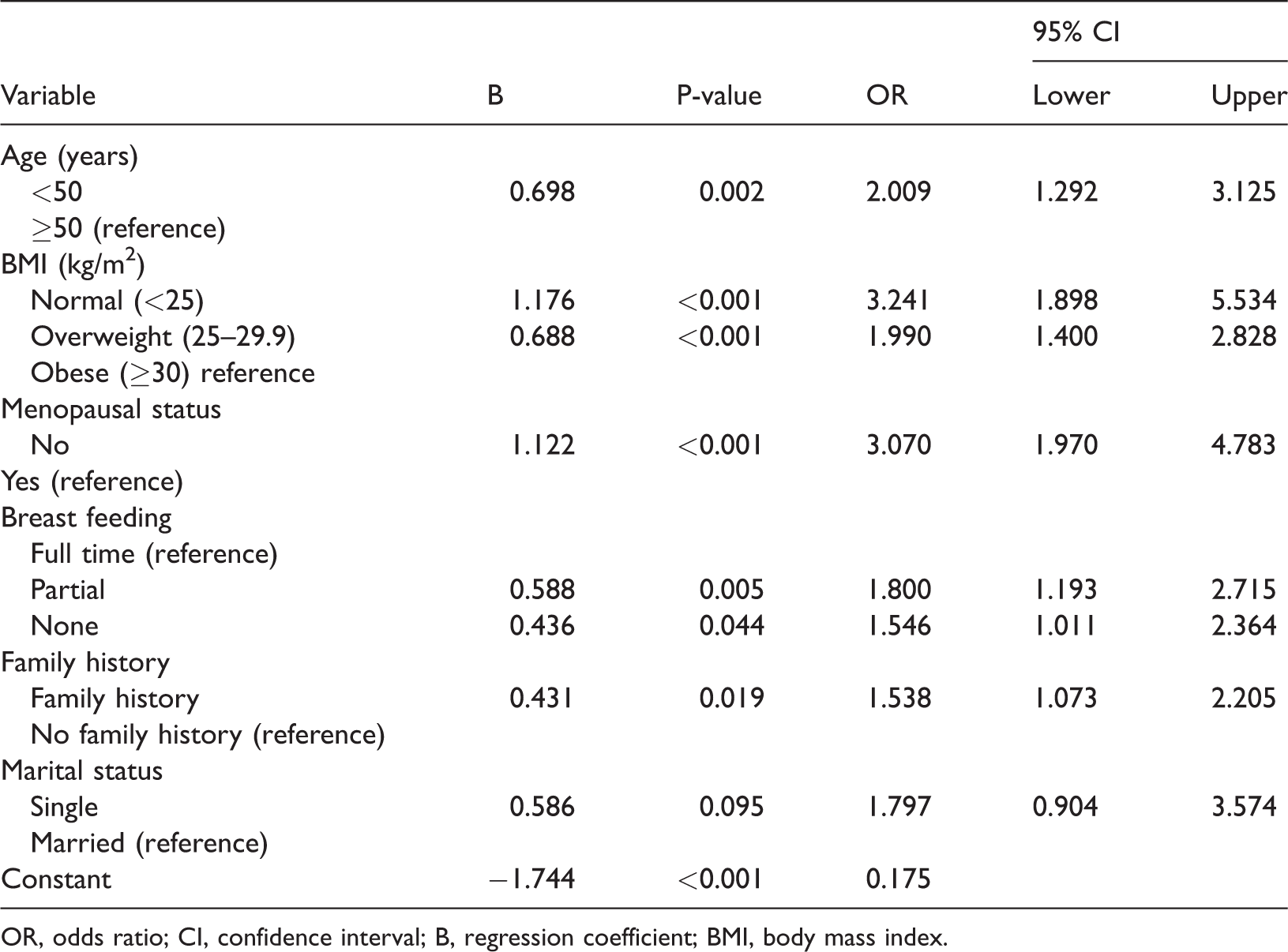
OR, odds ratio; CI, confidence interval; B, regression coefficient; BMI, body mass index.
Discussion
The Breast Disease Treatment Center in Sulaimaniyah city, Iraq, is the main center in Sulaimaniyah and includes breast radiologists, surgeons, physicians, and medical staff. Women visit this center for screening and diagnostic purposes. This cross-sectional study aimed to investigate the distribution of breast density among women in Sulaimaniyah city, and analyze the correlations between breast density and its potential risk factors.
Screening mammography of women aged 40 to 69 years was carried out in the present study according to the American Cancer Society. 14 Breast-density patterns were categorized according to the BI-RADS lexicon. Heterogeneous type C was the most frequent type, comprising >50% of the cases, and 54.3% of participants were categorized as ACR-BI-RADS C or D. These findings are similar to those of studies carried out in the USA (55.4%), 10 Lebanon (52.9%), 15 Iran (51.9%), 16 and China (52.8%), 17 but the percentage was higher than those in UAE (23.6%), 18 India (16%), 19 and Uganda (39%), 20 and lower than in Jordan (69.4%). 21
Regarding the age distribution of the participants, most were aged 45 to 49 years (30%), and breast density was inversely related to age. This is as expected given that ageing and decreasing levels of ovarian hormones lead to involution changes in the breast tissue, especially in post-menopausal women. The highest percentage of dense breasts (ACR-BI-RADS C and D) was found in women aged 40 to 44 years. We found a similar relationship for BMI, with an increased BMI related to decreased breast density. An elevated BMI is related to low estradiol levels, which is a potent enhancer of epithelial and stromal propagation in breast tissue, 22 and fatty tissue appears radiolucent on mammograms, leading to a lower breast-tissue density. 23 In this regard, Ishihara et al. found that BMI and delivery numbers were significantly associated with breast density in the Japanese population. BMI was not correlated with breast density during pre/postmenopause, and delivery numbers only significantly affected breast density in the postmenopausal stage, especially in women aged 50 to 60 years. 24
The present study reported that a longer period of breastfeeding was inversely associated with breast density, and 65.9% of women with dense breasts had no history of breastfeeding, compared with 42.7% of women with full-time breastfeeding. This suggests that breastfeeding might reduce the risk of breast tumors by altering the breast density. Similar outcomes were observed in studies by Yaghjyan et al. in Iraq, 25 Yang et al. in China, 26 and Ahmadinejad et al. in Iran, 16 but the findings were in contrast to those of Modugno et al. in the USA. 27 Nishiyama et al. concluded that MBD was a problematic issue for the development of breast cancer in Japanese women after accounting for other risk factors such as BMI, parity, and breastfeeding, but not age at menarche/first birth, or FHBC. Furthermore, this risk was particularly significant in postmenopausal women and women with higher BMI. 28
The incidence of dense breast was significantly higher among single women (77.3%) compared with married women (51%). However, although 50.4% of women with dense breasts were multiparous and 57.4% were nulliparous, the relationship between parity and breast density was not significant. This finding conflicts with those of Yaghjyan et al. in Iraq 25 and Ahmadinejad et al. in Iran. 16 The latter study found a significant positive association between breast density and parity (up to para 8). However, this apparent discrepancy may be due to differences in methodology, sample sizes, study locations, and ethnic groups.
Additionally, Woolcott et al. in the USA examined the impact of parity only in postmenopausal women, whereas most of the present study participants (60%–80%) were premenopausal. 29 Ochi et al. noted that parity was inversely correlated with dense breasts in pre/postmenopausal women, and nulliparous women should thus be aware of the increased risk of dense breasts. 30 However, the current results were mostly derived from young premenopausal women, and might thus differ from those in older and postmenopausal women.
In this study, the incidence of dense breasts was significantly higher among women with FHBC (59.6%) compared with those without FHBC (51.1%). Previous investigations also demonstrated that the correlation between breast density and the risk of breast cancer was higher in women with FHBC than in those without.31,32 However, Yang et al. reported that FHBC, breastfeeding, and the number of deliveries were not significantly correlated with dense breasts in Chinese women, while age and BMI were negatively related to MBD in premenopausal women. 26 Additionally, Kim et al. noted that dense breasts were associated with a high risk of breast tumors in pre/postmenopausal women. 33 However, Fatima et al. demonstrated no relationship of breast density with FHBC or age at menarche/first delivery in a study in Pakistan. 34 The present study also found no significant link between age at first birth/menarche and breast density, in accord with the results of Fatima et al. 34 In contrast however, Lim et al. showed that age at first birth interacted significantly with breast density and breast tumor risk among Korean women. 35
We found no relationship between OCP/HRT and breast density. However, these findings may be unreliable because of the small number of hormone users in this study and the lack of accurate data regarding the specific period of hormone usage. A further independent study focusing on this association is therefore required. In this respect, Lee and Oh reported that HRT for ≥2 years directly enhanced breast density in postmenopausal Korean women. They also found that parity, breastfeeding, and OCP affected the imaging outcomes of pre/postmenopausal women, while age, menarche at ≤15 years, ≤1 live birth, and former benign breast infection were associated with high breast density, regardless of menopausal status. 36
Collectively, women, especially those aged 50 to 74, should be informed about the risks of numerous screening examinations, additional imaging, their clinical automated BI-RADS breast density, and their overall breast tumor risk, to determine if they have dense breasts and their related risk of breast cancer, to allow them to consider routine biennial screening.37,38
Regarding the study limitations, it would have been more precise to measure breast density using automated methods; however, semi-automated/automated approaches are not currently available in the Kurdistan region of Iraq. Furthermore, there was selection bias because the study population was selected from a single institution and might thus not represent the target population as a whole; however, participants were selected from the only breast center in Sulaimaniyah, Iraq.
Conclusion
The distinct breast density profile of about 54% of the study population (categorized as ACR-BI-RADS C or D) indicates a significantly increased risk of breast cancer in these women. There were significant inverse relationships between breast density and age and BMI, and a significant positive association with FHBC. Higher breast density was also associated with no history of breastfeeding compared with partial or complete breastfeeding, and pre-menopausal women are more likely to have dense breasts than menopausal women. However, breast density was not significantly associated with parity, age at menarche/first birth, OCP, or HRT. However, further studies with larger sample sizes are needed to confirm the current findings.
Footnotes
Acknowledgement
The authors thank Prof. Dr. Namir Al-Taweel for assistance with the statistical analysis of the data.
Author contributions
SMH: Conceptualization; writing final manuscript. KASA: data collection and analysis; writing draft manuscript; writing final manuscript.
Data availability
All the necessary data will be available on request.
Declaration of conflict of interest
The authors declare that there are no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
