Abstract
Existing literature on primary carcinoma of the fallopian tube is limited because of the rarity of this disease. We report a patient with intermittent vaginal bleeding and vaginal discharge who underwent transvaginal ultrasound, magnetic resonance imaging, and 18-F-fluorodeoxyglucose positron emission tomography/computed tomography (18-F FDG PET/CT) in our hospital. Ultrasound showed a bilateral fallopian tube mass and a uterine lesion. Magnetic resonance imaging revealed typical sausage-shaped bilateral adnexal masses, but overlooked a small lesion in the uterus in the initial diagnosis. FDG PET/CT findings not only showed bilateral fallopian tube masses and uterine lesions, but also ruled out distant metastasis. Postoperative pathology confirmed bilateral primary high-grade serous adenocarcinoma of the fallopian tube with implants in the uterus. These findings suggest that 18-F FDG PET/CT imaging could be a good approach for the diagnosis and staging of primary carcinoma of the fallopian tube.
Keywords
Introduction
Primary carcinoma of the fallopian tube (PFTC) is rare and accounts for 0.14% to 1.8% of all gynecological cancers. 1 Bilateral PFTC and metastasis to the uterus are even rarer.2–3 The types of imaging routinely carried out for any suspicious gynecological cancers include ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI). We present a case of a 47-year-old woman with bilateral PFTC with metastasis to the uterus that was diagnosed not only using routine transvaginal ultrasound and MRI, but also using 18-F-fluorodeoxyglucose positron emission tomography/computed tomography (18-F FDG PET/CT). This diagnosis was finally confirmed by histopathological findings. To the best of our knowledge, there have been no reports in the literature on 18-F FDG PET/CT findings of bilateral PFTC and metastasis to the uterus.
Case report
A 47-year-old woman with a history of infertility presented to our hospital with intermittent vaginal bleeding and vaginal discharge in the past 2 months. There was no obvious abdominal pain and no fever during the course of the disease. One month previously, she had undergone diagnostic curettage in a local hospital because of vaginal bleeding and discharge. A preoperative examination and postoperative medical examination were unremarkable, and the patient reported that the medical examination was benign. The patient underwent transvaginal ultrasound and MRI in our hospital (Figure 1).

Magnetic resonance imaging shows bilateral adnexal sausage-shaped masses. The solid portions of the mass have T1 hypointensity (left adnexal mass: long arrow in (a), right adnexal mass: long arrow in (e)) and T2 hyperintensity (left adnexal mass: long arrow in (b), right adnexal mass: long arrow in (f)) and a high signal in diffusion-weighted imaging (left adnexal mass: long arrow in (c), right adnexal mass: long arrow in (g)). Magnetic resonance imaging also shows a focal abnormal signal in the uterine cavity (short arrow in (f) and (g)). Transverse-enhanced T1-weighted magnetic resonance image shows remarkable enhancement of these lesions (left adnexal mass: long arrow in (d), right adnexal mass: long arrow in (h), uterine cavity mass: short arrow in (h)). These findings suggest that a solid tumor and hydrosalpinx are the main components. However, in actual clinical practice, the uterine lesion was ignored by physicians in the initial diagnosis. Because the uterine lesion was small, it was only observed in one slice of the tomographic images. Transvaginal ultrasound shows bilateral cystic masses with spaces and mural nodules (long arrow in (i) and (j)), and a suspicious mass in the uterine cavity (short arrow in (k)).
At this time, the patient was diagnosed with bilateral adnexal masses and a uterine mass. The CA125 concentration was normal. To further confirm the diagnosis and understand the general situation, an 18-F FDG PET examination (Discovery STE16; GE) was then performed (Figure 2).
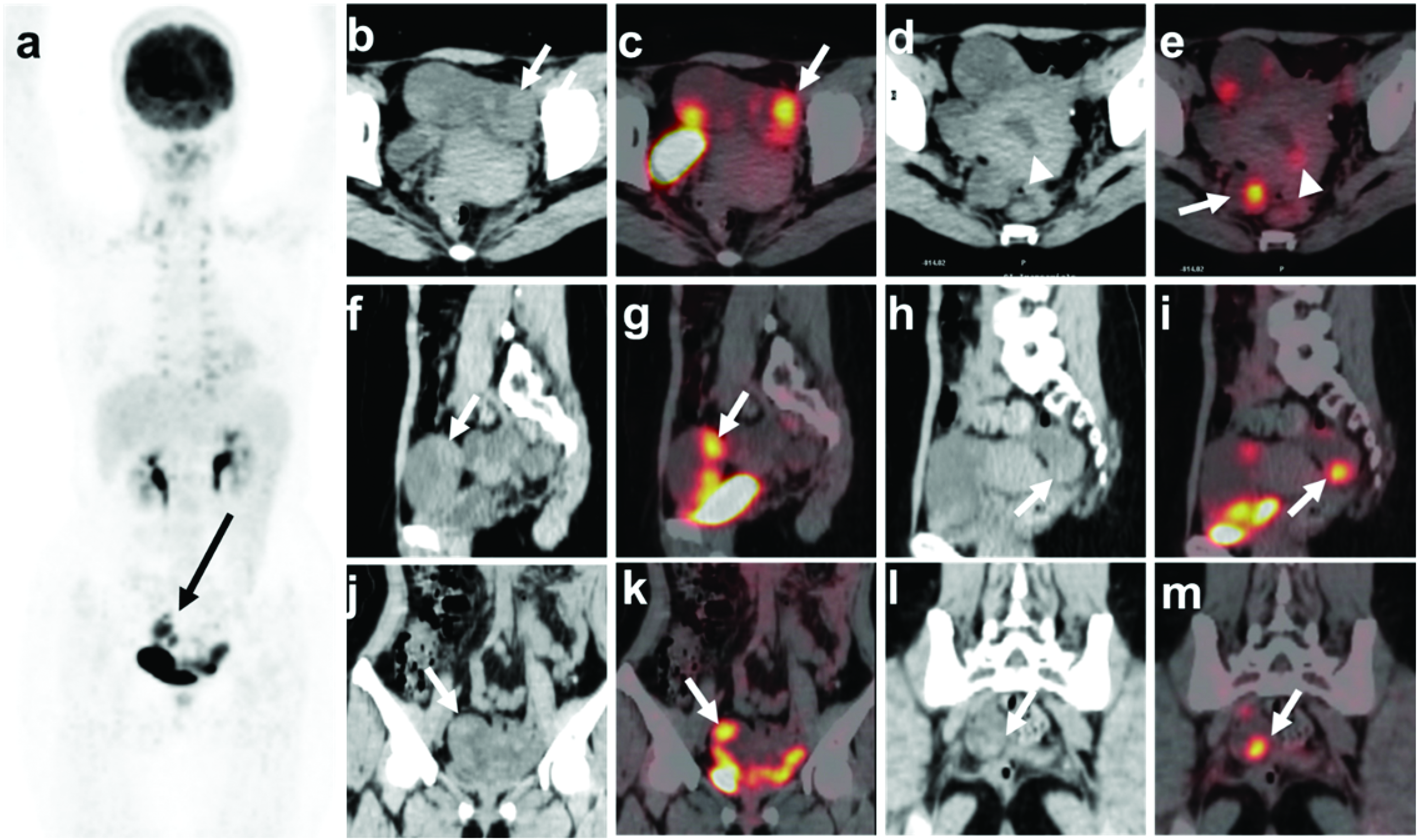
A combined positron emission tomography/computed tomography (PET/CT) skull base-to-mid-thigh scan was performed 60 minutes after intravenous administration of 6.31 mCi of 18-F fluorodeoxyglucose (FDG). CT shows a fluid-filled tubular adnexal structure, containing nodular or papillary solid components (long arrow; left adnexal mass: transverse [(b)], sagittal [(f)], and coronal [(j)] images; right adnexal mass: transverse [(d)], sagittal [(h)], and coronal [(l)] images). Uterine lesions are not clearly shown. The maximum long diameter and vertical diameter of the mass in the axial plane are approximately 43 × 97 mm (left) and 21 × 69 mm (right), respectively. Maximum intensity projection PET ((a)) and fused PET/CT images show nodular solid components of the masses presenting with intense FDG uptake, with a maximum standardized uptake value (SUVmax) of 10.91 (long arrow indicates the left adnexal mass in transverse [(c)], sagittal [(g)], and coronal [(k)] images) and 9.28 (long arrow indicates the right adnexal mass in transverse [(e)], sagittal [(i)], and coronal [(m)] images), respectively. PET/CT also shows focal intense FDG uptake with an SUVmax of 3.55 in the uterine cavity (short arrow in (e)).
Transvaginal ultrasonography, magnetic resonance imaging, and 18-F FDG PET/CT showed bilateral adnexal masses and a suspicious lesion in the uterus. However, the metastasis of fallopian tube cancer to the uterus is rare, and there have also been case reports of fallopian tube cancer combined with endometrial cancer. 4 Therefore, all three imaging studies failed to characterize whether the uterine lesion was metastatic or dualistic. Whole-body PET ruled out distant metastases and showed that the patient had the opportunity to undergo a comprehensive staging operation. Intraoperative rapid pathology suggested poorly differentiated cancer of the left fallopian tube. Therefore, a comprehensive staging operation for fallopian tube cancer was performed. Postoperative pathology confirmed bilateral primary high-grade serous adenocarcinoma of the fallopian tube and high-grade serous adenocarcinoma in the uterine cavity, which was ultimately considered metastatic. Immunohistochemical results showed the following: vimentin (−), estrogen receptor (40% moderately +), progesterone receptor (2% weakly +), p16 (+), p53 (100% strong +), paired box gene 8 (+), Wilms’ tumor 1 (+), Ki-67 (80%+), cytokeratin 7 (+), cytokeratin (+), inhibin-a (−), and calretinin (−) (Figure 3). These findings indicated a diagnosis of bilateral PFTC with stage IIA. 5

(a) Histology shows a multi-level papillary structure with stratified cells and a prominent solid growth pattern with cleft-like cavities. Shoe spike-like and localized clear cell and plastid-type calcifications, considerable necrosis and hemorrhage, and lymphovascular infiltration can be seen. The nuclei show moderate to severe atypia, mitotic figures are easily seen, and the cytoplasmic ratio is high (HE stain, original magnification, × 40) and (b and c) Tumor cells are positive for cytokeratin 7 and cytokeratin (original magnification, × 40).
The patient underwent one cycle of intraperitoneal chemotherapy (cisplatin 110 mg) and two cycles of intravenous chemotherapy (paclitaxel liposome 240 mg on day 1 + carboplatin 670 mg on day 2). Leukocytes were decreased (2.63 × 109/L) after chemotherapy and improved after symptomatic treatment. The patient will continue intravenous chemotherapy and regular follow-up.
Discussion
Previous literature on PFTC is limited because of the rarity of this disease. Together with primary ovarian and peritoneal carcinomas, PFTC is often grouped under the epithelial ovarian cancer (EOC) umbrella. 6 There are four major histological types of epithelial ovarian cancer, namely serous, endometrioid, mucinous, and clear cell carcinoma. The age group from 46 to 65 years old contains 65% of PFTC cases. Approximately 70% of patients with PFTC have pelvic inflammatory disease, and 49% complain of infertility according to the patients’ history and intraoperation revision data. 7 The incidence rate of PFTC is 0.36 to 0.41/100,000 women each year. However, from 2001 to 2014, the incidence of PFTC increased 4.19 fold. 8 This increase is likely multifactorial, such as a change in diagnostic practices, increased early detection, and improved pathological processing.
Despite an improvement in the diagnostic capacity of the contemporary methods of examination, a specific preoperative diagnosis is still challenging because of the silent course of PFTC. The usual clinical diagnosis is an ovarian tumor or pelvic inflammatory disease. 5 The most common symptoms and signs are abdominal pain or a pelvic mass, and vaginal bleeding or watery discharge. Preoperative diagnostic imaging routinely carried out for any suspicious gynecological cancers includes ultrasound, CT, and MRI.
Several previous studies9,10 have described the MRI findings of PFTC as a sausage-shaped adnexal mass or a multilocular cystic mass with a cog-and-wheel appearance, and the solid portion shows high intensity on diffusion-weighted images. There has even been a suggestion to include MRI in the International Federation of Gynecology and Obstetrics (FIGO) guideline for grading of malignancies in the female genital tract. A previous study showed that apparent diffusion coefficient values were significantly higher in women with FIGO grade 1 than in women with FIGO grade 3 (p < 0.0001, q = 16.591), and the Spearman correlation coefficient was 0.1012 between the mean apparent diffusion coefficient and FIGO grading. 9 However, in our case, the uterine lesion was ignored by physicians in the initial diagnosis because it was small, and it was only observed in one slice of tomographic images. According to the FIGO guideline, if there are uterine or ovarian metastases, the staging will change. Therefore, once fallopian tube cancer is suspected, the uterus and ovary should be carefully observed for suspicious lesions. There have been few relevant reports on the application of FDG PET in fallopian tube cancer.11,12 Potential advantages of FDG-PET/CT include its ability to detect distant metastases owing to its whole-body imaging properties and the detection of small lesions because of its high sensitivity. In this case, PET clearly showed a uterine lesion because of its high contrast (high target/non-target ratio). Studies have suggested a high specificity and moderate sensitivity for FDG-PET/CT to assess tumor resectability in EOC (including PFTC). 11 In our case, 18-F FDG PET/CT was a useful assessment tool for the staging of PFTC, including the detection of primary lesions and intrauterine metastases. In addition, many women in China, especially middle-aged and elderly women, have a history of intra-uterine device implantation. The findings in our case suggest that when MRI is not suitable, 18-F FDG PET/CT is an option to consider. A recent study showed that in ovarian cancer, 18-F FDG PET/MRI offers better sensitivity and specificity for the characterization and M staging than contrast-enhanced MRI and contrast-enhanced CT. 13 Additionally, the diagnostic value for T and N staging was equivalent for these two methods, which suggested that 18-F FDG PET/MRI is a useful diagnostic alternative to conventional imaging modalities. However, there were no cases of PFTC in this previous study. Whether PET/MRI has the same accuracy for diagnosing PFTC needs to be confirmed in future studies.
Among serological indicators, CA125 is the most commonly used to evaluate PFTC, although its diagnostic accuracy is controversial.14,15 CA125 is known as transmembrane glycoprotein or mucin 16 with a high molecular weight, and it is largely expressed in EOC. The median serum CA125 value in patients with PFTC preoperatively was reported to be 183 U/mL. 16 Mi and Zhang showed that serum human epididymis protein 4 (HE4) and CA125 concentrations were significantly higher in advanced patients than in controls with benign disease. 17 However, HE4 had higher specificity, but lower sensitivity, then CA125. Furthermore, serum HE4 concentrations were closely associated with the response of treatment and recurrence, and the effective response rate for therapy shown by HE4 was higher than that for CA125. 17
The optimal management of PFTC is still uncertain because it is rare. Treatment for PFTC is normally based on the same guidelines as those used for serous-type EOC. The mainstay of treatment is cytoreduction surgery and platinum-based chemotherapy. 6 Carboplatin and paclitaxel administered every 3 weeks is the standard-of-care, first-line chemotherapy for EOC. A clinical trial showed weekly dose-dense chemotherapy can be delivered successfully as the first-line treatment for EOC, but it did not significantly improve progression-free survival compared with standard 3-weekly chemotherapy in predominantly European populations. 18 The 5-year survival rate of PFTC is 47.4%, but the survival rate is as high as 92%, if the disease is caught in the early stages. 19 Fortunately, PFTCs are mostly found at an earlier stage than EOC because of the shorter history of symptoms of PFTC. The FIGO stage is a recognized prognostic factor of PFTC. Other possible prognostic factors remain controversial, such as the pathological type of tumor, the diameter of the residual tumor remaining after surgery, cycles of chemotherapy, tumor grade, preoperative CA125 concentrations, and the presence of ascites.20,21
Conclusions
PFTC is rare and its preoperative diagnosis remains challenging because of the silent course of this disease. The most common symptoms and signs of PFTC are abdominal pain or a pelvic mass, and vaginal bleeding or watery discharge. Among serological indicators, CA125 and HE4 are the most commonly used. Routine imaging methods for detecting PFTC include ultrasound, CT, and MRI. MRI findings of PFTC appear as a sausage-shaped adnexal mass or a multilocular cystic mass with a cog-and-wheel appearance. In the current case, FDG-PET/CT showed the following additional information. First, a small uterine lesion was clearly observed because of the high target/non-target ratio. Second, distant metastases were ruled out, which enabled the patient to undergo a comprehensive staging operation. Our findings suggest that 18-F FDG PET/CT is a good approach for the diagnosis and staging of PFTC. This is important because the FIGO stage is a recognized prognostic factor of PFTC. The mainstay of treatment for PFTC is cytoreduction surgery and platinum-based chemotherapy.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). The study was approved by ethics committee board of the First Affiliated Hospital of Soochow University of China. The patient provided written informed consent to publish the data in this study.
Funding
The present study was supported by the National Natural Science Foundation of China (grant no. 81601522), Medical Youth Talent Project of Jiangsu Province (grant no. QNRC2016749), Gusu Health Talent Program (grant no. GSWS2020013), Suzhou People’s Livelihood Science and Technology Project (grant no. SYS2019038), and Project of State Key Laboratory of Radiation Medicine and Protection, Soochow University (no. GZK1202127).
