Abstract
Excessive daytime sleepiness (EDS) is classified as a neurofunctional disorder that manifests as uncontrolled sleeping propensity in the daytime. Currently, consistent and effective therapeutic approaches for EDS are lacking. Stellate ganglion block (SGB) has a clear effect in various complicated pain syndromes, vascular insufficiency, hyperhidrosis, and posttraumatic stress syndrome. We report an EDS case that involved a patient who recovered after several sessions of SGB.
Keywords
Introduction
Sleep and wakefulness are among the most critical physiological rhythms in humans. A regular and healthy sleep cycle substantially refreshes the entire body, ensuring productive fitness. Conversely, abnormal sleep induces discomfort, and leads to detrimental outcomes in multiple diseases. 1 Sleep disorders interrupt normal sleep patterns, which might trigger excessive daytime sleepiness (EDS). In some severe cases, patients can be woken only by very strong stimuli, which reduces their quality of life and threatens daily safety. 2 Treatments for EDS comprise medication-based and non-medication approaches. Regarding the former, neurostimulants and dopamine receptor agonists are common choices, while the latter aim to recover healthy sleep routines through psychobehavioral therapies.3,4 The stellate ganglion refers to the assembly of the inferior cervical and superior thoracic ganglia, which when blocked by a local anesthetic, can resolve several complex pain syndromes, posttraumatic stress syndrome, and chronic regional pain syndrome. 5 Studies have revealed that repeat stellate ganglion blocks (SGB) effectively activate autonomic nerves, thereby adjusting autonomic nerve function.6,7 By maintaining the sympathetic and parasympathetic balance, SGB is effective for migraine, cluster headache, insomnia, and premenstrual syndrome.4,5 An imbalance between sympathetic and parasympathetic tone facilitates the disruption of the sleep–wake cycle. 8 Therefore, SGB might be helpful for EDS treatment. This hypothesis was confirmed in the present case, where a patient with EDS recovered dramatically after several sessions of SGB.
Case presentation
The present study fully conformed to the CAse REporting (CARE) guidelines. 9 A female patient in her early 30s (height: 158 cm, weight: 44 kg) visited our clinic with complaints of 1 year of daytime sleepiness, weakness in the lower limbs, fecal incontinence, and urgent or frequent urination. The patient’s sleep duration was more than 8 hours each night. However, she remained somnolent in the daytime, especially when alone, and even when speaking or eating. The resulting weariness reduced her ability to perform light physical activities, and she found it difficult to stay awake and alert. She went to sleep unintentionally or at the wrong time almost every day. She also mentioned a 3-month burning sensation in both lower extremities and a 1-week-duration of poor appetite. Except for the consistent oral administration of oryzanol (30 mg/day), no other medications were recorded. The patient’s condition had been deteriorating since the onset of the symptoms, with an Epworth Sleepiness Scale (ESS) score of 19. Physical examination revealed no abnormalities. On the basis of these findings, the patient was diagnosed with EDS accompanied by autonomic nervous dysfunction. After the patient agreed to receive treatment by providing informed consent, SGB was performed in the hospital. Specifically, the patient was placed in the supine position with her head deviated to one side, without the need for local anesthetic.
High-frequency ultrasonography was performed at the C6–C7 level, with the aim of detecting the longus colli muscle. Guided by the ultrasonographic images (Figure 1), 0.16% ropivacaine (5 mL) was infused below the prevertebral fascia and above the longus colli. SGB was performed once a day for 5 minutes, alternating between the left and right sides. Horner’s syndrome appeared after each injection, indicating the efficiency of the block. The patient remained in the hospital for a 30-minute observation period after each treatment; no hospitalization was required. She received eight sessions of SGB in total, during which oryzanol was continuously administered. After the first SGB, the weakness in both lower limbs resolved. The second SGB normalized the control of voiding and defecation, the third SGB markedly relieved the EDS, and the fifth SGB mitigated the burning sensation in the lower extremities and restored the patient’s normal eating behavior. The subsequent three sessions of SGB reduced the ESS score from 19 to 0. Throughout the treatment process, the patient reported no discomfort. Furthermore, no SGB-related complications occurred, such as infection and bleeding, and there was no damage to the nervous system. 10 After the final SGB, oryzanol administration was stopped. During the 3-month follow-up, no relapse of daytime sleepiness, weakness in the lower limbs, fecal incontinence, or urgent or frequent urination was reported.
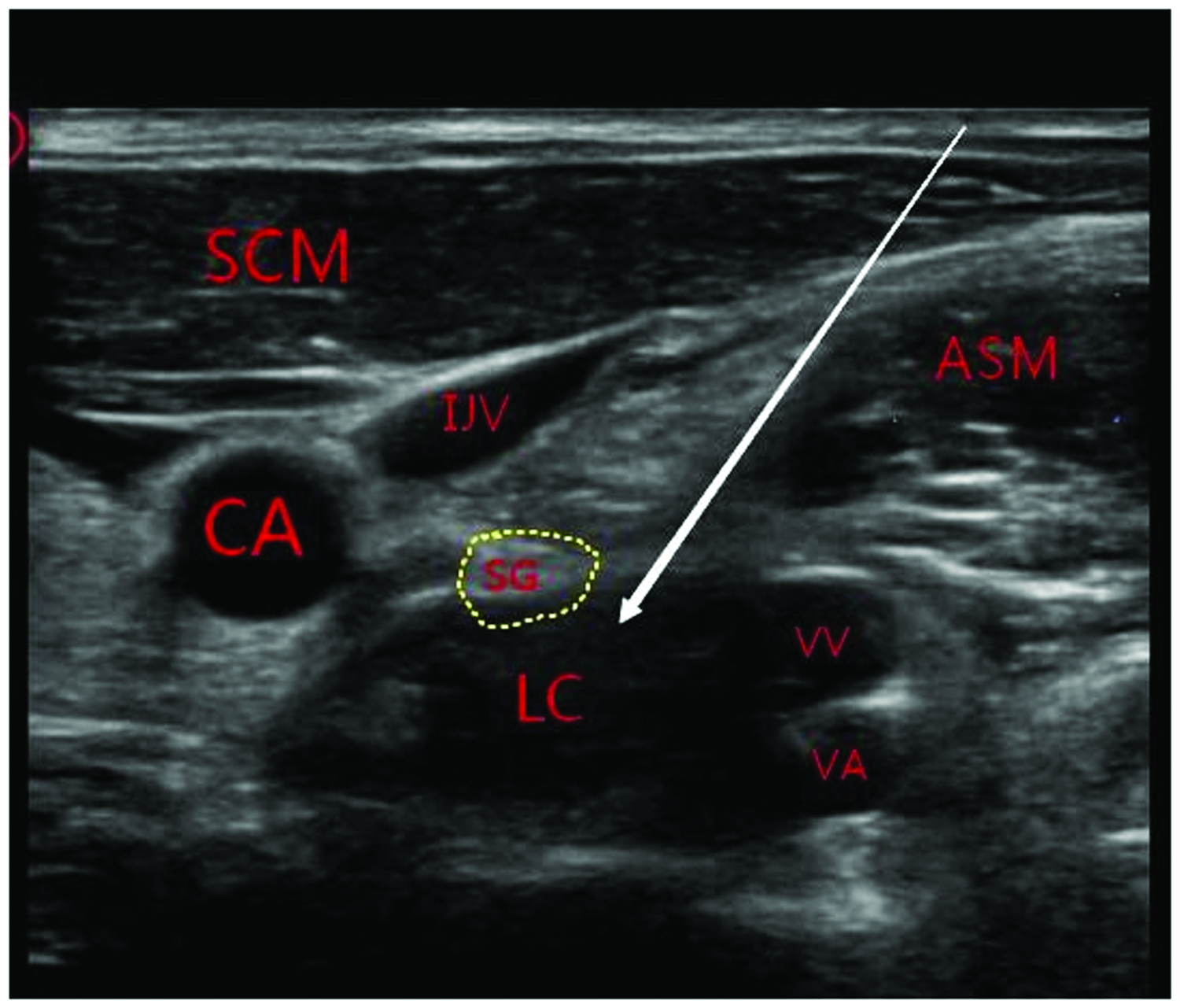
High-frequency ultrasonography of the neck (C6–C7 level), with the aim of detecting the longus colli (LC). The long arrow indicates the path of the injection needle.
Discussion
EDS leads to various adverse consequences, namely mind distraction, low working efficiency, and poor quality of life. The main causes are central nervous system (CNS) disorders, obstructive sleep apnea syndrome, metabolic diseases, 11 and depression. 12 Irregular sleepiness patterns; sleep fragmentation; 13 abnormal concentrations of γ-aminobutyric acid (GABA), growth hormone-releasing hormone (GHRH), and melatonin (MT); 14 and hypothalamic dysfunction may serve as underlying mechanisms. 15 In the present case, the patient reported no history of CNS-, respiratory-, metabolism-, or psychology-related abnormalities. The severity of the patient’s daytime sleepiness, graded by the ESS score, was evaluated independently by professional assessors or in sleep labs.
The reported sensitivity and specificity of ESS is as high as 93.5% and 100.0%, respectively, making this scoring method the currently accepted choice of patients and their families, clinically. 16 The initial ESS score of the present patient was 19, indicating a serious condition, and with her specific symptoms, the ESS score supported the diagnosis of EDS with autonomic nervous dysfunction. This condition might be correlated with abnormal function in the hypothalamus. 17 SGB has been used to adjust the activity of the hypothalamus by refining the local circulation. 18 This effect normalizes the homeostasis of the internal environment, stabilizes autonomic, endocrine, and immune functions, and facilitates the secretion of GHRH and MT. 19 As a result, SGB represents a potential treatment for sleep disorders. 20 In the present case, SGB resulted in a clear and desirable effect on the patient’s EDS and a decrease in the ESS score from 19 to 0. Additionally, the patient terminated the oryzanol when the somnolence and extreme fatigue were relieved after the final treatment. The 3-month follow-up revealed no relapse of any of the previous symptoms, namely daytime sleepiness, weakness in the lower limbs, fecal incontinence, or urgent or frequent urination, indicating that the SGB treatments recovered the patient’s autonomic activity and resolved the endocrine disorder. This result is similar to the results reported in other studies, suggesting the efficacy of SGB in stabilizing the autonomic system through reversing the autonomic imbalance. 21
Conclusion
There are currently few therapeutic approaches for EDS, and these have had undesirable outcomes. In the present case, SGB resulted in attractive effects regarding EDS recovery, providing a novel solution for severe functional complications related to EDS. In the future, clinical multicenter studies with a large sample size are warranted to further elucidate the therapeutic value of SGB in EDS and the mechanisms underlying its effects.
Footnotes
Author contributions
Study design: Yuenong Zhang.
Literature search: Tao Huang and Ruiqiong Zhong.
Writing of the original draft: Jian Xu.
Writing of the literature review and editing: Yuenong Zhang and Qingyan Liu.
Availability of data and materials
All the data supporting the findings in the study are included in the published manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics statement
This study was approved by the Ethics Committee and Institutional Review Board of The Third Affiliated Hospital of Sun Yat-sen University-Yuedong Hospital (2021–11). Written informed consent was obtained from the patient for the publication of this case report and the accompanying images.
