Abstract
Objective
To evaluate the effect of trigeminal nerve block (TNB) on patients’ quality of life (QOL) 15 days after the procedure in patients with refractory TN.
Methods
This retrospective observational cohort study involved patients receiving TNB (levobupivacaine, clonidine, corticosteroid) between 2014 and 2018 at a postoperative pain clinic in France. Change in QOL from Day 0 (before block) to Day 15 was assessed according to SF-12.
Results
21 patients (62 ± 14 y) were included in the study. Most patients (71%) were referred following surgery or dentistry. Of the 9 patients (43%) who exhibited >10% increase in SF-12 scores and so were deemed responders, SF12-physical and SF12-mental were increased by mean differences of 17 and 9 points, respectively. The mean duration of block lasted 15 ± 59 days (range 1 to 90 days) and no severe adverse effects were observed.
Conclusion
Improved QOL was observed in approximately 50% of patients with trigeminal neuralgia (TN) two weeks after specific nerve block. The technique was easy to administer and well accepted by the patients.
Keywords
Introduction
Trigeminal neuralgia (TN) is a clinical entity involving many aetiologies and causes a neuropathic pain that is sometimes refractory to all medical treatments. 1 The annual overall incidence of TN ranges from 4.3 to 27/100,000 people/year, and the incidence increases with advancing age. 1 First-line treatment for the management of TN is anticonvulsant medication (e.g., carbamazepine or oxcarbazepine) but approximately 25% of patients are refractory to carbamazepine, 2 and require additional treatment. Despite limited scientific evidence, alternative therapies such as peripheral trigeminal nerve block (TNB) are often used.1,3,4 The efficacy of TNB on chronic pain is controversial but when effective its beneficial effects have been reported to be more prolonged than pharmacological effects. 5 Indeed, the beneficial effect of TNB may last from several days to weeks.6–8 Guidelines recommend that improvement in functional status and quality of life (QOL) should be the main criteria to evaluate the usefulness and efficacy of loco-regional anaesthesia in treating chronic pain. 9 Therefore the primary objective of the present study was to evaluate the effect of TNB on change in QOL, 15 days after the procedure, in patients with refractory TN. Secondary objectives were to evaluate pain levels, analgesic consumption, duration of block, and patient satisfaction over the same 15-day period.
Patients and Methods
This retrospective observational cohort study was performed between July 2018 and March 2019. Eligible patients were those with refractory TN who received a nerve block between 2014 and 2018 in a postoperative pain clinic at Limoges University Hospital. Patients who did not provide informed consent, had received a different type of block during the previous year, had incomplete medical records or had died since the TNB, were excluded from the study.
During their first consultation, patients had undergone a complete neurologic examination. Topographic information on their facial pain was recorded. After patient consent, TNBs were performed in conscious patients at the level of emergence of trigeminal nerve in the supra/infra zygomatic fossa or the supra orbital fossa. 10 Following a negative aspiration test, patients received 5 ml levobupivacaine 0.5%, 1ml corticosteroid (i.e., cortivazol 3.75 mg/ml or betamethasone 7 mg/ml) and clonidine 1 μg . kg−1 injected near the nerve trunk. Patients were instructed to contact the clinic to arrange a new TNB if their pain reappeared; the time difference between the two blocks was defined as the duration of analgesia. Pain was evaluated before each TNB using a visual analogue scale (VAS) score that ranged from 0–10. In addition, patients completed the Douleur Neuropathique 4 (DN4) questionnaire (scored 0–10) before the block and again in the recovery room.
Follow-up telephone calls were made to the patients 15 days after their TNB procedure. QOL was evaluated using the SF-12 questionnaire on Day 0 (before block) and Day 15. Non-responders were defined as those patients with <10% variation in SF-12 scores between the two timepoints. Patient satisfaction, evaluated on a simple verbal scale that ranged from 0 (bad) to 10 (excellent), adverse effects and patients’ assessment of medication consumption were evaluated on Day 15.
The reporting of this study conforms to STROBE guidelines. 11 All participants provided written informed consent. Patient data were anonymized prior to analysis. The study was approved by the hospital ethics committee (CPP Sud Méditerranée, Montpellier, France, 11 September 2018, N° ID-RCB 2018-A01979-46) and it was also registered at ClinicalTrials.gov (NCT03669744).
Statistical analyses
Statistical analyses were performed using XLSTAT software. Data obtained on Day 0 and Day 15 were compared using paired t-tests. Linear regression analysis was used to analyse any link between SF-12 scores and other quantitative data. A P-value <0.05 was considered to indicate statistical significance.
Results
In total, 25 patients received TNB at our centre between 2014 and 2018. Of these, four patients were excluded from the analysis (i.e., lost to follow-up; other block; did not wish to participate; death). Demographic and clinical data of the 21 eligible patients are shown in Table 1. Most patients were older than 50 years (mean ± SD age was 62 ± 14 years) and with the exception of two patients, all patients had at least one concomitant disease. Most common aetiologies were post-surgical (8 patients, 38%) and post dental care (7 patients, 33%) and the average duration of symptoms before the first block was 4 ± 5 years. On average, TNB had been performed at least four times in each patient.
General characteristics of patient population.

Values are shown as n, n (%) or mean ± SD.
*Some patients had more than one concomitant disease.
∞Some patients had more than one aetiology.
αNerve blocks were performed in the supra orbital foramen (V1), supra zygomatic fossa (V2) or in the infra zygomatic fossa (V3).
Abbreviations: ENT, ear, nose and throat; F, female; M, male.
Before receiving the TNB, SF-12 physical (SF12-PS) and mental (SF-12 MS) scores were 35 ± 14 and 29 ± 11, respectively. At Day 15, mean SF-12 PS and mean SF-12 MS values had increased by 3 and 5 points, respectively. In total, 11 patients (52%) exhibited <10% increase in SF-12 PS and 12 patients (57%) <10% increase in SF-12 MS (Figure 1). Following exclusion of the non-responders, SF-12 PS increased by 16.6 points (P < 0.01), and SF-12 MS by 9.2 points (P = 0.01). Combining, SF-12-PS and SF-12 MS scores, 12 (57%) patients were deemed non responders and nine (43%) were responders.

Individual SF-12 physical (SF-12PS) and mental (SF-12 MS) status scores. Patients with less than 10% improvement are shown by black solid lines. For SF-12PS there were 11 (52%) non-responders (two patients had same value). For SF-12MS, there were 12 (57%) non-responders. Following exclusion of the non-responders, SF-12 PS increased by 16.6 points (P < 0.01), and SF-12 MS by 9.2 points (P = 0.01).
No correlations were observed between the SF-12 scores and sex, age, time from first symptoms to first block or location of pain. Although there was no correlation between SF-12 MS scores and patient satisfaction, a significant correlation was observed between SF-12 PS scores and patient satisfaction (r2 = 0.30, P = 0.01).
Mean VAS pain scores were reduced by 3 points over the 15-day period (P < 0.001). DN4 scores also decreased significantly following the block (P < 0.001). The mean duration of the block was 15 ± 59 days (range 1 to 90 days). Unsurprisingly, patient satisfaction scores correlated with duration of the block (r2 = 0.51; P < 0.01). All nine (43%) responders had a satisfaction score ≥7. All 12 (57%) non-responders had a satisfaction score ≤5.
Patients reported that their pharmacological treatments were reduced over the 15 days (Figure 2). Six transitory adverse effects were observed (i.e., three cases of pain during injection; one case each of diplopia, edema at the puncture site, and local dysesthesia at the block area).
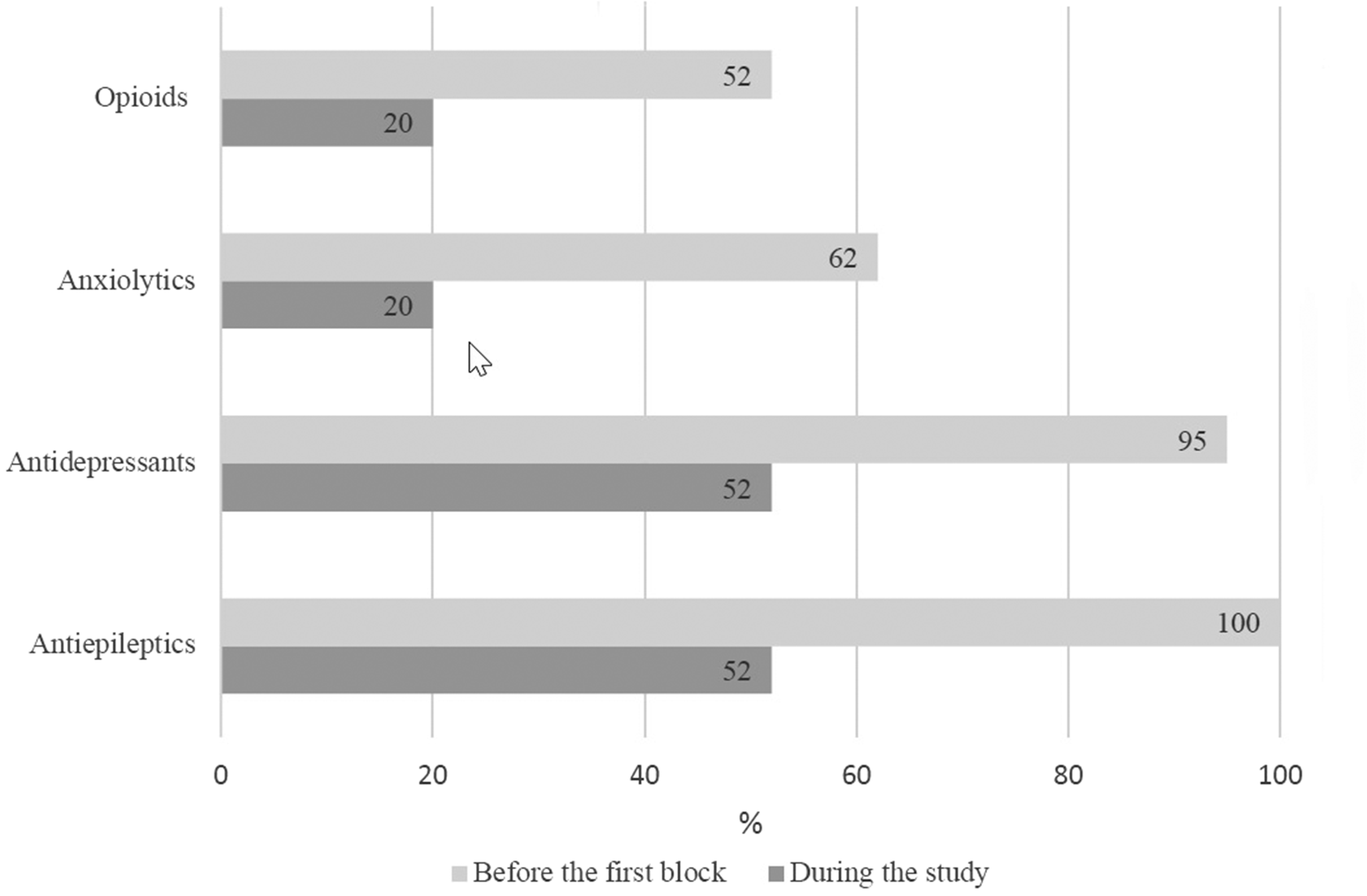
Patient reported pharmacological treatments over the 15-day study period. All treatments were reduced following the block (x axis is % patients, some patients received several medications).
Discussion
Although approximately half of patients with refractory TN did not show an improvement in QOL as assessed by SF-12 survey, the remaining half showed a significant improvement in QOL that correlated with patient satisfaction. Pain scores were reduced over the 15-day period as was use of background pain therapies. Moreover, no major adverse effects were observed.
We searched the PubMed database for similar studies using “trigeminal nerve block,” OR “peripheral nerve block,” AND “trigeminal neuralgia.” as keywords/terms. Our literature search identified eight articles from 2013 to 2019; three were isolated clinical cases and five were small cohort studies (Table 2).7,8,12–17 The selected patients and type of blocks used in these studies were heterogeneous and so it is difficult to compare their results with ours. The choice and dose of drugs used in these studies were probably based on routine clinical practice. The most frequently used treatment was a combination of local anaesthetic and a long-lasting corticosteroid (usually dexamethasone). Interestingly, in several studies, the use of short acting lidocaine resulted in blocks that lasted longer far longer than we achieved in this present study with a long-acting local anaesthetic (i.e., levobupivacaine).15,17 The combination treatment used in this present study included three drugs, a long-lasting local anaesthetic (0.5% levobupivacaine), a long-lasting corticosteroid and clonidine. Clonidine is widely used with loco-regional anaesthesia to increase and deepen analgesia in acute pain management 18 and prevent neuropathic pain. 19
Summary of studies reporting trigeminal nerve block in refractory trigeminal neuralgia 2013–2019.

NA, not assessed; QOL, quality of life; RCT, randomized controlled trial; SF-36: Short Form 36; TNB, trigeminal neuralgia block.
We found only one other study that had assessed QOL after nerve block for refractory TN. 12 The randomised study had been performed in 13 patients with TN who were receiving pharmacotherapy and six patients of these patients were randomized to also receive a lidocaine block. Using the SF-36 QOL questionnaires, the investigators found that patients on the combination therapy had improved QOL at follow-up visits up to 90 days. These findings are consistent with our results. We chose the SF-12 questionnaire because of its ease, rapidity and feasibility for a telephone interview. On Day 15, in our responder group, we observed score improvements of 17 for SF-12 PS and 9 for SF-12 MS. Opinions of clinically appropriate SF-12 score improvements are heterogeneous across the literature. For example, in patients with lumbago, 3- and 4-point variations in SF-12 PS were considered clinically pertinent but lower difference were observed in patients with chronic pain and a better baseline QOL. 20 By contrast, in another study, 9- and 14-point variations in SF-12 PS and MS were the threshold for clinically pertinent values following knee surgery. 21 In this present study, we decided that percentage variations were more relevant than variations expressed as absolute values. Indeed, the good correlation between 10% improvement in SF-12 PS and patient satisfaction suggests that this 10% threshold was clinically pertinent.
Other therapeutic options are available for refractory TN including microvascular decompression and thermocoagulation22,23 However, they require general anaesthesia and sometimes repeated procedures to obtain optimal efficacy. In addition, reports suggest that 30–45% of patients must continue medical treatment after surgery. 24 Furthermore, prolonged adverse effects such as 15 to 50% sensory impairment have been observed 24 and, although rare, mortality has been reported following microvascular decompression. 23 The advantage of TNB is that it can be performed in an ambulatory setting, thereby avoiding general anaesthesia and related complications.
The study had several limitations. For instance, our sample size was small which limited the study’s statistical power. In addition, it was retrospective and uncontrolled. Furthermore, most patients (71%) were referred following surgery or dentistry and so post-traumatic trigeminal pain was over-represented in the group. Importantly, bias may have been introduced because 57% of patents were excluded from the analysis because they were deemed ‘non-responders.’ Nevertheless, this retrospective study observed improved QOL in approximal 50% patients with refractory TN, 15 days after specific nerve block. The technique is simple, well tolerated and well accepted by the patients. Further randomized controlled trials are needed to confirm these results, determine why some patients are unresponsive to the technique and identify the best combination of drugs to use.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221132027 - Supplemental material for Quality of life after trigeminal nerve block in refractory trigeminal neuralgia: A retrospective cohort study and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605221132027 for Quality of life after trigeminal nerve block in refractory trigeminal neuralgia: A retrospective cohort study and literature review by Nicolas Jacques, Simon Karoutsos, Loïc Marais and Nathalie Nathan-Denizot in Journal of International Medical Research
Footnotes
Acknowledgements
The authors wish to thank Arthur Baïsse for his critical review, Chantal Rougier and Laurent Nougier for their availability and Dr Jeanne Cook-Moreau for translation assistance.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
