Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection caused the pandemic of coronavirus disease 2019 (COVID-19). Gastrointestinal (GI) involvement is common among patients with COVID-19, and GI symptoms can appear earlier than respiratory symptoms. Except for direct infectious effects, patients infected with SARS-CoV-2 are at risk of complications requiring gastroenterological management. Diarrhea is the most common GI symptom in patients with COVID-19 and occurs in up to half of them. Other GI symptoms, such as anorexia, discomfort, nausea, abdominal pain, loss of taste sensation, and vomiting, have been reported. GI symptoms are associated with a poor prognosis. Fecal viral excretion may have clinical significance because of the possible fecal–oral transmission of infection. In the present narrative review article, six different aspects of studies published to date are summarized as follows: GI manifestations of COVID-19, the roles of fecal–oral transmission, poor prognosis of GI symptoms; abnormal abdominal imaging findings, COVID-19 in patients with irritable bowel disease, and prevention and control of SARS-CoV-2 infection in the digestive endoscopy room. Timely understanding of the association between COVID-19 and the digestive system and effective preventive measures are critical to improve this disease and help clinicians take appropriate measures to mitigate further transmission.
Keywords
Introduction
Coronavirus disease 2019 (COVID-19), which was first detected in Wuhan City, Hubei Province, China, is caused by the novel human severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which was isolated on 7 January 2020. 1 On 11 March 2020, the Director-General of the World Health Organization (Geneva, Switzerland) defined COVID-19 as a pandemic because of its rapid global spread.2,3 As of 28 August 2022, more than 598 million confirmed cases and more than 6.4 million deaths have been reported globally (https://www.who.int/emergencies/diseases/novel-coronavirus-2019).
Respiratory symptoms, especially cough and dyspnea, are the most common clinical manifestations in patients with COVID-19. 4 However, SARS-CoV-2 affects the lungs of infected patients and damages other organs of the body. 5 The most attacked organs, such as the heart, blood vessels, kidneys, intestines, nervous system, and brain, have more angiotensin-converting enzyme 2 (ACE-2) receptors attached to the SARS-CoV-2 surface, which allows it to penetrate into infected cells.3,6 SARS-CoV-2 is more contagious than other known coronaviruses because of its stronger affinity for ACE-2, but it is not as serious as other viruses that cause atypical pneumonia.7,8 The median incubation period of this virus is approximately 4 days, but can be as long as 12 days, based on findings of previous reports. 6 Transmission appears to be unrelated to clinical symptoms, but it is most closely related to viral load, which reaches its peak 10 days after the onset of symptoms.
The respiratory tract is the main entry point for SARS-CoV-2, although many reports describe gastrointestinal (GI) involvement, which can cause nausea, vomiting, diarrhea, and highly persistent viral particles in the stool. 5 The presence of ACE-2 and transmembrane protease serine 2 in intestinal epithelial cells and the oral mucosa can explain the fecal–oral route of transmission (Figure 1). This observation poses new challenges for the diagnosis and control of COVID-19. The intestinal manifestations of patients with COVID-19 are reviewed in this review, using published studies that provide the diagnosis and prevention strategies from the perspective of gastroenterology. Understanding the association between COVID-19 and the digestive system and taking effective preventive measures are critical for an improvement of this disease, which could help clinicians take appropriate measures to reduce further transmission.
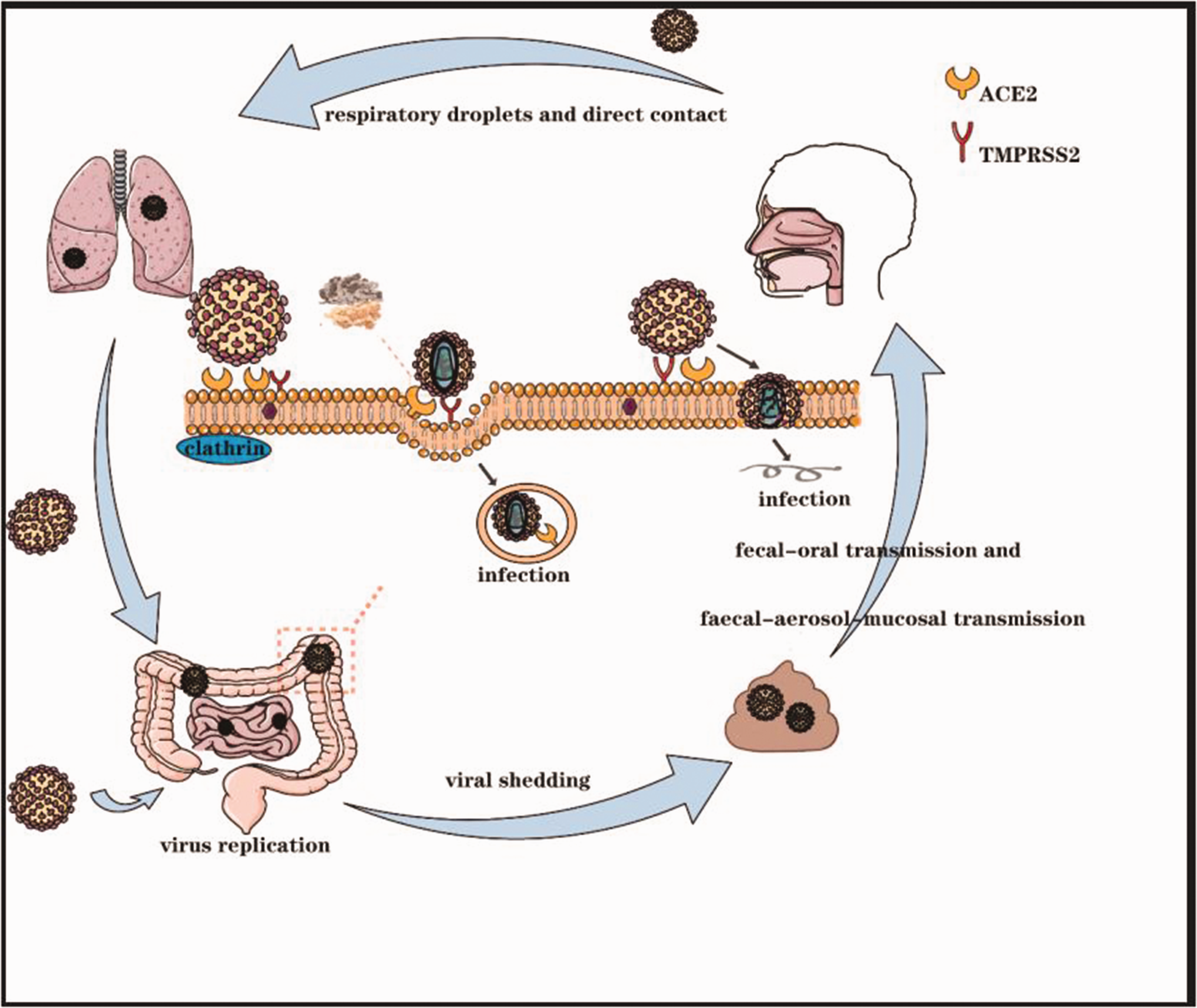
Schematic diagram of the transmission and pathogenesis of severe acute respiratory syndrome coronavirus 2 in the gastrointestinal tract.
Materials and methods
Databases, such as PubMed and EMBASE, and diverse websites of the International Gastroenterology Society were searched to obtain various published digestive system-related retrospective clinical studies, case reports, and society recommendations. In addition, articles, reports, and papers listed in the references and cross-referencing were extensively manually searched. The search terms in our review included “SARS-CoV-2,” “COVID-19,” or “2019-nCoV” and “digestive,” “gastrointestinal,” “IBD,” “abdominal imaging,” or “endoscopy.”
Results
GI manifestations of COVID-19
During an outbreak of viral infections with respiratory symptoms, such as influenza virus H1N1, GI symptoms (e.g., vomiting, diarrhea, and abdominal pain) are often present. GI symptoms similarly occur in up to 30% of patients with SARS and Middle East respiratory syndrome.9,10 Data show that GI involvement in SARS-CoV-2 infection is relatively less than that in Middle East respiratory syndrome and SARS infection. 11 However, some reports have suggested that diarrhea is a prognostic factor for COVID-19.12,13 Studies have also shown that a small number of patients only have GI symptoms at the beginning of infection, and these manifest before respiratory symptoms. 13 Furthermore, the first patient with COVID-19 in the United States had general and respiratory symptoms, nausea, and vomiting, and later had abdominal discomfort and diarrhea. 14 GI involvement in patients with COVID-19 may be underestimated because respiratory symptoms initially occur in most patients. In the early stages of the COVID-19 outbreak, GI symptoms were overlooked or not recognized because the incidence of GI symptoms is less common and greatly varies among different study populations. 15 During the COVID-19 pandemic, the available medical resources and expertise were rapidly outstripped by the substantial medical demands because of the tremendous increase in the number of patients. The obvious respiratory symptoms and life-threatening complications of these patients were the primary concerns of medical staff because of the rapidly increasing requirement for medical care. Therefore, GI symptoms may be neglected in patients, particularly in critically ill patients. 11
Fecal–oral transmission is a potential route of transmission of COVID-19
Cross-species infection, numerous asymptomatic cases, and unusual and rapid modes of transmission are reminders that other potential modes of SARS-CoV-2 transmission may exist, although the main mode of spread is through droplets. In addition to the clinical symptoms, endoscopic specimens of the duodenum, rectum, stomach, and esophagus have shown the presence of SARS-CoV-2 RNA. Recent studies have estimated that 35.7% to 54.5% of all confirmed cases of infection were detected in SARS-CoV-2 RNA-positive stool samples. Stool specimens of patients with COVID-19 have been continuously shown to have a large amount of SARS-CoV-2 RNA.16,17 Notably, the first confirmed patients with COVID-19 in the United States reported experiencing vomiting and diarrhea for 2 days before admission and positive fecal (not serum) viral RNA testing results. 14
In addition, the feces of infected patients can remain positive for SARS-CoV-2, although its clearance has occurred in the respiratory tract. 18 SARS-CoV-2 has a remarkably longer duration in stool samples than in respiratory and serum samples. 17 Therefore, the management of stool samples should be strengthened to prevent and control the epidemic. In a prospective study by Sun et al. 17 on 49 patients with COVID-19 in China, persistent viral RNA shedding in body fluids, especially fecal and nasopharyngeal samples, was observed. These results indicate that SARS-CoV-2 is an enterovirus, although the detection of the fecal high copy number of viral RNA is not equal to viral shedding or the spread of disease. Moreover, the high ACE-2 protein expression in the gland cells of the stomach, duodenum, and rectal epithelium support the entry of SARS-CoV-2 into host cells. SARS-CoV-2 attaches to ACE-2 receptors of host cells to enter them, followed by the activation of viral internalization using proteases (especially transmembrane protease serine 2). This process is the underlying mechanism of viral entry into a human host (Figure 1). Fecal–oral transmission is a potential route of transmission; therefore, infected patients should undergo routine fecal testing. 19 Fecal–oral transmission of the virus can still occur in “cured” patients.20,21 Therefore, fecal viruses should routinely be tested for in patients with COVID-19. Postponed discharge or other isolation measures should be performed in patients with positive fecal viral testing.
After an extensive observation of viral shedding, Xiao et al. 16 reported successfully isolating infectious SARS-CoV-2 from fecal matter. In addition, Zhang et al. characterized the presence of fecal live virus, and in Vero cells, they successfully cultured SARS-CoV-2 isolated from a stool specimen of a patient with severe COVID-19. 22 Furthermore, SARS-CoV RNA was previously detected in the sewage of patients with SARS. 23 Viral infections in the sewage lasted up to 2 weeks under experimental conditions. 23 At least two recent studies have shown that SARS-CoV-2 also appears to have a similar stability in the environment. SARS-CoV-2 was detected in untreated sewage in Australia. 24 When people eat food or water contaminated by SARS-CoV-2, they may be infected with SARS-CoV-2 through the fecal–oral route. Yeo et al. 12 reported that the fecal–oral transmission of COVID-19 may have clinical implications for infection control, particularly in areas with poor sanitation. Therefore, strict management and effective disinfection of the environment or objects that may be contaminated by feces of patients with COVID-19 are important to control the COVID-19 pandemic.
Poor prognosis of GI symptoms
Wan et al. studied 232 patients with COVID-19 from hospitals in Hubei, Jiangxi, and Guangdong. They found that diarrhea symptoms developed in 21% of patients during hospitalization, and this rate is much higher than 3% reported in another study. 25 There are also reports in the literature of acute hemorrhagic colitis as well as GI bleeding.26–28 Importantly, one study showed that diarrhea symptoms were strongly associated with severe infection with COVID-19, use of a ventilator, and admission to the intensive care unit (ICU). 25 Patients with SARS with diarrhea symptoms were similarly more likely to require ventilator support and be admitted to ICUs than those with SARS without diarrhea during the outbreak in 2003. 10 Zhang et al. 29 suggested that GI symptoms may have prognostic implications, and patients with diarrhea are more likely to present with storms of lymphopenia, cytokines, and neutrophilia, which lead to multiorgan damage. Inflammatory patterns are independent factors related to the course of illness of the survivors and in-hospital death due to severe COVID-19.
Portal venous gas and pneumatosis are usually present in patients with mesenteric ischemia, which is common in critically ill patients. Data from major health care networks in New York City have also suggested that there is a significant association of GI symptoms (defined as nausea/vomiting and diarrhea at presentation with positive test results) with people presenting for COVID-19 testing. GI symptoms are associated with a 70% increase in the relative risk of a positive test. However, investigators have also found that the appearance of GI symptoms among patients with COVID-19 is associated with a longer duration of this disease, lower ICU admission rates, and lower mortality during short-term follow-up.30,31 Therefore, more data are required to verify whether GI symptoms are associated with severe COVID-19. In addition, novel intervention strategies can be developed with an understanding of the underlying mechanisms of these correlations to reduce or prevent severe outcomes of COVID-19. An example of such strategies is that residents are urged to do pay attention to hand hygiene, wash hands frequently, pay attention to food hygiene, and keep toilets clean. Furthermore, the management of drinking water in rural areas should be strengthened, and fecal water in rural areas needs to be properly disposed of. We should also strengthen the monitoring and control of domestic sewage, and actively carry out environmental sanitation work in families and communities. At the same time, terminal disinfection should be performed in an environment where an infected person is located after the nucleic acid test is negative.
Abnormal abdominal imaging findings
The abdominal phenomena of SARS-CoV-2 infection can be understood by using imaging findings. Investigators of a retrospective study that included 412 patients with COVID-19 proposed the following symptom that cannot be ignored: patients with new coronary artery disease are prone to intestinal abnormalities. 32 Bowel wall thickening, portal venous gas, and pneumatosis are common findings on computed tomography. Computed tomographic images show that mesenteric ischemia occurs in some patients with portal vein gas and emphysema after further surgery. The remaining patients may present with intestinal infarction or corresponding yellow patchy discoloration on the antimesenteric sides of the transverse colon of unknown origin. To date, published studies have also reported abdominal imaging findings, which include small and large bowel wall thickening, a fluid-filled colon, pneumatosis, pneumoperitoneum, intussusception, and ascites. 33 Culver et al. 34 also found that one patient with COVID-19 and ascites, as indicated by ultrasound, had SARS-CoV-2 RNA-positive ascites samples. Abnormal abdominal imaging findings, which include direct viral infection, nonocclusive mesenteric ischemia, and small-vessel thrombosis, in patients with COVID-19 may be caused by different mechanisms. 32
The abundant ACE-2 expression on the surface of the alveolar epithelium, vascular endothelium, and enterocytes of the small intestine suggests that the vasculature and small intestine may be susceptible to SARS-CoV-2 infection. The presence of fibrin clots indicates edema, inflammation, ischemia, or cytokine storms caused by intestinal epithelial SARS-CoV-2 infection. 33
COVID-19 in patients with inflammatory bowel disease
Treatment of uncontrolled inflammation is the main therapeutic strategy for inflammatory bowel disease (IBD), and immunotherapy is warranted for most patients with IBD. 35 However, these treatments may weaken the immune system, thereby increasing the risk of infection by pathogens, such as SARS-CoV-2, and the risk of IBD complications. 36 SARS-CoV-2 infection is not more likely to be present in patients with IBD, and no evidence has demonstrated that an increased risk of COVID-19 is associated with IBD treatment. 35
Some manifestations in patients with COVID-19 may be similar to deterioration in IBD. Patients with infectious bursal diseases are recommended to receive SARS-CoV-2 testing before making a diagnosis because there is no evidence to support the notion that SARS-CoV-2 infection is the cause of IBD. 37
Atypical presentations of viral diseases usually occur in immunosuppressed patients. 38 However, the use of immunomodulators has not been recognized as a risk factor for the severity of COVID-19 even though viral burdens are theoretically increased by a reduced immune surveillance. 39 Patients with IBD infected with SARS-CoV-2 are presumably at a higher risk of developing complications owing to the increased ACE-2 expression and trypsin-like protease activities of host cells in their inflamed guts. This possibility indicates that an inflamed gut is a channel for the virus to enter host tissues. However, reports have shown that the risk of developing COVID-19 is not increased in patients with IBD because gut ACE-2 and TMPRSS2 expression is not elevated by inflammation or IBD medication. 40
The anti-COVID-19 vaccination changed dramatically the pandemic, and patients with IBD had reduced immune responses following vaccination compared with the general population. Factors including the use of immunomodulators and anti‐tumor necrosis factor agents reduce response rates. Despite decreased efficacy associated with immune-modifying medication, most vaccines are broadly recommended for patients with IBD.41,42
Endoscopy is the mainstay for the diagnosis and management of patients with IBD. During the COVID-19 epidemic, a complete change in management is required for patients with IBD who need to undergo endoscopic procedures. Several preventive measures, such as the use of personal protective equipment, greater attention to endoscopic room hygiene, and rescheduling of nonurgent procedures, must be taken to avoid the spread of infection among health care professionals and patients with IBD. 43
Prevention and control of SARS-CoV-2 infection in the digestive endoscopy room
Gastroduodenoscopy is a common method of examination and treatment of upper digestive tract diseases. SARS‐CoV‐2 is highly infectious and spreads through respiratory droplets and direct contact. COVID-19 is primarily regarded as a respiratory disease, 44 although there is considerable evidence of possible fecal–oral transmission in SARS-CoV-2. 45 A recent proof-of-concept study, which used a laser particle counter, showed that esophagogastroscopy was related to an increase in aerosol-sized particles. 46 This finding provides evidence to support the general consensus that upper endoscopy results in aerosol generation. 47 During the height of SARS-CoV-2 infection, medical personnel involved in endoscopy were in close contact with patients. Therefore, occupational exposure and cross-infection easily occurred, which posed a great danger to endoscopy personnel.
After the occurrence of pneumonia in SARS-CoV-2, gastroenterologists should know how to prevent it further as follows. First, patients with potential SARS-CoV-2 infection should be risk-stratified, and patients should be pre-examined. If endoscopy is considered for suspected cases, the best practice is to conduct polymerase chain reaction testing or detect SARS-CoV-2 antibody. If any test is positive, endoscopy should be postponed, except for emergency endoscopy. Second, semi-emergency endoscopic surgery for patients with confirmed or suspected COVID-19 should be postponed as long as possible. Appropriate reduction of endoscopy is helpful in minimizing exposure to SARS-CoV-2. Finally, all staff working in endoscopy units should receive training on infection control of SARS-CoV-2, including the proper use of personal protective equipment during endoscopy. Personal protective equipment should be optimized, with regard to the risk of infectious atypical pneumonia. 48
SARS-CoV-2 has caused unprecedented public health emergencies in the world and has become a serious threat because of its strong human-to-human transmission abilities. This new coronavirus is continually undergoing new mutations and is extremely contagious. SARS-CoV-2 can bypass ordinary epidemic prevention measures, such as wearing masks or washing hands. Therefore, individuals must be more vigilant about potential fecal–oral transmission. Furthermore, fecal SARS-CoV-2 has been identified in a large proportion of patients with COVID-19. This phenomenon poses new challenges to the diagnosis and treatment of COVID-19, which requires proper hygiene and sanitation.
The penetration of SARS-CoV-2 into host cells is dependent on surface ACE-2 expression. Therefore, tissues with high ACE-2 expression levels are presumed to be susceptible to direct infection. ACE-2 surface expression is most abundant in alveolar epithelial cells, vascular endothelium, and enterocytes of the small intestine. Liver injury and GI symptoms caused by SARS-CoV-2 infection may be associated with a large amount of ACE-2 surface expression in the GI tract. A previous review showed that SARS-CoV-2 and SARS-CoV, which belong to the same β-coronavirus family, have highly similar genomes. 49 In addition, ACE-2 is the receptor of SARS-CoV and SARS-CoV-2. Therefore, the digestive symptoms caused by SARS-CoV-2 are similar to those caused by SARS-CoV.
After a patient is infected with SARS-CoV-2, the overactive host immune system releases inflammatory mediators, leading to a “cytokine storm.” This increase in inflammatory mediators leads to high permeability of the lungs, which allow viruses and inflammatory mediators to migrate to the intestine through the circulation and bind to ACE-2 receptors that are highly expressed on intestinal cells. SARS-CoV-2 reduces the expression of the ACE-2 receptor, which affects the microbial composition and host immune system. Inflammatory mediators destroy the permeability of the intestinal tract, which causes intestinal microbes and related metabolites to leak into the circulation. The leaked microorganisms and products migrate through the circulation to the organs, including the lungs, and produce abnormalities, which lead to acute respiratory distress and multiorgan failure (Figure 2). In addition, the amino acid transport function of ACE-2 is associated with GI microbial ecology, thereby indicating that COVID-19 may be associated with the intestinal microflora to some extent. Microbial disorders may explain the main GI symptoms (such as diarrhea) of patients with new coronary pneumonia.

The lung–gut axis in coronavirus disease 2019. ACE2 receptors are expressed in the lungs and intestinal epithelium. SARS-CoV-2 recognizes ACE2 receptors in host cells, and the virus begins to replicate in host cells. In the lungs, once the virus enters the cell, it activates the immune system, which is characterized by pro-inflammatory cytokines and immune cell recruitment. If the virus is not sufficiently cleared, the immune response further develops into cytokine storm syndrome. In the gut, the virus targets cells and triggers an immune response in a similar way. The immune response also destroys intestinal permeability, causing intestinal microorganisms and related metabolites to enter the bloodstream, leading to further biological disorders.
Conclusion
The fecal–oral route of viral transmission of SARS-CoV-2 is possible. Therefore, exposure to possible sources of contamination, such as vomitus, feces, and other body fluids, should be avoided by using additional precautions. Moreover, washing hands properly, taking additional precautions for using public toilets, and controlling open defecation in developing and poor countries should be emphasized.
Footnotes
Author contributions
Chaoyang Xu and Shuxun Jin designed the review and analyzed and interpreted the data. Shuxun Jin drafted the manuscript. Chaoyang Xu and Xiaofeng Lu critically revised the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Zhejiang non-profit technology applied research projects of China (Grant no: LGF20H160017) and Jinhua non-profit technology applied research projects of Zhejiang China (Grant no: 2018-4-016).
