Abstract
Objectives
Internal tandem duplications of the Fms-like tyrosine kinase 3 gene (FLT3-ITD) and additional chromosomal abnormalities (ACA) are prognostic factors in patients with acute promyelocytic leukemia (APL). This study aimed to determine the effect of the association between FLT3-ITD and ACA in the prognosis of APL.
Methods
This was a retrospective cohort study including 60 patients with APL treated with all-trans retinoic acid (ATRA) and chemotherapy. Five-year overall survival (OS) and progression-free survival (PFS) were analyzed in patient groups according to the presence of FLT3-ITD and ACA.
Results
FLT3-ITD was an independent adverse factor for 5-year PFS, and ACA was an independent adverse factor for 5-year OS. There were significant differences in OS and PFS among the groups: FLT3-ITD-negative without ACA, FLT3-ITD-positive without ACA, FLT3-ITD-negative with ACA, and FLT3-ITD-positive with ACA. The OS times were 52.917, 45.813, 25.375, and 23.417 months, and the PFS times were 48.833, 38.563, 23.250, and 17.333 months, respectively.
Conclusion
FLT3-ITD and ACA are associated with the poorest OS and PFS outcomes in patients with APL treated with chemotherapy plus ATRA.
Keywords
Introduction
Acute promyelocytic leukemia (APL) is a subtype of acute myelogenous leukemia characterized by the proliferation of abnormal promyelocytic cells, with rearrangements involving the retinoic acid receptor alpha (RARα) located at 17q21. About 95% of patients with APL show the translocation (15;17)(q22;q21) including promyelocytic leukemia (PML)/RARα, 1 while the remaining patients show the nucleophosmin1 (NPM1)/RARα translocation (5;17)(q35;q21) or the promyelocytic leukemia zinc finger (PLZF)/RARα translocation (11;17)(q23;q21). 2 Abnormal promyelocytic cells with the (15;17)(q22;q21) translocation are considered to be susceptible to all-trans retinoic acid (ATRA) and arsenic trioxide (ATO), and APL is thus considered to be a curable malignant disease. 3 However, in addition to this major translocation, patients with APL may have other chromosomal abnormalities that may interfere with the therapeutic efficacy of these specific drugs. Previous studies found that these additional chromosomal abnormalities (ACA) were associated with poorer treatment outcomes and survival times.4–6 Unfortunately, a relatively high percentage of patients with APL (29%–43%) may have ACA at diagnosis. 7
Internal tandem duplication of the Fms-like tyrosine kinase 3 gene (FLT3-ITD) is associated with increased survival and proliferation of hematopoietic progenitors and is related to leukocytosis in patients with APL, and thus plays a role in the pathogenesis of APL.8–10Given the high incidence of FLT3-ITD in APL (13%–40%), it is important to understand its potential effect on patient survival.11,12 However, few studies have investigated the association between FLT3-ITD and ACA and their effect on survival in patients with APL. The current study aimed to determine the role of the association between FLT3-ITD and ACA in the prognosis of APL.
Materials and Methods
Patients
This was a retrospective cohort study conducted at the Center for Hematology and Blood Transfusion, Bach Mai Hospital, Hanoi, Vietnam, from January 2015 to December 2019. The study enrolled all consecutive patients diagnosed with new APL according to the FAB classification with positive PML/RARα, who were treated with chemotherapy plus ATRA. 13
The Institutional Review Board of Hanoi Medical University waived the need for approval and patient consent because of the retrospective observational nature of the study. All patient details were de-identified.
Reverse transcription polymerase chain reaction
Bone marrow samples obtained from all patients at the time of diagnosis were analyzed by reverse transcription polymerase chain reaction to detect PML/RARα and FLT3-ITD.
Cytogenetics
Bone marrow samples obtained at diagnosis were also subjected to chromosome analysis to detect t(15;17)(q21;q22) and ACA.
Treatment
Patients were treated with an induction, consolidation, and maintenance regimen. The induction regimen consisted of oral ATRA (twice daily, 45 mg/m2/day) until complete remission, with daunorubicin (45 mg/m2/days 1–3) in the low/intermediate-risk group or daunorubicin (45 mg/m2/days 1–3) + cytarabine (100 mg/m2/days 1–7) in the high-risk group. The consolidation regimen consisted of daunorubicin (45 mg/m2/days 1–3) and ATRA (45 mg/m2/day for 15 days) for two to three courses, monthly. Maintenance therapy with oral ATRA (45 mg/m2/day for 15 days every 3 months) and oral mercaptopurine (60 mg/m2/day) was administered for 2 years.
Definition
Risk groups were classified according to the Sanz score. 14 Disseminated intravascular coagulation (DIC) was defined according to the criteria of the International Society for Thrombosis and Hemostasis. 15 The response criteria to induction therapy were determined according to the International Working Group criteria. 16
Statistical analysis
The patients were grouped according to the presence of FLT3-ITD and ACA. Patients were further divided into three groups based on the number of ACA: no ACA, one ACA, two or more ACA. Differences in quantitative variables (hemoglobin, white blood cells, platelets, fibrinogen, D-dimer, bone marrow cell count, bone marrow blast percent) among groups were analyzed by one-way ANOVA, and differences in qualitative variables (DIC and risk group) were analyzed by χ2 or Fisher’s exact test. Univariate and multivariate analyses were performed using the Kaplan–Meier method and Cox proportional hazards model to identify independent prognostic factors for overall survival (OS) and progression-free survival (PFS). OS was defined as the time from diagnosis to the last follow-up, or death, and PFS was defined as the time from remission to relapse or death.
There were no missing data in this study and the bias was therefore controlled. The reporting of this study conforms to the STROBE guidelines. 17
Results
Clinical data
Sixty patients were included in the study and their data were analyzed retrospectively. There were 28 men (46.7%) and the mean age was 38.6 years (range: 15–68 years). The characteristics of the patients are presented in Tables 1 and 2. There were no significant differences in laboratory indices, except for Hb levels, including the distribution of risk groups or the frequency of DIC, between the groups of patients according to the presence of FLT3-ITD and ACA.
Laboratory indices in patients according to presence of FLT3-ITD and additional chromosomal abnormalities.

ACA, additional chromosomal abnormalities; SD, standard deviation; SE, standard error; Hb, hemoglobin; WBC, white blood cells; FEU, fibrinogen equivalent units.
Characteristics of patients according to the presence of FLT3-ITD and additional chromosomal abnormalities.
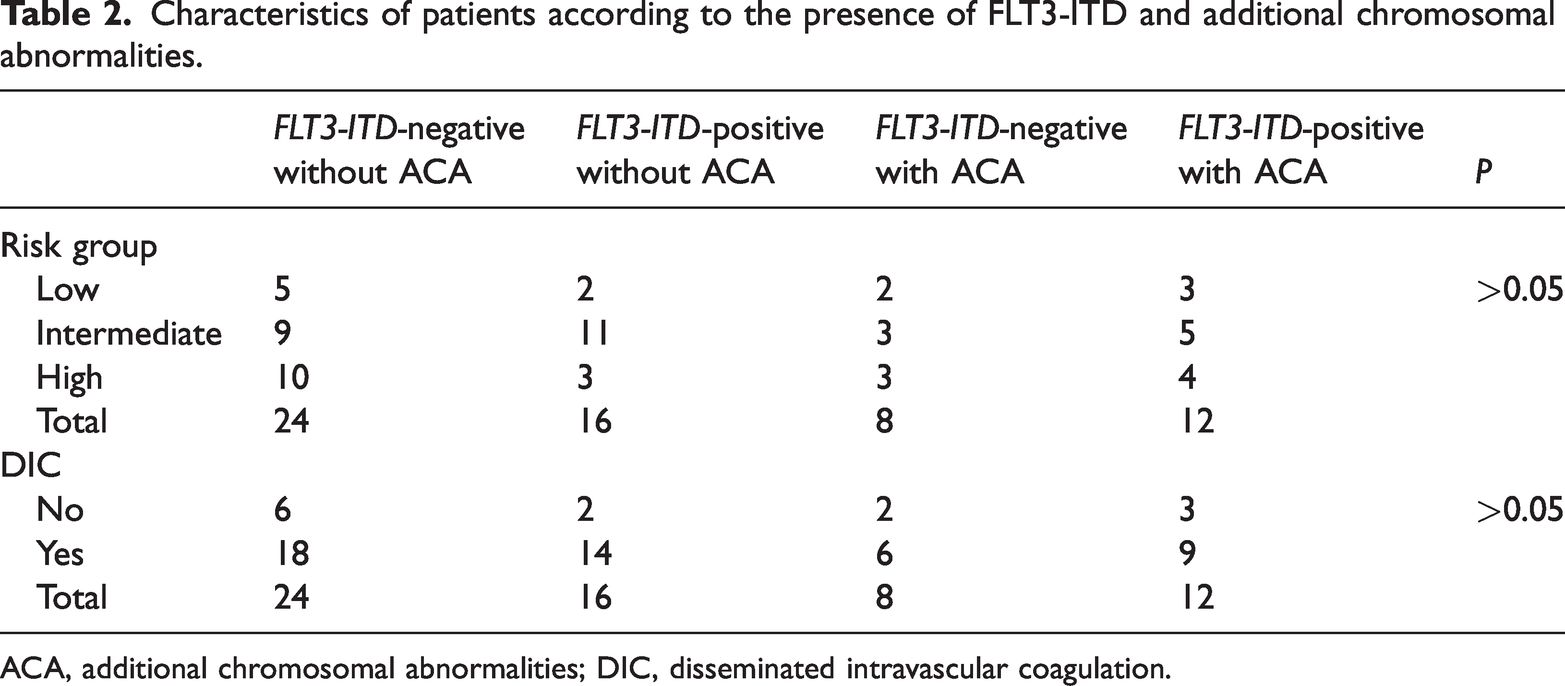
ACA, additional chromosomal abnormalities; DIC, disseminated intravascular coagulation.
Survival analysis
Univariate analysis showed that both FLT3-ITD and ACA were associated with poor 5-year OS and 5-year PFS (FLT3-ITD: P = 0.027, P = 0.008; respectively; ACA: P = 0.007, P = 0.015; respectively) (Table 3). However, multivariate analysis indicated that FLT3-ITD was only an adverse prognostic factor for PFS (P = 0.032, hazard ratio [HR] = 2.854, 95% confidence interval [CI]: 1.095–7.437) while ACA was only an independent adverse prognostic factor for OS (P = 0.033, HR = 2.905, 95% CI: 1.090–7.746) (Table 3).
Univariate and multivariate analyses of survival times.

ACA, additional chromosomal abnormalities; OS, overall survival; HR, hazard ratio; CI, confidence interval; PFS, progression-free survival.
aLog-rank; bCox regression.
We also analyzed the survival time of the patient groups based on the number of ACA (no ACA, only one ACA, ≥2 ACA). OS and PFS differed significantly among the three groups according to univariate analysis (P = 0.02, P = 0.019; respectively), but multivariate analysis found no significant difference (Table 3).
It should be noted that 5-year OS and 5-year PFS differed significantly among the following groups of patients: FLT3-ITD-negative without ACA, FLT3-ITD-positive without ACA, FLT3-ITD-negative with ACA, and FLT3-ITD-positive with ACA (P = 0.006, P = 0.003; respectively). The OS times were 52.917, 45.813, 25.375, and 23.417 months, respectively, and the PFS times were 48.833, 38.563, 23.250, and 17.333 months, respectively. FLT3-ITD-positive patients with ACA had the worst OS and PFS (Table 4, Figures 1 and 2).
Overall and progression-free survival according to presence of FLT3-ITD and additional chromosomal abnormalities.

ACA, additional chromosomal abnormalities; OS, overall survival; PFS, progression-free survival.

Cumulative overall survival (Cum Survival) according to the presence of FLT3-ITD and additional chromosomal abnormalities (ACA).

Cumulative progression-free survival (Cum Survival) according to the presence of FLT3-ITD and additional chromosomal abnormalities (ACA).
Discussion
The PML/RARα fusion gene derived from translocation t(15;17)(q22;q21) generates the oncogenic PML/RARα fusion protein, which is considered to act as the main pathogenic factor in APL via deregulation of transcriptional control of the RARα gene and disruption of PML function. 18 ATRA is the main drug used to treat APL. Its mechanisms of action include relocalizing PML and degrading PML/RARα protein, and converting PML/RARα from a transcription inhibitor to a transcription activator. 3 However, ACA and/or FLT3-ITD may be associated with other comorbid mechanisms, resulting in reduced treatment efficacy and survival.
Pantic et al. showed that ACA was an independent adverse factor for survival time in patients with APL treated with ATRA, 19 and Wiernik et al. also suggested that APL patients without ACA had better OS and disease-free survival (DFS) than those with ACA, following treatment with ATRA. 20 Cervera et al. showed that ACA was associated with lower relapse-free survival according to univariate analysis, but there was no significant association in multivariate analysis. 21
Epstein-Peterson et al. suggested that ACA was not always an adverse prognostic factor for event-free survival in patients with APL treated with ATO, but did have an adverse effect in cases with a complex karyotype, 22 while Chen et al. found that an abnormal karyotype was associated with a high risk of early mortality, even in patients treated with ATO. 23 Poiré et al. also suggested that patients with ACA had poorer OS, despite ATO therapy. 24
Overall, these studies suggest that ACA may be an adverse factor affecting survival time. The current multivariate analysis showed that ACA was an independent adverse prognostic factor for OS but not PFS in patients with APL treated with ATRA plus chemotherapy. This result appears to be similar to that of Pantic et al. and Wiernik et al.,19,20 suggesting that ATRA has little effect on ACA.
The FLT3-ITD mutation is associated with a poor prognosis in patients with acute myelogenous leukemia with a normal karyotype. 8 However, FLT3-ITD is often present in patients with APL and is associated with hyperleukocytosis, suggesting that it is also an adverse risk factor for treatment and survival outcomes in APL. 1
Breccia et al. showed that FLT3-ITD was an independent unfavorable factor for OS, relapse-free survival, and DFS in patients with APL treated with ATRA plus chemotherapy, 11 but Lucena-Araujo et al. suggested that this mutation only had an impact on OS, but not on DFS. 25 However, Singh et al. compared patients with FLT3-ITD and wild-type FLT3 and concluded that FLT3-ITD was a poor prognostic factor for DFS, but not for OS. 26
Poiré et al. and Deka et al. assumed that FLT3-ITD had no effect on OS or DFS in patients treated with ATO,24,27 while Song et al. suggested that FLT3-ITD was an adverse factor in terms of OS and event-free survival, even after ATO therapy. 28
These studies have thus produced controversial results regarding the role of FLT3-ITD as a poor prognostic factor, regardless of ATRA or ATO treatment. However, the current multivariate analysis suggested that FLT3-ITD was an independent adverse prognostic factor for PFS, but not for OS, in accord with the findings of Lucena-Araujo et al. 25
It is generally difficult to treat patients with APL with ACA or FLT3-ITD. Furthermore, the prognostic effect of the combination of these two abnormalities is unclear, and studies examining the effects of these two factors on survival time using multivariate analysis are lacking. We therefore analyzed the effect of the association between FLT3-ITD and ACA on survival time using the Kaplan–Meier method and log rank test. We found significant differences in both OS and PFS among the groups: FLT3-ITD-negative without ACA, FLT3-ITD-positive without ACA, FLT3-ITD-negative with ACA, and FLT3-ITD-positive with ACA (P = 0.006, P = 0.003; respectively), with FLT3-ITD-positive patients with ACA having the worst OS and PFS. Appropriate treatment strategies thus need to be considered for patients with both FLT3-ITD and ACA.
Our study had some limitations. The European Leukemia Network 2019 considers normal chromosomal acute myelogenous leukemia with NPM1mut, without FLT3-ITD or with low-level FLT3-ITD, as having a favorable prognosis. 29 Low levels of FLT3-ITD may thus not cause adverse effects, and the adverse prognostic influence of FLT3-ITD may also depends on its expression level. This may also help to explain some of apparently conflicting results regarding the prognostic significance of FLT3-ITD. Unfortunately, we did not quantify FLT3-ITD levels in the current study. As noted above, some studies showed that FLT3-ITD had no effect on survival time in patients treated with the ATO regimen.24,27 The prognostic factors thus change in line with advancements and changes in treatments, and different prognostic factors should be analyzed and applied at different stages of treatment development. The current study is applicable to patients treated with the ATRA regimen; however, further studies are needed in patients treated with the ATO regimen. More research is also needed to analyze treatment outcomes in relation to FLT3-ITD levels.
Conclusion
FLT3-ITD may be an independent adverse prognostic factor for PFS and ACA may be an independent adverse prognostic factor for OS in APL patients treated with chemotherapy plus ATRA, with FLT3-ITD-positive patients with ACA having the worst OS and PFS times.
Research Data
Research Data for Association between FLT3-ITD and additional chromosomal abnormalities in the prognosis of acute promyelocytic leukemia
Research Data for Association between FLT3-ITD and additional chromosomal abnormalities in the prognosis of acute promyelocytic leukemia by Minh Phuong Vu, Cuc Nhung Nguyen, Hoang Vu, Tuyet Mai Nguyen, Tuan Tung Nguyen and Phuong Thao Pham in Journal of International Medical Research
Footnotes
Acknowledgements
The authors thank all the technicians in the Genetics and Molecular Biology Laboratory, Center for Hematology and Blood Transfusion, Bach Mai Hospital, for their efforts in supporting this research.
Author contributions
Minh Phuong Vu: conceptualization, formal analysis, methodology, writing – original draft, writing – review & editing; Cuc Nhung Nguyen: formal analysis, investigation, methodology, writing – original draft; Hoang Vu: investigation, writing – original draft; Tuyet Mai Nguyen: formal analysis, writing – original draft; Tuan Tung Nguyen: methodology, writing – original draft; Phuong Thao Pham: formal analysis, investigation, Writing – original draft. All authors approved the version to be published
Data availability
The dataset used for this paper is available upon reasonable request.
Declaration of conflicting interests
The authors declare no conflicting interests in preparing this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
