Abstract
Objective
To investigate the effect of drainage on heterotopic ossification (HO) after total hip arthroplasty (THA) and to evaluate other postoperative complications and joint dysfunction between patients with and without drainage.
Methods
In this retrospective cohort study, the medical records of patients who underwent THA from 2017 to 2019 were reviewed. The patients were divided into a drainage group and non-drainage group. Standard preoperative anteroposterior and lateral radiographs were assessed by senior radiologists for HO analysis. Clinical indicators included the hemoglobin concentration, superficial infection, blood transfusion, hematoma formation, hip range of motion (ROM), erythrocyte sedimentation rate, C-reactive protein concentration, dressing changes, visual analogue scale score, and Harris Hip Score (HHS).
Results
The incidence of HO was significantly higher in the drainage than non-drainage group (32.0% vs. 16.3%). The presence of severe HO (Brooker grade III or IV) was also different between the groups. Patients in the non-drainage group had smaller ROM early after surgery, but the final ROM and HHS did not differ significantly between the groups.
Conclusions
The rate and degree of HO after THA were significantly different between patients with and without drainage. There is no added advantage of closed suction drainage over no drainage in primary THA.
Keywords
Introduction
Heterotopic ossification (HO) is the most frequent complication following primary total hip arthroplasty (THA), occurring at a mean incidence of 24% to 32%.1,2 Scarce evidence exists regarding the influence of drainage on the occurrence of HO. Generally, HO is asymptomatic. In severe cases, however, local pain and limitation of joint excursion may occur. 3 A postulated etiology is that osteoinductive growth factors are released as a consequence of soft tissue trauma, thus inducing the formation of HO; however, the exact mechanism remains unclear. 4
As a routine step in THA, drainage is frequently used before closure of the joint capsule. This has historically been believed to be able to prevent hematoma formation, significantly decrease tension over the incision, reduce the delay in wound healing, and reduce the risk of infection. 5 However, recent studies have indicated that drainage can cause other problems and that it does not reduce hematoma formation or the incidence of wound-related complications. 6 Furthermore, some studies have shown that drainage increases blood loss and the risk of retrograde infection after THA. 7
The primary purpose of the present study was to investigate the influence of drainage on HO. We comprehensively collected radiological data, clinical data, and laboratory test results of patients undergoing THA. The differences between patients with and without drainage were assessed to provide help in the decision-making of treatment strategies.
Patients and Methods
Inclusion and exclusion criteria
The inclusion criterion for this study was treatment by primary unilateral THA for osteoarthritis or osteonecrosis of the femoral head. The exclusion criteria were previous surgery or post-traumatic disorders in the same limb, hip arthrodesis, blood coagulation disorders, intraoperative fractures, and loss to follow-up with the inability to be contacted. Based on the inclusion and exclusion criteria, 198 patients who underwent primary unilateral THA at our hospital from 2017 to 2019 were retrospectively screened and included in this retrospective cohort study.
Demographic and general information
The patients’ demographic and general information was obtained from their medical records, including age, sex, body mass index, hospitalization time, side of surgery, duration of the operation, and intraoperative blood loss. The study was approved by the Institutional Review Board of the Third Hospital of Hebei Medical University (approval number: K2020-004-1, date of approval: 13 January 2020) and was conducted in accordance with the Declaration of Helsinki and the regulations of the Health Insurance Portability and Accountability Act (HIPAA). Because this was a retrospective study and all patient information was de-identified before the analysis, informed consent was only required for patients whose radiological images would be published; consequently, these patients provided written informed consent. The reporting of this study conforms to the STROBE guidelines. 8
Clinical evaluations
To evaluate the onset of HO and compare its incidence between patients with and without drainage, radiological images were collected preoperatively and 1 week, 3 months, 6 months, and 12 months postoperatively and compared using a radiological display monitor (Figure 1). HO was classified according to the Brooker classification. Brooker grades I and II were considered radiologically mild HO, and Brooker grades III and IV were considered radiologically severe HO. Clinical data included the performance of blood transfusion, the incidence of superficial infection, and the frequency of dressing changes during hospitalization. The hemoglobin (Hb) and C-reactive protein (CRP) concentrations were collected preoperatively and 1 and 5 days postoperatively. The erythrocyte sedimentation rate (ESR) was collected preoperatively and 1 day postoperatively. Prior to removal of the drains in the drainage group and 24 hours after surgery in the non-drainage group, the patients’ pain symptoms were assessed using a visual analogue scale (VAS). The wound hematoma thickness was assessed by ultrasonography on postoperative day 3. Range of motion (ROM) of the hip was assessed preoperatively and at discharge. Functional results were evaluated using the Harris Hip Score (HHS) preoperatively and 1 year postoperatively.

Images of a 30-year-old man with osteonecrosis of the femoral head in the left hip. The patient had undergone total hip arthroplasty 2 years previously. (a) The radiograph showed no significant abnormalities around the prosthesis at discharge and (b) The radiograph showed severe heterotopic ossification around the prosthesis 2 years after surgery.
Operative technique
All patients underwent the operations by the same operation team. All procedures were performed under general or spinal anesthesia using a biologic total ceramic total hip prosthesis (Tri-Lock COC; DePuy Synthes, Raynham, MA, USA), and the patients were placed in a lateral position using a standard posterolateral approach and minimally invasive techniques. In the drainage group, a silicone tube was inserted and the drain was removed 24 hours postoperatively. In the non-drainage group, direct closure was performed. Tranexamic acid (1 g) was intravenously administered immediately prior to the incision, and a second dose was applied topically immediately prior to closure. All wounds were covered with sterile dressings and pressure bandaged. Low-molecular-weight heparin was administered subcutaneously 12 hours after the operation and was continued until discharge for thrombosis prophylaxis. All patients were asked to perform ankle flexion and extension exercises in bed immediately after surgery. On the second day after surgery, the patients were asked to perform progressive ambulation exercises with partial weight bearing with the assistance of a walker or crutches. Transfusion therapy was initiated when the Hb concentration reached <8 g/dL (or <10 g/dL with clinical signs of hypovolemia).
Statistical analysis
Excel 2016 for Windows (Microsoft Corporation, Seattle, WA, USA) and SPSS 26.0 statistical software for Windows (IBM Corp., Armonk, NY, USA) were used for the statistical analysis. Categorical variables are expressed as frequency. Continuous variables are expressed as mean ± standard deviation. Student’s t test and the chi-square test were used. A P value of <0.05 was considered statistically significant.
Results
Patients’ characteristics
Among all 198 hips, 100 patients were included in the drainage group and 98 in the non-drainage group. The patients’ baseline characteristics are summarized in Table 1. None of the demographic and baseline characteristics differed between the two groups, including age, sex, body mass index, hospitalization time, side of surgery, duration of the operation, and intraoperative blood loss.
Characteristics of patients in drainage and non-drainage groups.
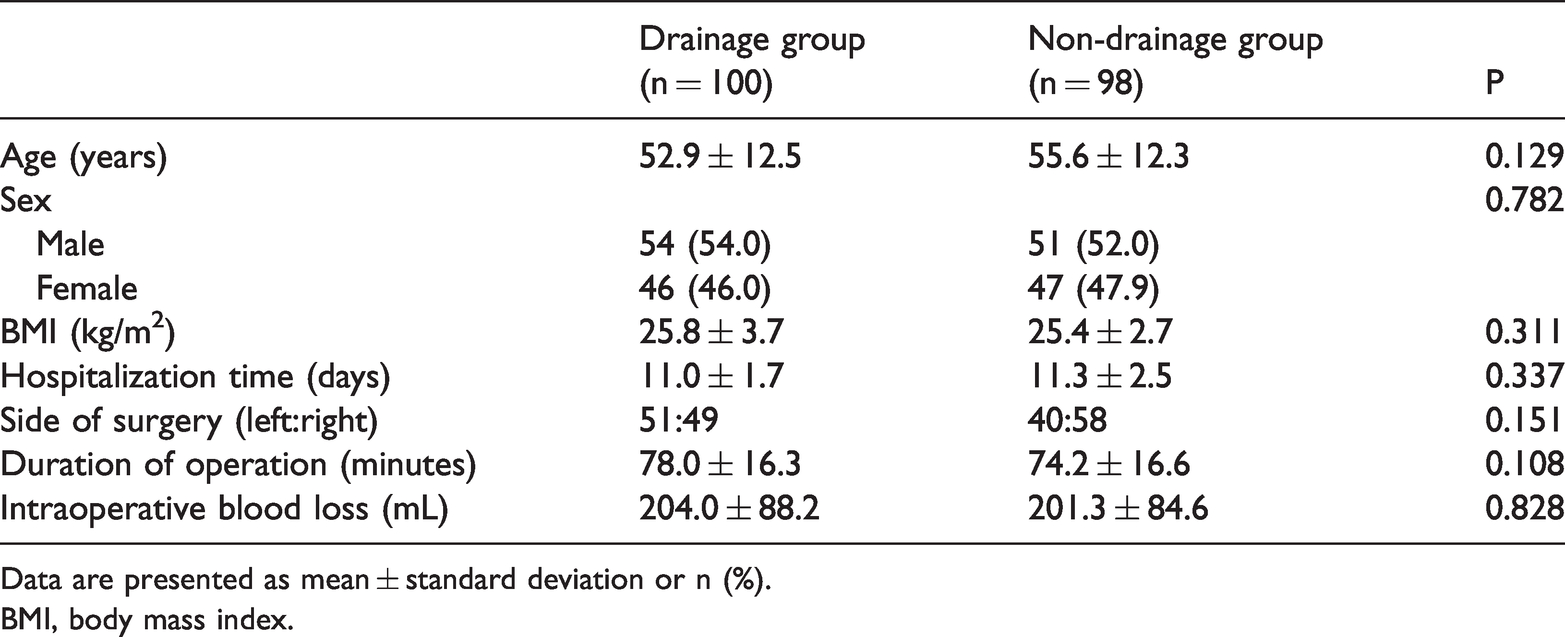
Data are presented as mean ± standard deviation or n (%).
BMI, body mass index.
Effect of drainage and no drainage on various clinical parameters after THA
Of the 198 patients included in our analysis, 48 (24.2%) developed HO (Table 2). Among the 24.2% patients who developed HO, 11.1% had Brooker grade I HO, 9.6% had grade II, and 3.5% had grade III. The incidence of HO was significantly higher in the drainage than non-drainage group (32% vs. 16.3%, P = 0.010). When considering Brooker grade III and IV as radiologically severe, five (15.6%) patients in the drainage group and two (12.5%) in the non-drainage group had HO after 12 months. The majority of the 48 patients with HO had evidence of it on radiographs by 3 months postoperatively (Table 3).
Comparison of variables between drainage and non-drainage groups.
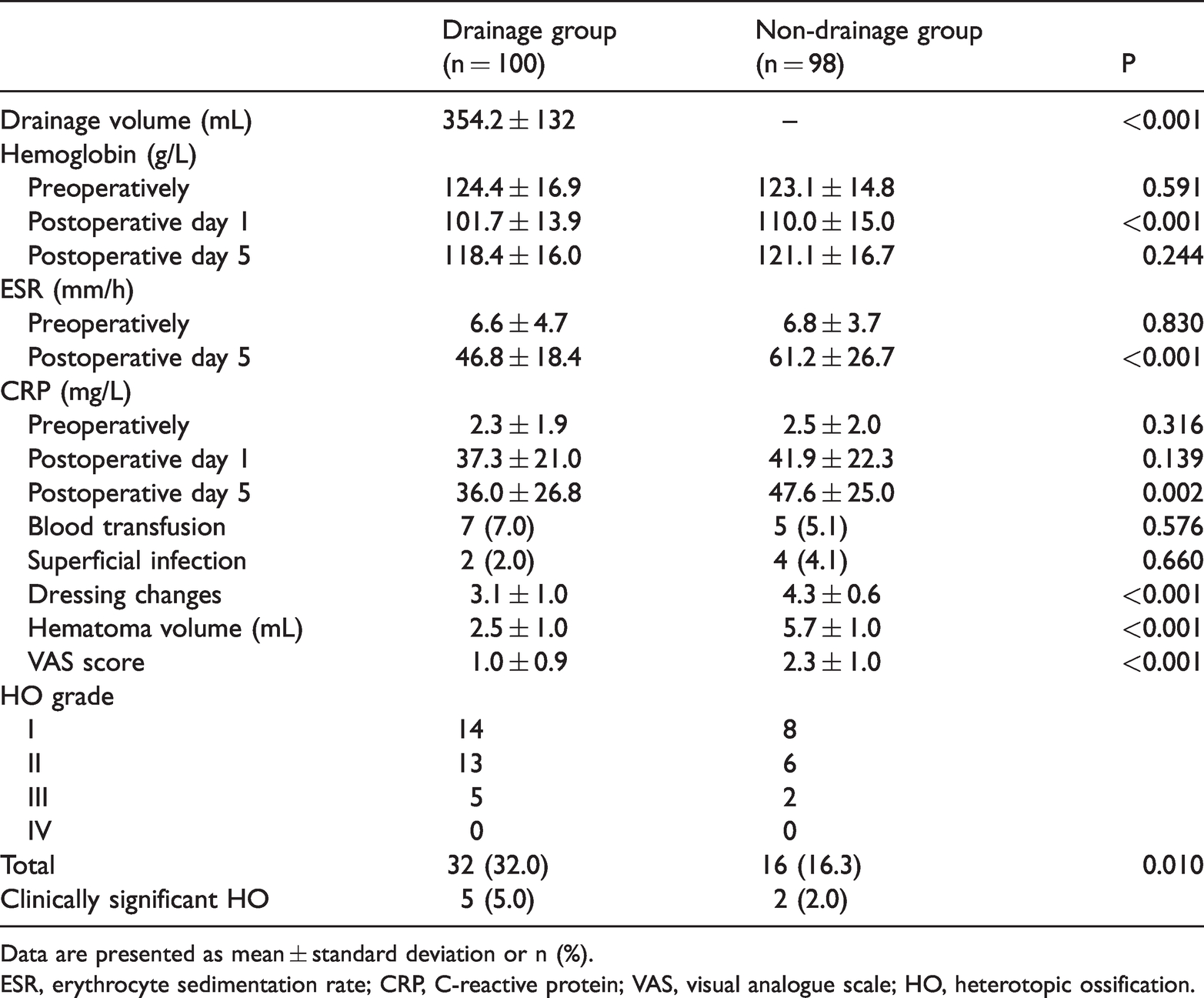
Data are presented as mean ± standard deviation or n (%).
ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; VAS, visual analogue scale; HO, heterotopic ossification.
Timing of HO diagnosis after THA by Brooker classification.

Data are presented as n (%).
HO, heterotopic ossification; THA, total hip arthroplasty.
None of the clinical parameters after THA differed between the drainage and non-drainage groups, including the Hb concentration on postoperative day 5 (118.4 ± 16.0 vs. 121.1 ± 16.7 g/L, respectively), transfusion rate (7.0% vs. 5.1%, respectively), and rate of superficial infection (2.0% vs. 4.1%, respectively). However, significant differences were found between the two groups in the ESR on postoperative day 5 (46.8 ± 18.4 vs. 61.2 ± 26.7 mm/h, respectively; P < 0.001), the CRP concentration of postoperative day 5 (36.0 ± 26.8 vs. 47.6 ± 25.0 mg/L, respectively; P = 0.002), dressing changes (3.1 ± 1.0 vs. 4.3 ± 0.6, respectively; P < 0.001), hematoma volume (5.7 ± 1.0 vs. 2.5 ± 1.0 mL, respectively; P < 0.001), and immediate postoperative pain level (VAS score) (1.0 ± 0.9 vs. 2.3 ± 1.0 points, respectively; P < 0.001). Longer follow-up revealed no difference in ROM between the two groups (Table 4). At the 1-year follow-up, no significant difference was found in the HHS between the two groups (83.0 ± 4.5 vs. 83.3 ± 5.1).
Comparison of ROM and HHS between drainage and non-drainage groups.

Data are presented as mean ± standard deviation.
ROM, range of motion; HHS, Harris Hip Score.
Discussion
This is one of the few studies comparing the effects of drainage and no drainage on the development of HO. The overall incidence of HO in this study was 24.2%, which is a moderate level compared with some similar studies.9–11 For instance, Toom et al. 12 reported an HO incidence of 32% among 178 patients who underwent THA. Comeau-Gauthier et al. 13 noted an incidence of 19.9% among 1441 patients who underwent THA. The incidence of severe HO (grade III or IV) was 3.5% (7 of 198) in the present study, which is lower than the previously reported prevalence of 3.6% to 7.0% for grades III and IV HO after THA or HA.14,15
The established risk factors for HO are male sex, hypertrophic arthrosis, obesity, ankylosing spondylitis, rheumatoid arthritis, the surgical approach used, post-traumatic arthritis, Paget’s disease, and idiopathic skeletal hyperostosis.16–18 Other risk factors, although not yet studied in depth, may be related to surgery, such as the use of drainage. In our study, the incidence of HO was significantly higher in the drainage than non-drainage group. Cohn et al. 19 reported that the development of a hematoma under the fascia was considered as a factor affecting HO. Hematoma formation induces the release of bone-inductive factors, thereby affecting the formation of HO. 20 Based on this pathogenesis, the use of drainage after THA is an important measure. However, some studies have shown that drainage is a contributing factor to the development of HO after THA. 21 The use of drainage is an additional source of trauma to the soft tissues. 22 Nauth et al. 23 considered that the trauma to the surrounding soft tissues creates an environment favorable to local proliferation of fibroblasts and accumulation of extracellular matrix, which will evolve into HO through endochondral ossification. Huang et al. 24 considered that when tissue is damaged, a local hypoxic microenvironment forms. Under hypoxic stimulation, hypoxia-inducible factor is activated and regulates the expression of cytokines, promoting the differentiation of ectopic bone precursor cells and mesenchymal cells into cartilage and osteogenic cells, which ultimately develop into ectopic bone.25,26 The presence of drainage also provides another route for the spread of osteogenic mediators through soft tissues. 22 Furthermore, the presence of a foreign body such as a drain can favor local recall of inflammatory factors that can stimulate the initiation of HO development. 27 According to the data obtained, we believe that the presence of drainage could favor the development of HO after THA.
Another important finding of this study is the result of the direct comparison of the postoperative complications and joint dysfunction between the two groups. We assessed blood loss by comparing the Hb concentration preoperatively and postoperatively and found that the patients in the drainage group may have had more blood loss in the first few days after THA; 5 days postoperatively, however, the difference was not significant. Seven (7.0%) patients in the drainage group and five (5.1%) in the non-drainage group required blood transfusion. However, this difference was not significant. These findings indicate that non-drainage may reduce postoperative blood loss but that it has no benefits regarding reduction of blood transfusion. Moreover, we found that no drainage can increase the number of dressing changes. This was probably because the hematomas caused high incision tension and wound discharge, increasing the need for dressing changes in the non-drainage group. 28 Another finding is that the CRP concentration and ESR were higher on postoperative days 1 and 5. Moreschini et al. 29 reported that the CRP concentration peaked on day 3 followed by a comparatively rapid return to the baseline level compared with the ESR. Katoh et al. 30 reported that the CRP concentration and ESR increased early in the postoperative period and remained high even at 14 days postoperatively compared with the respective baseline levels of each. The ESR and CRP concentration were significantly lower in the drainage than non-drainage group on postoperative day 5. However, Smolle et al. 31 considered that there is no correlation between this phenomenon and post-THA infection. Several studies have suggested that drainage reduces the incidence of postoperative hematomas and thus the incidence of infection. 32 However, some other researchers have argued that the placement of a drainage tube may increase the infection rate because it provides a portal of entry for bacteria and their retrograde colonization. 33 Six patients in our study developed superficial infection, but no periprosthetic infection was found in our series. Wound drainage has benefits in terms of pain relief and reduced hematoma formation, but the VAS score was generally low (<3 points) in both groups of our study. Drainage can also cause restriction of early postoperative exercise, but it has no effect on the final hip ROM and HHS. 28 There are advantages and disadvantages to both types of management, but non-drainage is more advantageous in terms of the long-term outcome, particularly in reducing the incidence of HO. Similar to our findings, Xu et al. 34 concluded that drain use was associated with a higher transfusion rate and a longer postoperative length of stay in patients undergoing routine primary THA. Some recent studies have recommended no closed-suction drainage after THA because the routine use of drainage may be of more harm than benefit. 35
This study has several limitations. First, whether the drain was clamped and the clamping time were not recorded in detail in the database; thus, we were unable to determine the effect of drain clamping on the incidence of HO. Second, the drain removal criteria were not recorded, which may have affected the blood loss volume. Third, the details of drainage use (other than yes or no) were not recorded in the database, and the starting time for drainage differed for each hospital, which may have affected the blood loss volume and transfusion rate. Despite these limitations, to the best of our best knowledge, our study is one of the few studies to compare the effects of drainage versus no drainage on HO. Our study provided conclusive results regarding the use of drainage in THA and its effect on HO, postoperative complications, and joint dysfunction. The results of this study could become an important component of orthopedic surgeons’ decision-making regarding drainage in routine primary THA.
Conclusion
The current study showed that drainage was associated with a significantly higher incidence of HO in patients undergoing primary unilateral THA. We are of the opinion that the traditional practice of draining the wound has no effect on the postoperative control of infection and blood loss. Although no drain usage is associated with more dressing changes postoperatively, higher VAS scores, and a greater hematoma volume, the benefits of drain usage are still very limited. In addition, the presence or absence of drainage showed no significant effect on the long-term function of the hip joint.
Footnotes
Availability of the data and materials
All the data generated or analyzed during this study are included in this published article.
Authors’ contributions
All authors have read and approved the manuscript.
Declaration of conflicting interests
All the authors declare that they have no conflicts of interest with any organization that sponsored the research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Nature Science Foundation of Hebei, China (No. H2019206609).
