Abstract
Paraquat is a non-selective contact herbicide that is absorbed through the digestive tract and skin and can cause multiple organ damage. The toxicokinetics of paraquat poisoning in specific patients are rarely reported. Case 1 was a 76-year-old man who intermittently immersed his perineum in diluted paraquat solution for 3 consecutive days because of eczema of the perineal skin. On admission, the patient’s scrotal skin was severely corroded and his blood paraquat concentration was 0.5 μg/mL. He developed severe kidney and lung damage after admission and died on Day 6 of admission. Case 2 was a 23-year-old woman who ingested paraquat during gestational week 36. Her initial blood paraquat concentration was 0.81 μg/mL. The patient refused hemoperfusion and a cesarean section. She birthed a baby girl 83 hours after ingesting paraquat. Paraquat concentrations in postnatal maternal blood, fetal blood, umbilical cord blood, and amniotic fluid were 0.19 μg/mL, 0.23 μg/mL, 0.20 μg/mL, and 0.47 μg/mL, respectively. The baby died within hours of birth and the mother died of refractory respiratory failure 2 days after delivery. This paper provides clues about paraquat toxicokinetics in specific patient types and indicates that paraquat can be absorbed through the scrotal skin and the placental barrier.
Keywords
Introduction
Paraquat is a non-selective contact herbicide that is predominantly used to control weeds in agricultural fields. 1 The herbicide is absorbed through the digestive tract and skin and causes multiple organ damage. 2 Skin exposure to paraquat can result in local redness and swollen cankers. 3 Only a few cases of visceral damage from paraquat poisoning via skin absorption have been recorded. 1 In most cases, this damage can be clinically cured and poor prognosis is rarely reported. 4 Paraquat poisoning in pregnant women is likewise rarely reported. Paraquat may cross the placenta;5,6 however, its toxicokinetics in pregnant women and fetuses are unclear.6,7 Here, we report two cases of unusual paraquat poisoning. This paper highlights the toxicokinetics of paraquat and suggests that this herbicide can penetrate the scrotal skin and placental barrier.
Case report
Case 1
A 76-year-old man complained of eczema of the scrotum. The patient had soaked his scrotum in diluted paraquat solution 5 days before the hospital visit. The treatment was continued 3 to 4 times a day and spanned 10 to 20 minutes per treatment. The patient initially felt tingling from eczema pain. Three days before the hospital visit, he discovered severe damage to his scrotal skin and experienced pain, swelling, and erosion of the scrotum. The patient subsequently increased treatment length and paraquat concentration. A day before the visit, he developed mild chest tightness and visited another local hospital, where scrotal cleaning and rehydration were performed. Scrotal damage did not resolve and the patient’s chest tightness worsened. Thereafter, the patient was transferred to our hospital. Physical examination on admission revealed a temperature of 36.8°C, a heart rate of 94 beats/minute, a respiratory rate of 18 breaths/minute, blood pressure of 169/90 mmHg, and oxyhemoglobin saturation of 98%. The patient had clear consciousness but was in poor spirits. Wet rales were heard in both lungs. The scrotum was red, swollen, and eroded and the right groin was marked with blood scabs and exudation (Figure 1). The remaining physical parameters were normal on examination. Urine turned mild blue with sodium bicarbonate and sodium bisulfite testing, leading to a preliminary assessment that paraquat was present. Laboratory test results were as follows: white blood cell (WBC), 12.86 × 109/L (normal value: 3.5–9.5 × 109/L); red blood cell (RBC), 4.71 × 1012/L (normal value: 4.3–5.8 × 1012/L); neutrophil ratio (NEU%), 80.70% (normal value: 40%–75%); lymphocyte ratio (LYM%), 6.00% (normal value: 20%–50%); alanine transaminase (ALT), 166 IU/L (normal value: 21–72 IU/L); aspartate aminotransferase (AST), 85 IU/L (normal value: 17–59 IU/L); total bilirubin (TBIL), 28 μmol/L (normal value: 3–22 μmol/L); and creatinine (Cr), 296 µmol/L (normal value: 58–133 µmol/L). Blood paraquat concentration was 0.5 μg/mL. Initial treatment mainly included dexamethasone (40 mg, once a day), pantoprazole (40 mg, twice a day), reduced glutathione (2.4 g, once a day), alanyl-glutamine (10 g/50 mL, once a day), torasemide (20 mg, twice a day), and flucloxacillin (1.0 g, once every 8 hours). A kangfuxin solution (Chinese Medicine Approval: Z53020054, Kunming Sino Pharmaceutical Co. Ltd., Kunmin, China) was applied externally to the patient’s scrotal skin. On the second day of admission, the patient complained of pain and scrotal bleeding and the exudation persisted. On Day 3, the patient presented with obvious abdominal distension and pain and decreased bowel sounds.

The patient’s scrotal ulceration upon admission.
Repeated laboratory test results were as follows: WBC 12.85 × 1012/L; RBC, 4.23 × 109/L; NEU%, 80.90%; ALT, 148 IU/L; AST, 111 IU/L; TBIL, 15.8 μmol/L; Cr, 440 µmol/L; and blood urea nitrogen (BUN), 28.14 mmol/L. Coagulation function, myocardial enzyme, blood amylase, and lipase levels were normal. Gastrointestinal decompression was performed. In the afternoon, the patient complained of chest tightness and increased chest pain, and his blood oxygen saturation level decreased to 70%. Chest computed tomography showed diffused infiltration with bilateral lung distribution. On Day 4 of hospitalization, the patient continued to deteriorate, appeared drowsy, presented with shortness of breath, and had difficulty breathing. Oxygen was administered through a mask at 5 L/minute. The patient’s condition subsequently deteriorated and blood oxygen saturation decreased. Arterial blood gas analysis results were as follows: pH, 7.23; PaCO2, 28 mmHg; PaO2, 42 mmHg; Lac, 4.6 mmol/L; and K+, 3.35 mmol/L. After communication with the patient's family, endotracheal intubation and ventilator were refused. On physical examination, wet rales could be heard in both lungs. On Day 6 of admission, the patient died of cardiac arrest.
Case 2
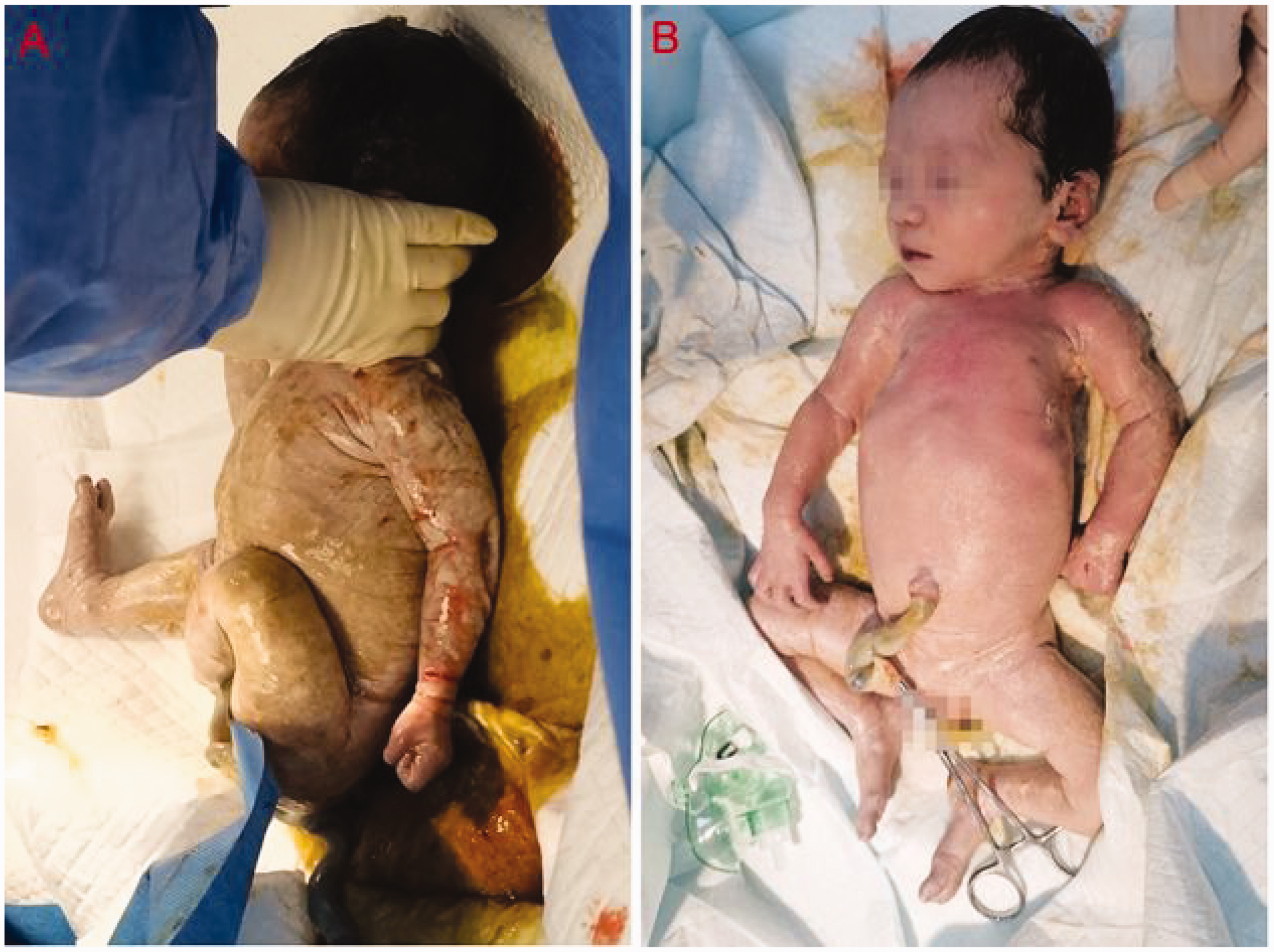
A 23-year-old pregnant woman with psychosis ingested approximately 100 mL of paraquat (W/V, 20%) during gestational week 36. Gastric lavage was performed and the patient was transferred to our department for further treatment 6 hours after exposure. At admission, the patient had a Glasgow Coma Scale score of 15, a heart rate of 75 beats/minute, a blood pressure of 133/89 mm Hg, and a blood oxygen saturation level of 99%. The fetal heart rate was 135 beats/minute. The patient’s laboratory test results were as follows: WBC, 7.56 × 109/L; NEU%, 83.6%; hemoglobin, 102 g/L; D-dimer, 4.38 μg/mL; and potassium, 3.41 mmol/L. The patient’s myocardial enzyme levels, and liver and renal function were normal. The patient’s urine turned blue when tested with dithionate (Figure 2) and her blood paraquat concentration was 0.81 μg/mL. The patient refused hemoperfusion and a cesarean section. She received activated carbon (Smecta®) and mannitol by oral administration for gastrointestinal decontamination. Other treatments included dexamethasone (40 mg, once a day), lansoprazole (30 mg, twice a day), reduced glutathione (1.8 g, once a day), alanyl-glutamine (10 g, once a day), flucloxacillin (1.0 g, once every 6 hours), furosemide (20 mg, twice a day), and symptomatic therapy. On Day 3, the patient experienced mild chest congestion. Urine volume was normal, and laboratory results showed Cr of 222 μmol/L and BUN of 15.3 mmol/L. Eighty-one hours after ingestion, the patient’s dyspnea worsened. She experienced throat and abdominal pain and regular contractions. The fetal heart rate was 143 beats/minute. Two hours later, the patient vaginally birthed a baby girl (weight, 2250 g; Apgar score 5 minutes after birth, 5; Figure 3
The mother’s urine was tested with dithionate 6 hours after paraquat ingestion. (a) The baby’s skin was soaked with contaminated amniotic fluid at the moment of birth. Text boxes in Figure 3a correspond to Figure 4b–f and (b) The baby 20 minutes after birth, immediately following cardiopulmonary resuscitation.

Pathology of the placenta: (a) H&E (hematoxylin and eosin ) × 20 and (b–f) H&E × 200. (a) Placental tissue without appreciably abnormal lesion, (b) well-developed villi and intact chorionic vessels, but capillary diffusion and congestion; (c) fibrin deposition between intervillous lacunae; (d) normal venous tissue; (e) normal arterial tissue; (f) polyps and karyorrhexis in trophoblast membranes. Figure 4b–f corresponds to text boxes in Figure 3a.
Discussion
Paraquat is an effective, broad-spectrum, and highly poisonous water-soluble quaternary ammonium salt herbicide. 1 Paraquat is highly toxic to humans; the main prognostic factors of paraquat absorption are pulmonary damage and fibrosis.1,2,8 Paraquat can rapidly enter the body through ingestion and skin absorption. 9 After absorption, paraquat is rapidly distributed to the liver, kidneys, lungs, and other organs. 2 The volume distribution of paraquat is 1.2 to 1.6 L kg−1, and the unchanged drug excretion rate up to 48 hours (not metabolized, predominantly urinary) is 82.9%.10,11 Once paraquat is distributed into deeper body tissues, it is slowly eliminated over several days to weeks. 12 This may explain the continued aggravation of organ damage after paraquat is eliminated from the blood through medical intervention. In this case series, Patient 1’s case indicated that paraquat can be absorbed through the scrotal skin and can lead to adverse consequences. The case of Patient 2 indicated that paraquat can be absorbed through the placental barrier and can lead to fetal death. However, the toxicokinetics of acute paraquat poisoning in such special patients is rarely reported.
In 1983, scrotal skin exposure to a concentrated paraquat solution resulted in liver and kidney damage and respiratory failure in a patient, indicating that paraquat can be absorbed through perineal skin and produce significant systemic toxic effects. 13 Damaged skin is a prerequisite for a considerable level of paraquat absorption.1,14 Paraquat is highly corrosive, easily damages the skin and mucous membranes and may further aggravate previous skin diseases, which increases the likelihood of absorption. 14 Scrotal skin is rich in blood supply, is thin and tender, and has many folds—characteristics that facilitate paraquat residue and skin damage. Patient 1 was repeatedly exposed to paraquat solution for nearly 4 days. His blood paraquat concentration was 0.5 μg/mL at admission. The average distribution half-life of plasma paraquat concentration is 5 hours while the average elimination half-life is 84 hours. 15 In contrast, the major indicator of plasma paraquat concentration remained high despite in Patient 1 despite the patient being transferred to our hospital 2 days after cessation of skin exposure to the herbicide. The patient initially presented with localized redness and swelling. He received periodic iodophor disinfection, repeated blood perfusion, and high-dose hormonal shock treatment. However, the patient developed severe renal impairment and pulmonary fibrosis and ultimately died from his illness. Therefore, we suspect that the patient’s condition was related to the presence of scrotal eczema and associated surface skin damage, repeated immersion in paraquat solution, and the accelerated absorption of the chemical through the skin, leading to the extended presence of paraquat in the body.16,17
Paraquat poisoning by oral ingestion in pregnant women is rare.5–7 The case of Patient 2 provides clues about the toxicokinetics of paraquat in pregnant women and fetuses. Recent studies indicate that the toxic effects of fetal exposure to paraquat are complicated, and the toxicokinetics of the placenta may be independent of maternal and fetal metabolizing organs. 18 Previous reports showed that after paraquat was administered intravenously to pregnant rats, high concentrations were found in the placenta and throughout the fetuses 0.5 hours after administration. 19 In our case, childbirth occurred 83 hours after paraquat ingestion. Therefore, paraquat's absorption, distribution, metabolism, and elimination were relatively complete. Despite a lack of serious injury to the patient’s placenta, paraquat had nevertheless easily crossed the placenta during gestational week 36 and the fetus had initially released paraquat into the amniotic fluid.
A specific antidote for paraquat is not available. In the case of paraquat poisoning through skin absorption, early skin decontamination interventions are critical. In paraquat poisoning during the late trimester of pregnancy, a timely cesarean section should be performed.6,7 Simultaneously, early blood perfusion is an essential prerequisite for treating severe paraquat poisoning. 17 Further understanding of the toxicokinetics of paraquat in specific patient types has important clinical significance and will guide better treatment strategies.
Ethics statement
Written informed consent for publication of patient information and images was obtained from the patients’ families. Ethics approval was obtained from the Qilu Hospital Ethics Committee for Human Research (KYLL-202106(KS)-040).
Footnotes
Data availability statement
The reporting of this study conforms to the CARE guidelines. The original research data can be obtained from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Qilu Hospital, Shandong University under Grant KYLL-2019-296.
