Abstract
With the extensive use of immune checkpoint inhibitors (ICI) in advanced-stage cancers, immune-related adverse events (irAEs) have been noted in various systems. While most irAEs are reversible and manageable, cardiac toxicities are rare but life-threatening, with high mortality rates. We present a case of a 71-year-old man with cholangiocarcinoma who developed myocarditis related to ICIs 29 days after the first infusion of teriprizumab combined with albumin-bound paclitaxel and gemcitabine. He was initially asymptomatic after admission but with substantial elevations of troponin I and myocardial enzymes. Sixteen hours after admission, he developed palpitations, dizziness, and syncope. Electrocardiography confirmed third-degree atrioventricular block and frequent ventricular premature contractions for which he received high-dose corticosteroids and a permanent pacemaker. The patient survived and permanently discontinued immunotherapy. Early identification and intervention are the keys to improving the prognosis of immune myocarditis.
Keywords
Introduction
Immune checkpoint inhibitors (ICI) have revolutionized the treatment of cancers by removing immune suppression by cancer cells, thus achieving tumor regression. However, the augmented immune autoreaction can affect any organ or system, creating a variety of toxicities, such as colitis, hepatitis, pneumonitis, thyroiditis, myositis, hypophysitis, and dermatitis. Although ICI-associated myocarditis is rare, with an incidence of 0.27% to 1.14%, 1 the condition is serious, with a morbidity rate of up to 50%. 2 Additionally, the combination of ICI and non-ICI therapies increases the risk, severity, and fatality of myocarditis resulting from ICI monotherapy. 3 Teriprizumab, a human monoclonal antibody targeting programmed death-1 (PD-1), was developed and approved in China in December 2018. Here, we describe a patient with metastatic cholangiocarcinoma who developed ICI-induced myocarditis after receiving the first cycle of teriprizumab and chemotherapy.
Case Report
The reporting of this study conforms to the CARE guidelines. 4
A 71-year-old Chinese man was diagnosed with intrahepatic cholangiocarcinoma in the second hepatic portal (stage II, clinical tumor-node-metastasis stage: cT2N0M0) on 20 October 2021, in Shanghai Zhongshan Hospital. The pathological results of a liver biopsy indicated small bile duct cholangiocarcinoma, grade II to III differentiation, with arginase (ARG)-1−, cytokeratin (CK)7+, cluster of differentiation (CD)34+, CK19+, glutamine synthetase (GS)+, Hepa−, alpha-fetoprotein (AFP)−, Ki-67 (40+), hepatocyte nuclear factor (HNF)-1β+, CD56+, and S-100P focal + by immunohistochemical analysis. The patient had a history of type 2 diabetes but no history of cardiovascular disease other than hypertension. Baseline cardiovascular examination, namely myocardial zymography, electrocardiography, echocardiography, and coronary computed tomography angiography (CTA) indicated no abnormalities. He enrolled in a phase II clinical study of teriprizumab combined with gemcitabine and oxaliplatin (GEMOX) as first-line therapy for advanced or unresectable intrahepatic cholangiocarcinoma on 27 October 2021. He was scheduled to undergo GEMOX (gemcitabine 160 mg d1/8; oxaliplatin 120 mg d1/8) combined with teriprizumab (200 mg d1) every 3 weeks. After the first stage of treatment of the first cycle, he dropped out of the clinical study and came to our hospital for the remaining treatment, for which he provided fully informed written consent. The scheduled dose of gemcitabine in the second stage was reduced to 140 mg owing to second-degree myelosuppression; treatment was delayed until 4 November 2021. Routine examination (electrocardiography (ECG); serum myocardial markers) (Figure 1a and Figure 2) indicated normal findings, and the chemotherapy was completed successfully. When the patient was admitted for the second cycle of treatment at 4:30 pm on 23 November 2021, he had no complaints of discomfort. His blood pressure, heart rate, pulse, and oxygen saturation were in the respective normal ranges. Although he was still asymptomatic the next morning, laboratory tests revealed increases in myocardial enzyme levels to varying extents, and the cardiac troponin I (cTnI) level was 14 times higher than the normal value. Creatine kinase-myocardial band (CK-MB), CK, and myoglobin levels were elevated to 47 U/L (normal value: <19 U/L), 938 U/L (normal value: <164 U/L), and 706.1 µg/L (normal value: <70 µg/mL), respectively. Additionally, the cTnI level was markedly elevated (0.87 ng/mL vs normal: <0.06 ng/mL) (Figure 2). Although he presented with a normal (77%) left ventricular ejection fraction (LVEF) on ECG (normal value: 50%–78%), repeat ECG revealed an abnormal change to complete right bundle branch block and left anterior branch block with a heart rate of 95 bpm (Figure 1b). Suspecting immune myocarditis, intravenous methylprednisolone was initiated immediately at 200 mg (4 mg/kg/day). However, at 1:30 am on 24 November 2021 (16 h later), he suddenly experienced palpitations, dizziness, and syncope. His heart rate was 30 to 45 bpm with a sinus arrest on the cardiac monitor. ECG further revealed third-degree atrioventricular block and frequent ventricular premature contractions (Figure 1c and d). He was diagnosed with ICI-related myocarditis and was immediately transferred to the cardiac care unit (CCU) for temporary pacemaker placement and intravenous methylprednisolone administration. After 4 days of methylprednisolone (200 mg; 4 mg/kg/day) therapy, the CK level decreased to 303 U/L, and CK-MB increased slightly to 59 U/L (Figure 1e); however, the low heart rate did not improve. On 29 November 2021, a permanent pacemaker was installed. CK and CK-MB levels decreased to 216 U/L and 55 U/L, respectively, after 160 mg methylprednisolone treatment. On 10 December 2021, the dose of intravenous methylprednisolone was reduced to 80 mg (1.6 mg/kg), and CK, CK-MB, and cTnI levels decreased to 190 U/L, 30 U/L, and 1.6 µg/L, respectively (Figure 2). The patient was discharged from the hospital and was maintained on a tapering regimen of oral methylprednisolone for 2 weeks (Figure 3). He permanently discontinued immunotherapy and refused to undergo further examination or any anti-tumor treatment. The patient was still alive and able to perform light physical activity with no discomfort, with an Eastern Cooperative Oncology Group (ECOG) score of 1, at the last follow-up on 11 September 2022.

ECG findings.
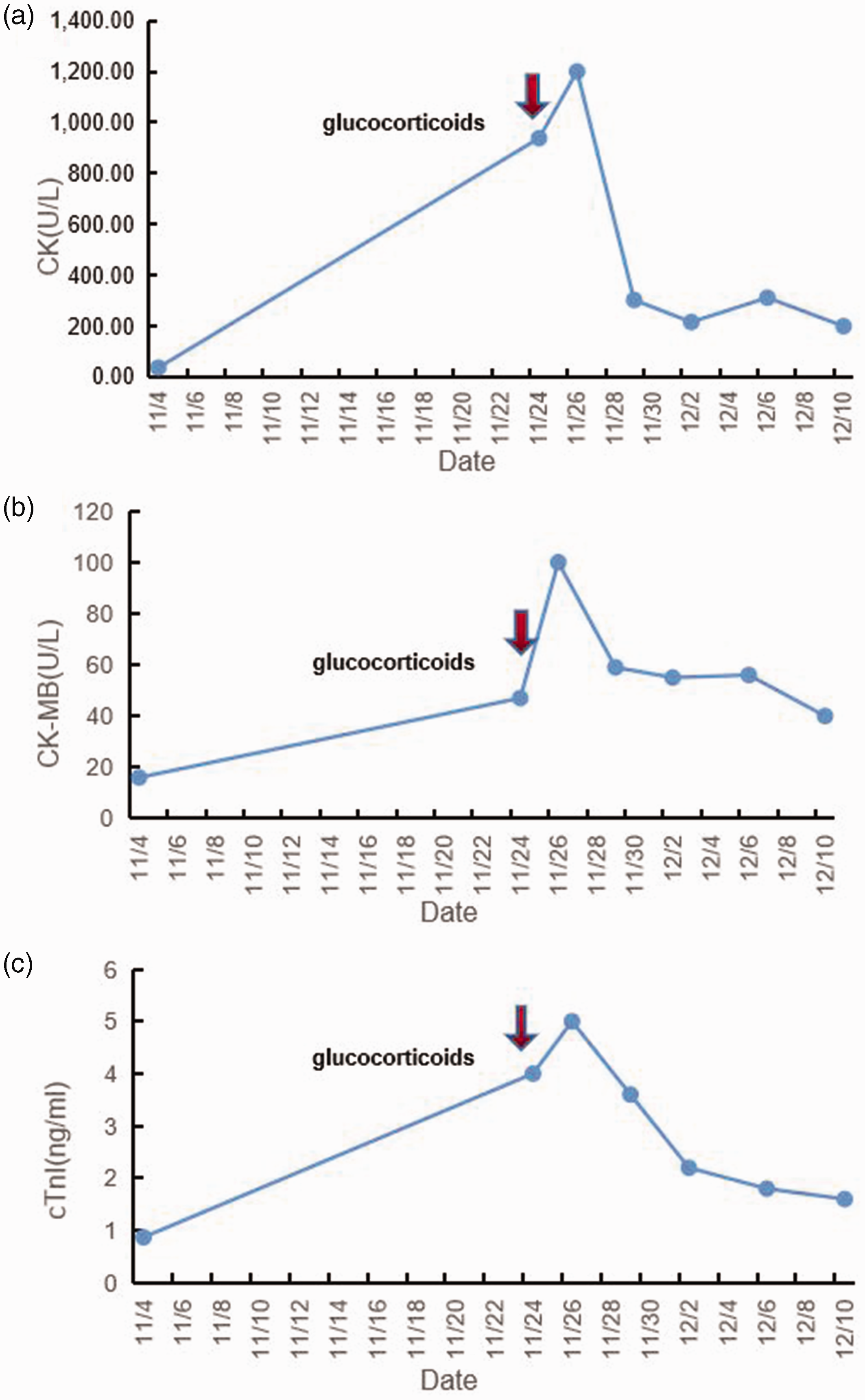
Changes in serum myocardial biomarkers before and after glucocorticoid therapy with methylprednisolone 4 mg/kg/day, intravenously, which was decreased to 1.6 mg/kg after 17 days and tapered after another 14 days. (a) CK changes over time; (b) CK-MB changes over time and (c) cTnI changes over time.
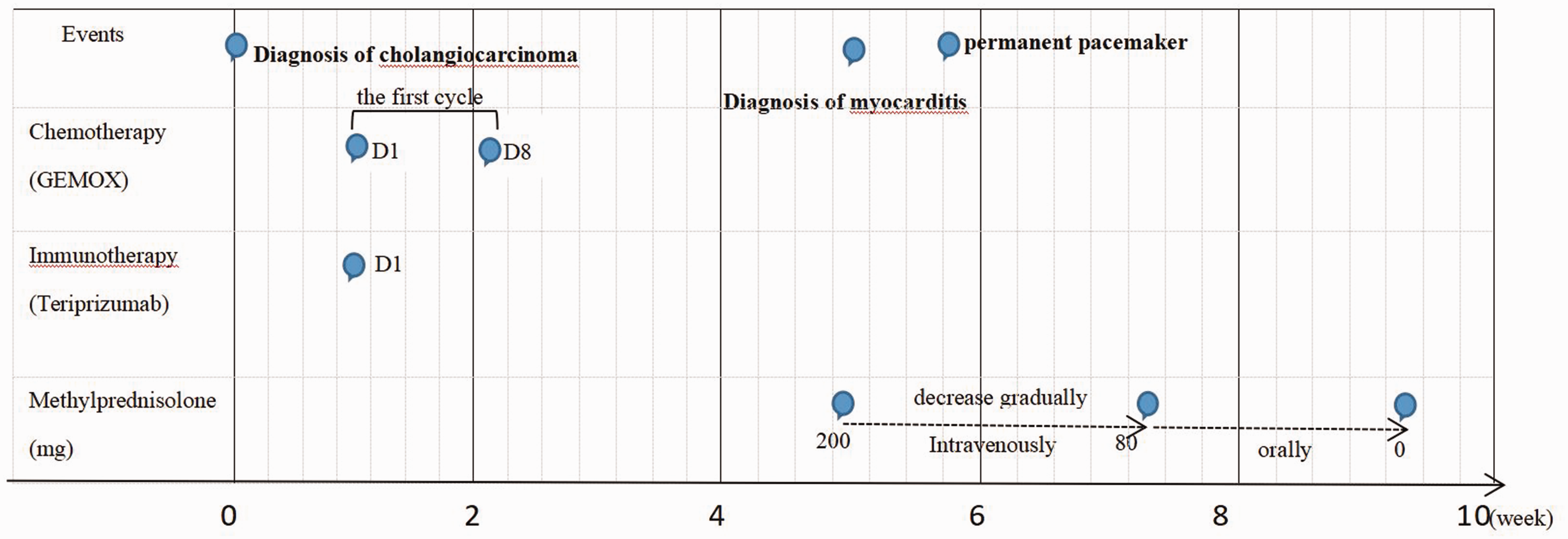
Summary of the clinical course of the case.
Discussion
With the widespread application of immunotherapies, immune-related adverse events (irAEs) are increasingly attracting attention. Although any organ or system can be involved, irAEs most commonly affect the gastrointestinal tract, endocrine glands, skin, and liver. 5 ICI-related cardiovascular toxicities often occur as myocarditis, pericardial disease, or vasculitis, and patients less commonly present with Takotsubo-like syndrome and other non-inflammatory left ventricular dysfunctions. These presentations almost never overlap. 6 The mechanism of ICI-related myocarditis is unclear. Off-target attack of immunotherapy is suggested to originate from the presence of common epitopes shared by tumor cells and immune cells. 7
ICI-associated myocarditis usually occurs early, with a median time of 17 to 34 days after onset of ICI therapy, approximately after the first or second ICI infusion. 1 Our patient was diagnosed with immune myocarditis 29 days after the initial treatment with teriprizumab, which was consistent with this timeframe. However, there are also reports of the occurrence of immune myocarditis after 28 cycles of immunotherapy or even after the cession of immunotherapy. 1 Therefore, the possibility of immune myocarditis should not be excluded at any time when immunotherapy has been used. The prevalence of myocarditis varies with the type of ICI, in the decreasing order of anti-cytotoxic T-lymphocyte antigen-4 (anti-CTLA-4) monotherapy (3.3%), and anti-PD-ligand 1 (anti-PD-L1) (2.4%) and anti-PD-1 agents (0.5%). 8
The risk factors for myocarditis due to ICI therapy are not fully clarified; however, combination immunotherapy is considered the predominant risk factor. Additionally, many chemotherapies or targeted drugs may cause drug-related myocarditis. especially in patients who received ICIs combined with chemotherapies, targeted therapies, or other non-ICIs or who received non-ICIs previously. This issue makes determining the exact cause of cardiac toxicity very challenging. Moreover, combination therapy exacerbates cardiotoxicity and increases treatment difficulty. Chemotherapy drugs that can cause cardiotoxicity mainly comprise anthracyclines, antimicrotubule molecules, and 5-fluorouracil. 9 Rhythm and conduction disturbances are often caused by antimicrotubule molecules, such as vinca alkaloids or taxanes, whereas cardiotoxicity caused by gemcitabine and oxaliplatin is reported rarely in the literature. In our case, the patient experienced severe conduction disturbances after only one cycle of an ICI and GEMOX. The fulminant course and the specific types of chemotherapeutic drugs used in this case indicated a relationship between myocarditis and ICIs. A study using a mouse model found that cardiac irradiation increased the risk of and morbidity associated with cardiac toxicity from immunotherapy alone. Some researchers suspected that cardiomyocyte injury from non-immune therapy exposed cardiac antigens and, combined with immunotherapy, activated T cells, triggering or exacerbating cardiomyocyte immune damage. 10 Additional possible risk factors are pre-existing cardiac disease, diabetes mellitus, obesity, and autoimmune disease. 11 Our patient had no history of heart disease or autoimmune disease but did have a history of type 2 diabetes for more than 5 years. Therefore, patients must be fully evaluated at baseline, and patients with high-risk factors must be closely monitored after immunotherapy initiation, especially in the early stages.
The presentation of immune myocarditis is non-specific and varies, but the condition shows a fulminant course. The symptoms are those of acute heart failure and arrhythmias, varying from shortness of breath, chest pain, and syncope to cardiac shock. 12 In our case, the patient was asymptomatic at first but showed abnormal laboratory findings. Just 16 hours later, his symptoms developed rapidly to palpitations, dizziness, and syncope. Various arrhythmias have been described in patients receiving ICI therapy, namely conduction delays, ventricular arrhythmias, and atrial fibrillation. Notably, arrhythmic complications have been implicated as a major driver of mortality and sudden cardiac death. 11
The diagnosis of myocarditis can be very challenging and requires a very high index of clinical suspicion. All of our cases with prior ICI use had elevated troponin and abnormal ECGs at the time of clinical presentation. Mahmood et al. noted that nearly all cases of myocarditis had increased troponin levels (94%) and abnormal ECGs (89%) at the time of presentation. 1 A combination of clinical symptoms, biochemistry and imaging can be used to diagnose ICI-associated myocarditis. With its 94% to 100% sensitivity, cardiac troponin appears to be one of the most important markers for ICI-induced myocarditis, not only in the diagnosis but also in predicting prognosis. 13 Our patient’s condition was initially notable for the obvious elevation of cTnI. In ICI-associated myocarditis, CK-MB levels almost always increase to varying degrees, and the elevation is not limited to myocarditis but also occurs with skeletal muscle inflammation. Clinically, many patients treated with ICIs show varying degrees of CK-MB elevation; however, very few patients develop immune myocarditis, which needs to be excluded as a diagnosis by combining troponin and ECG data. ECG is a quick and simple test, and is the first option when patients present with cardiovascular symptoms. In our case, abnormal ECG changes were observed even when the patient was asymptomatic, and significant abnormalities were observed in the ECG when the patient showed obvious heart symptoms. Thus, dynamic ECG examination is necessary for patients with suspected immune myocarditis. Cardiac ECG is also preferred for the first-line diagnosis of immune myocarditis; however, compared with cardiac markers and ECG, its sensitivity is relatively poor. Even a normal LVEF does not exclude immune myocarditis. 14 The gold standard for the diagnosis of myocarditis is endomyocardial biopsy. 15 However, this method is not recommended as a first-line test because it is invasive, is associated with the lethal risk of cardiac perforation, and has sampling limitations, which could result in false-negative outcomes. 16
When a patient is diagnosed with suspected immune myocarditis, ICI treatment should be discontinued, and corticosteroids should be administered immediately. In a retrospective study by Wang et al., 17 a patient developed marked elevations of myocardial enzymes on zymography only 12 days after the first nivolumab infusion, with no obvious symptoms. The patient did not receive any treatment, including corticosteroids. The patient was then admitted to the hospital with palpitations and chest tightness only 16 days later; she subsequently developed third-degree atrioventricular block and died soon after. 17 For patients with severe ICI-related myocarditis, high-dose glucocorticoids and even immunosuppressants are recommended. An awareness of immune myocarditis and early corticosteroid intervention are crucial for improving the prognosis and even saving a patient’s life. High-dose corticosteroids (i.e., methylprednisolone 1000 mg/day for 3 days followed by prednisone 1 mg/kg) is recommended as the first-line therapy in the acute phase. 18 However, the use of corticosteroids, especially in high doses, is a double-edged sword that could aggravate some underlying diseases or result in new diseases.1 In our patient, considering the diabetes history, we initially chose 200 mg/day methylprednisolone (4 mg/kg/day), and the patient developed a noticeable increase in blood glucose during treatment. For patients with underlying diseases, choosing the dose of corticosteroids to maximize the benefits while minimizing the side effects is a challenge. ICI-related myocarditis is a rare but fatal disease. The key in cases of suspected immune myocarditis is to raise awareness of the condition and initiate corticosteroid therapy immediately to alleviate symptoms and decrease the risk of death.
Footnotes
Acknowledgements
The authors are indebted to the support provided by the physicians and nursing staff of the Oncology Department, Taizhou People’s Hospital.
Author contributions
Yunyao Ye contributed to the data analysis, witting the article, and editing the figures. Yiping Li contributed to witting the article and editing the figures. Sihui Zhang contributed to the literature search and clinical record collection. Gaohua Han contributed to the conception and revision of the article.
Data availability statement
The original data evaluated in this study are available from the corresponding author upon request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
All patient-specific details have been deleted from this article. Signed consent for publication was obtained from the patient and his son, and this case report was approved by the Ethics Committee of Taizhou People’s Hospital.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Jiangsu Provincial Six Talent Peaks [grant number WSW-264] and the “311 Project of Taizhou” [grant number RCPY201813].
