Abstract
Background
The diagnosis of constrictive pericarditis (CP) is challenging as there are currently no standard echocardiographic diagnostic criteria. In this retrospective case series, we analyzed and summarized the features of 25 patients with CP and proposed echocardiographic diagnostic criteria. It is hoped that the suggested criteria help professionals make decisions in their daily practice so that patients receive timely diagnosis and effective treatment.
Methods
Twenty-five patients with CP were selected for this retrospective study. The clinical and echocardiographic imaging data were analyzed and summarized, and echocardiographic diagnostic criteria for CP were proposed.
Results
The main clinical manifestations were fatigue, breathlessness, exertional dyspnea (88%), lower-limb edema (84%), hepatomegaly, and jugular vein filling (84%). Echocardiographic features comprised pericardial thickening (88%) and calcification (60%), pulmonary hypertension (52%), inferior vena cava dilation (80%), left and/or right atrial enlargement (100%), diastolic flattening of the left ventricular (LV) posterior wall (72%), septal shudder and bounce (64%), restrictive LV and right ventricular diastolic filling pattern (100%), early filling changes of mitral and tricuspid flow (80% and 60%, respectively), and mitral annulus reversus (73%).
Conclusions
Echocardiography is a simple and valuable examination for CP. The echocardiographic diagnostic criteria are valid and worth promoting.
Keywords
Introduction
Constrictive pericarditis (CP) is characterized by restricted diastolic filling of the ventricles that is caused by thickening, fibrosis, and calcification of the pericardium, with or without myocardial fibrosis or atrophy. The reported incidence rate of pericarditis is 27.7 per 100,000 people, and approximately 1.8% of acute pericarditis patients progress to CP. 1 The most common cause of CP is idiopathic or viral, accounting for 42% to 49% of all CP cases. 1 In developed countries, other causes include post-cardiac surgery (11%–37%), postradiation therapy (9%–31%), connective tissue disorders (3%–7%), post-infectious causes (tuberculosis (TB) or purulent pericarditis, 3%–6%), and miscellaneous causes (malignancy, trauma, drug-induced, asbestosis, sarcoidosis, and uremic pericarditis (<10%). 1
The diagnosis of CP is challenging. Patients often experience fatigue, breathlessness, exertional dyspnea, abdominal distension, peripheral edema, pleural effusion, or ascites. However, these presentations lack specificity. Multimodal cardiovascular imaging comprising echocardiography, computed tomography (CT), and cardiac magnetic resonance (CMR) imaging, is recommended in patients suspected of having CP. 2 Echocardiography is a simple and valuable examination for accurate evaluation of cardiac function; however, there is currently no diagnostic standard for CP. This likely explains why the diagnostic accuracy of CP is relatively low at approximately 51%–73%. 3
This case series is based on a retrospective analysis of 25 patients with CP confirmed by surgery and multimodal imaging in our hospital. Additionally, possible CP diagnostic criteria are proposed to optimize the diagnostic approach and enable early diagnosis and effective treatment.
Methods
Twenty-five patients diagnosed as having chronic CP who were evaluated at our hospital from August 2010 to December 2021 were selected, namely 15 men and 10 women, aged 25 to 78 years. Twenty-three patients were diagnosed by multimodal imaging. Among these 23 patients, 21 underwent pericardiectomy, and CP was confirmed by pathology; 2 cases were treated conservatively. The two remaining cases were missed diagnoses that were confirmed by pathology during cardiac surgery. The patients’ in-hospital and follow-up clinical and paraclinical data were collected from the medical records, physical examination findings, epidemiological data, echocardiographic data, and additional test results (i.e., thoracic radiography and cardiac imaging data). The imaging equipment used for echocardiographic examination were Phillips Epic 7C (Philips Medical Systems, Bothell, WA, USA), Siemens SC2000 (Siemens Medical Solutions USA, Inc., Mountain View, CA, USA), and Vivid 7 (GE Healthcare, Milwaukee, WI, USA). Routine two-dimensional, M-mode, pulse wave Doppler, and tissue Doppler echocardiography were performed. Continuous variables are expressed as mean ± standard deviation (SD), and the categorical variables are presented as percentages.
The Institutional Review Board (IRB) and Ethics Committee of the Qingdao University Medical School, Qingdao, China, approved this study, and we have de-identified all patient details. All patients provided written informed consent to participate in the study. The reporting of this study conforms to the STROBE guidelines. 4
Results
Fatigue, breathlessness, and exertional dyspnea were the most frequent symptoms and these occurred in 22 patients (88%), among which 10 patients (40%) experienced recurrent attacks. Edema of the lower limbs occurred in 16 patients (84%), and hepatomegaly and jugular vein filling occurred in 16 patients (84%). Four patients (16%) had pleural effusion and ascites. Five patients (20%) had a history of confirmed other symptoms, such as cough or palpitation.
Normal pericardial thickness (2 mm) was measured in three cases. Pericardial thickening was identified in 22 cases (88%), and the thickness ranged from 0.3 cm to 1.2 cm (0.61 ± 0.58 cm). The most commonly thickened sites were the posterior and inferior walls of the left ventricle (LV) (n = 15, 60%). Diffuse thickening was identified in 2 cases (8%), and pericardial calcification was identified in 15 cases (60%) in different areas and to varying degrees (Figure 1).

Pericardial calcification (yellow arrow) and the resulting abnormal cardiac morphology in a patient with CP.
Severe abnormal cardiac morphology (Figure 1) was identified in 2 cases (8%) owing to severe thickening and calcification of the pericardium. Almost all cases had left and/or right atrial enlargement, and 5 cases (20%) had small ventricles. Severe mitral regurgitation was identified in one patient (4%), which was owing to abnormal cardiac morphology caused by severe pericardial calcification. The remaining patients had mild-to-moderate mitral and tricuspid regurgitation. Pulmonary artery systolic pressure (PASP) ranged from 22 mmHg to 55 mmHg (37.4 ± 17.84 mmHg), and pulmonary hypertension was identified in 13 cases (52%). The inferior vena cava (IVC) was dilated to different degrees (2.5 ± 1.35 cm) in 20 cases (80%) (Figure 2).

M-mode echocardiography of a patient with CP. An obvious V-shaped cut (yellow arrow) is seen.
Diastolic flattening of the LV posterior endocardial wall was identified in 18 patients (72%). Abrupt posterior motion of the ventricular septum in early diastole (septal shudder and bounce) was seen in 16 patients (64%). Among these 17 patients, 13 (52%) showed an obvious V-shaped cut (Figure 2). Ventricular septal respiratory drift, indicated by the ventricular septum moving toward the LV with inspiration and toward the right ventricle (RV) with expiration, was identified in 9 patients (36%). All patients showed restrictive LV and RV diastolic filling patterns. Fifteen patients (60%) had high early (E) velocity values and E/A ratios >2, 18 patients (72%) had deceleration time (DT) values <160 ms (Figure 3), 4 patients (16%) had E/A ratios <1, and 6 patients (24%) had E/A ratios >1. The respiratory variability of mitral and tricuspid inflow velocities could not be measured in patients with atrial fibrillation. Change in mitral inflow velocity of >25% was identified in 16 patients (80%). The data for the tricuspid inflow velocity pulse waves indicated variability rates >40% during expiration and inspiration in 12 patients (60%) (Figure 4).
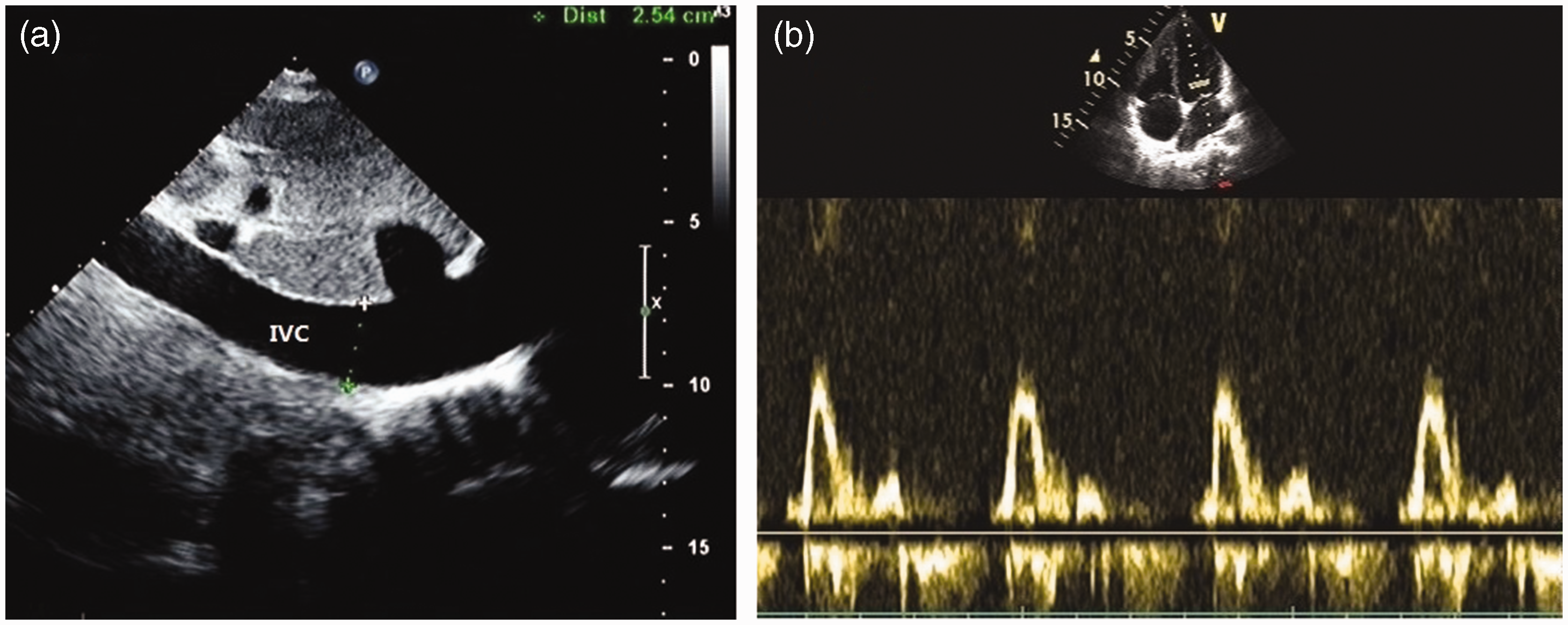
Restrictive ventricular diastolic filling pattern in a patient with CP; (a) dilated IVC (2.5 cm) in the subxiphoid view and (b) mitral inflow wave: E/A: 3.6, DT: 71 ms.

Mitral and tricuspid inflow pulse waves in patients with CP. The variability rates in expiration and inspiration were 34% (mitral inflow) (a) and 40% (tricuspid inflow) (b), respectively.
Early diastolic velocity (e′) data from the medial and lateral mitral annulus by tissue Doppler were obtained in 15 patients. A normal or increased mitral media annulus velocity (e′ ≥ 8 cm/s) was seen in 12 patients (80%). Mitral annulus reversus, defined as a lateral mitral annulus e′ that is usually lower than e′ from the medial annulus, was identified in 11 patients (73%) (Figure 5).
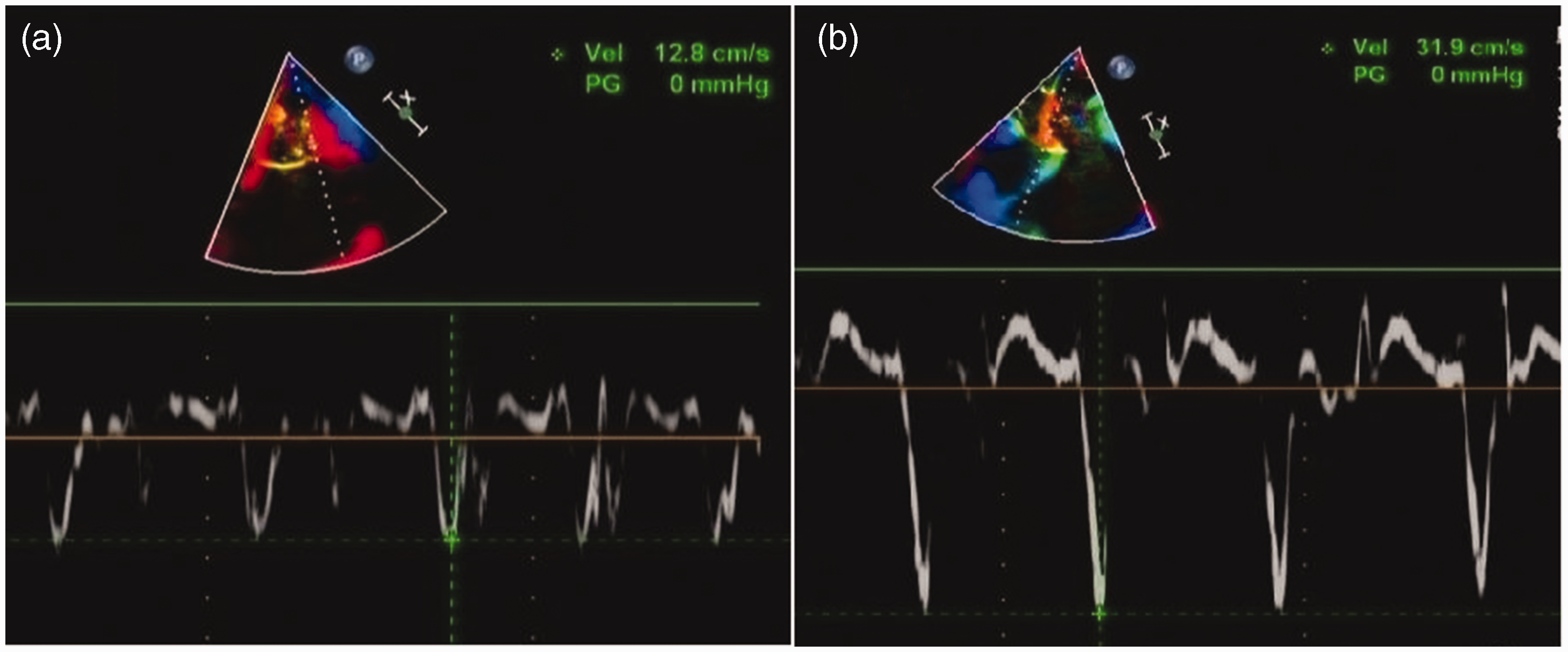
Tissue Doppler imaging in a patient with CP: (a) e′ from the lateral mitral annulus was 12.8 cm/s and (b) e′ from the medial mitral annulus was 31.9 cm/s. These findings indicate mitral annulus reversus.
Discussion
CP is a condition in which pericardial thickening, adhesions, and often calcification, limit diastolic filling of the ventricles. 5 Pericarditis may impact the epicardial myocardium, and chronically, this pathological condition may lead to myocardial atrophy, fibrosis, fat infiltration, and calcification. 6 Furthermore, thickening and calcification of the pericardium compresses the coronary arteries, resulting in decreased blood flow, which leads to myocardial ischemia. 7 Chronic CP, myocardial degeneration, and ischemia may lead to irreversible cardiac dysfunction; therefore, it is very important to make an early diagnosis and perform pericardiectomy.
The normal pericardium is less than 2 mm thick. In patients with CP, the pericardium loses elasticity and is replaced by dense fibrous tissue with calcification. This rigid shell limits ventricular filling. Echocardiography is usually the initial diagnostic procedure in patients with suspected CP. Pericardial thickening and calcification and abnormal ventricular filling patterns can be measured using this method. The characteristic changes on M-mode echocardiography are flattening of the LV posterior wall in mid-to-late diastole and abrupt posterior motion of the ventricular septum in early diastole with inspiration (septal shudder and bounce). 8 In this study, most patients had diastolic flattening of the LV posterior wall, and the pericardium was especially damaged in this location.
In CP, ventricular relaxation is limited and is caused by obstruction of venous reflux, resulting in jugular and inferior vena cava venous distension. Blood flows into the ventricles from the atria very quickly in early diastole owing to the increased pressure in the atria. However, ventricular relaxation is suddenly restricted by the inelastic pericardium in mid-to-late diastole, and the pressure in the ventricles rises rapidly. Thereby, the pressure gradient between the atria and ventricles quickly reaches equilibrium. The characteristic changes on pulse wave Doppler are high E velocity, short DT, and reduced atrial (A) wave. 9 All patients in this study showed varying degrees of restrictive ventricular filling disorder. Because the ventricles are located in a “fixed” space, the interaction between them is strengthened. Septal shudder and bounce in early diastole, a V-shaped cut, and ventricular septal respiratory drift can be seen in CP. 10
Restricted by the thickened and calcified pericardium, the radial and rotational movements of the ventricles decrease. Therefore, the movement of the ventricular long axis is enhanced, as compensation. A mitral media annulus velocity (e′ ≥ 8 cm/s) on tissue Doppler imaging that is in the normal range or increased can be detected. 11 This might be a clue that early constriction is present, especially for elderly patients, and is the opposite of what is seen with other causes of heart failure. 12 Additionally, adhesions and pathological changes in the LV lateral pericardium further decrease LV motion. Furthermore, mitral annulus reversus can be seen on tissue Doppler imaging, which is consistent with strain analysis. 13 In the traceable tissue Doppler data in this study, the incidence of annulus reversus was low, which was related to the location of the pericardial adhesions and the degree of damage to the epicardium and myocardium.
Usually, the pressure in the pericardial cavity changes with the pressure in the thoracic cavity during breathing. In patients with CP, the inflexible pericardium prevents the pressure change in the thoracic cavity from being transmitted to the pericardial cavity, which leads to a pressure difference between the thoracic and cardiac cavities. At the end of inspiration, the pressure in the pulmonary vein decreases, and the gradient from the pulmonary vein to the left atrium (LA) also decreases. In response, the filling volumes of the LA and LV decrease. In contrast, at the end of expiration, the RV filling pressure increases, and the pressure gradient between the RA and RV decreases, then the RV filling volume decreases. Mitral inflow velocity may decrease by as much as 25% to 40%, and tricuspid velocity increases dramatically (>40%) during the first beat after inspiration. 14 Mitral and tricuspid inflow velocity changes were found in most patients (80% and 60%, respectively) in this study.
The pathological changes and ultrasonographic findings in patients with CP were summarized in this study. The underlying pathological changes in CP comprise pericardial lesions (thickening, adhesions, fibrosis, or calcification), heart morphological and structural changes, pathophysiological triad (ventricular dependence, thoracic-intracardiac pressure difference, compensatory enhancement of long axis motion in early diastole), and a restrictive pattern of ventricular diastolic filling, which is the final result of the pathological factors. Different pathological changes manifested as different ultrasonographic characteristics, in this study (Table 1).
Basic pathological changes and echocardiographic findings in CP.

CP, constrictive pericarditis; LV, left ventricle; RV, right ventricle; E/A, early (E) velocity to atrial (A) wave ratio; DT, deceleration time; IVC, inferior vena cava.
Our results showed that even patients with a confirmed diagnosis of CP may not have every typical ultrasonographic manifestation, even when they undergo two-dimensional (2D), M-mode, and tissue and pulse Doppler examinations. Furthermore, it is challenging to make a correct diagnosis for patients with atypical clinical and echocardiographic features and those in the early stages of CP. Unfortunately, the ultrasonographic diagnostic criteria of CP are not clearly defined in the current guidelines and in previous studies. On the basis of the results of this study, we proposed the following echocardiographic diagnostic criteria for CP: evidence of a restrictive pattern of ventricular diastolic filling combined with one ultrasonographic manifestation: suspicious for CP; restrictive pattern combined with three ultrasonographic manifestations: confirmed CP; and restrictive pattern combined with two ultrasonographic manifestations: highly suspicious for CP (Table 2). All patients in this study met these diagnostic criteria well, namely 15 confirmed cases, 8 highly suspicious cases, and 2 suspicious cases (misdiagnosed cases). It should be emphasized that we discussed only the echocardiographic diagnostic criteria for CP in this study. The clinical diagnosis of CP should not ignore the importance of other imaging techniques, such as CT or CMR imaging. Comprehensive evaluation of multimodal imaging findings is more conducive to making a correct diagnosis than relying on a single imaging modality.
Echocardiographic diagnostic criteria for constrictive pericarditis.

Echo, echocardiography; CT, computed tomography; LV, left ventricle; RV, right ventricle; E/A, early (E) velocity to atrial (A) wave ratio; DT, deceleration time; IVC, inferior vena cava.
Conclusion
CP is a serious clinical syndrome that is not easy to diagnose. Based on this retrospective case series, effective echocardiographic diagnostic criteria were proposed. We hope that the criteria will be adopted widely to facilitate the standardized diagnosis of CP. Thereby, patients can be diagnosed correctly and treated effectively.
Research Data
Research Data for A case series of constrictive pericarditis and suggested echocardiographic diagnostic criteria
Research Data for A case series of constrictive pericarditis and suggested echocardiographic diagnostic criteria by Junfang Li, Rong Li, Guangting Cheng, Changhong Lu, Weigang Liu, Dongmei Sun, Xue Li and Zhibin Wang in Journal of International Medical Research
Footnotes
Data availability statement
All data supporting the conclusions of this article are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
Author contributions
JL and CL conceptualized the study and reviewed the manuscript. RL and GC acquired and analyzed the data and critically reviewed the manuscript. DS, WL, and XL acquired and analyzed the data. ZW reviewed and finalized the manuscript. All authors had full access and take full responsibility for the data. All authors read and approved the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
