Abstract
Objective
To investigate the effects of hand–foot syndrome (HFS) and fatigue on disease progression and survival in patients treated with sorafenib followed by regorafenib for advanced hepatocellular carcinoma.
Methods
A retrospective analysis of patients with advanced hepatocellular carcinoma treated with sorafenib in our hospital from 1 October 2018 to 31 October 2021 was performed, and clinical and pathological data and follow-up results were obtained. Patients were divided into groups according to the severity of HFS and fatigue. Survival analysis among the groups was performed using the Kaplan–Meier method, continuous variables were analyzed using the
Results
The study included 150 men and 23 women with a mean age of 60.77 years (range: 40–85 years). The median overall survival (OS), progression-free survival (PFS), and time to tumor progression (TTP) increased with increasing severity of HFS. Conversely, the median OS, PFS, and TTP decreased with increasing severity of fatigue
Conclusion
HFS and fatigue were independent risk factors affecting TTP, PFS, and OS among patients treated with sorafenib followed by regorafenib for advanced hepatocellular carcinoma.
Keywords
Introduction
Primary liver cancer is a common malignant tumor of the digestive system globally. According to global data released by GLOBOCAN 2018, liver cancer is the sixth most common malignant tumor (up to 841,000 cases per year) and second most common cause of cancer-related death (up to 782,000 deaths annually).1,2 Hepatocellular carcinoma (HCC) accounts for 75% to 85% of all liver cancers, and it is the fifth most common cancer in men (7.5% of all cancers) and ninth in women (3.4% of all cancers).3,4 HCC is the main cause of liver cancer-related burden and death.
At present, the treatment of HCC follows recognized guidelines.3,5 Treatment options are extremely limited for patients diagnosed with intermediate- or advanced-stage disease, but unfortunately, most patients are diagnosed with intermediate- or advanced-stage disease. According to the results of the Sharp study, the first-line systematic treatment for patients with advanced or metastatic liver cancer and Child–Pugh class A cirrhosis includes the oral multikinase inhibitor (MKI) sorafenib 400 mg twice a day. 6 According to a global open-label randomized phase III study (REFLECT), lenvatinib was the first new drug approved for advanced liver cancer in a first-line setting in more than 10 years. 7 Sorafenib and lenvatinib remain the most effective single-drug therapies.
Regorafenib (BAY 73-4506) is a novel MKI. The primary protein kinases inhibited by this drug include vascular endothelial growth factor receptors 1–3 (VEGFR1–3), TIE2, KIT, RET, RAF-1, BRAF, FGF, and platelet-derived growth factor receptor.8,9 Regorafenib can effectively block the activities of various protein kinases related to tumor angiogenesis, tumorigenesis, and the tumor microenvironment. The antitumor activity of regorafenib was confirmed in a series of clinical trials of advanced solid tumors, including HCC, gastrointestinal stromal tumor (GIST), and colorectal cancer (CRC).10–13 Some results also suggest that regorafenib exhibits anti-immunosuppressive properties and promotes anti-tumor activity. 14 Regorafenib enhances anti-tumor immunity by modulating macrophages and increasing the proliferation and activation of CD8+ T cells. 15 Because patients who tolerated sorafenib and recorded sorafenib progression had a survival advantage over those treated with placebo, regorafenib was the first drug approved for second-line treatment in HCC. 12 The average treatment durations of the regorafenib and placebo groups were 5.9 and 3.3 months, respectively, which were significantly different. 12
In randomized trials with approved indications (CRC, GIST, or HCC), regorafenib recipients experienced a large number of adverse events (AEs), including hepatotoxicity, infection, and bleeding. 16 A large number of studies revealed that liver function should be monitored before and during treatment with regorafenib, and hepatotoxicity should be managed by interrupting treatment, adjusting the dose, or terminating therapy according to its severity and persistence.
In the RESORCE trial, the incidence of any-grade treatment-related AEs (TRAEs) was approximately 2-fold higher in the regorafenib group than in the placebo group (93% vs. 52%). 12 Hypertension, hand–foot–skin reaction (HFSR), fatigue, and diarrhea are the most common grade 3 to 4 events. 6 The rates of grade 3 or 4 TRAEs in patients receiving regorafenib or placebo were 50% and 17%, respectively, with the most common events being HFSR (13% vs. 1%), hypertension (13% vs. 3%), bilirubinemia (7% vs. 2%), and fatigue (6% vs. 2%).
Although some studies demonstrated that fatigue and HFSR are related to the prognosis of patients receiving sorafenib, few reports have described the relationship between adverse reactions and the prognosis of patients with advanced HCC who received sorafenib combined with regorafenib. This study investigated the relationships of these AEs with the time to tumor progression (TTP) and overall survival (OS) to determine their utility as robust outcome predictors for patients who received sorafenib followed by regorafenib for advanced HCC.
Patients and methods
Patients
The clinical data and follow-up results of patients treated with sorafenib followed by regorafenib for advanced HCC in the Zhaoyuan branch of the Second Hospital of Shandong University (Linglong Yingcheng Hospital, Zhaoyuan, China) from 1 October 2018 to 31 October 2021 were analyzed retrospectively. Clinical data included sex, age, previous treatment (resection, ablation, transcatheter arterial chemoembolization [TACE], others), and adverse reactions (HFSR, diarrhea, fatigue), drug dose adjustment, hepatitis, cirrhosis, portal hypertension, alcoholism, diabetes, Child–Pugh classification, TNM staging, survival time and disease progression time from start of sorafenib treatment, and survival time and disease progression time from start of regorafenib treatment. All patients were diagnosed with HCC
The inclusion criteria of this study were as follows: confirmation of HCC by imaging or surgical resection and the receipt of sorafenib for more than 3 months and regorafenib for more than 1 month. Meanwhile, patients with Child–Pugh grade C cirrhosis and those with Eastern Cooperative Oncology Group performance status ≥3 before treatment were excluded.
Sorafenib and regorafenib administration
The initial dose of sorafenib was 800 mg/day delivered in two divided doses. The initial dose of regorafenib was 160 mg once daily for the first 21 days of each 28-day cycle. AEs caused by sorafenib and regorafenib were recorded, and the criteria were based on the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) v5.0. 16 AEs were examined on the 10th day and 1 month after drug administration. For each grade ≥ 3 AE, dose adjustment or temporary treatment interruption was performed, and the duration of withdrawal did not exceed 7 days.
Follow-up
Patients administered sorafenib and regorafenib were followed up once a month in the clinic. Liver function and alpha-fetoprotein levels were reviewed monthly. CT was performed once every 2 months, and enhanced CT, enhanced MRI, or related examinations (such as radionuclide bone scan or positron emission tomography–CT) were performed when disease progression was suspected. TTP was calculated by referencing the date of imaging diagnosis.
Statistical analyses
Univariate analysis was used to identify predictors of survival using the Kaplan–Meier method, and comparisons were performed using the log-rank test.
Results
Baseline characteristics
Baseline data are presented in Table 1. The study included 150 men and 23 women with a mean age of 60.77 years (range: 40–85 years). The cohort included 143 cases of viral hepatitis B. Regarding the treatment history before study enrollment, 85 patients underwent hepatectomy, 3 patients underwent liver transplantation, and 73 patients underwent TACE. The cohort included 164 patients with Child–Pugh grade A and 9 patients with Child–Pugh grade B. All patients in this study received only sorafenib + regorafenib.
Baseline data.

HBsAg, hepatitis B virus surface antigen; TACE, transcatheter arterial chemoembolization; ALB, albumin; Tbil, total bilirubin; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; AFP, alpha-fetoprotein; ECOG PS, Eastern Cooperative Oncology Group performance status.
The survival factors of the patients in this group were analyzed. Based on the results of multivariate analysis presented in Table 2, fatigue and hand–foot syndrome (HFS) were independent risk factors for the outcomes of patients with HCC.
Factors influencing survival after regorafenib therapy.
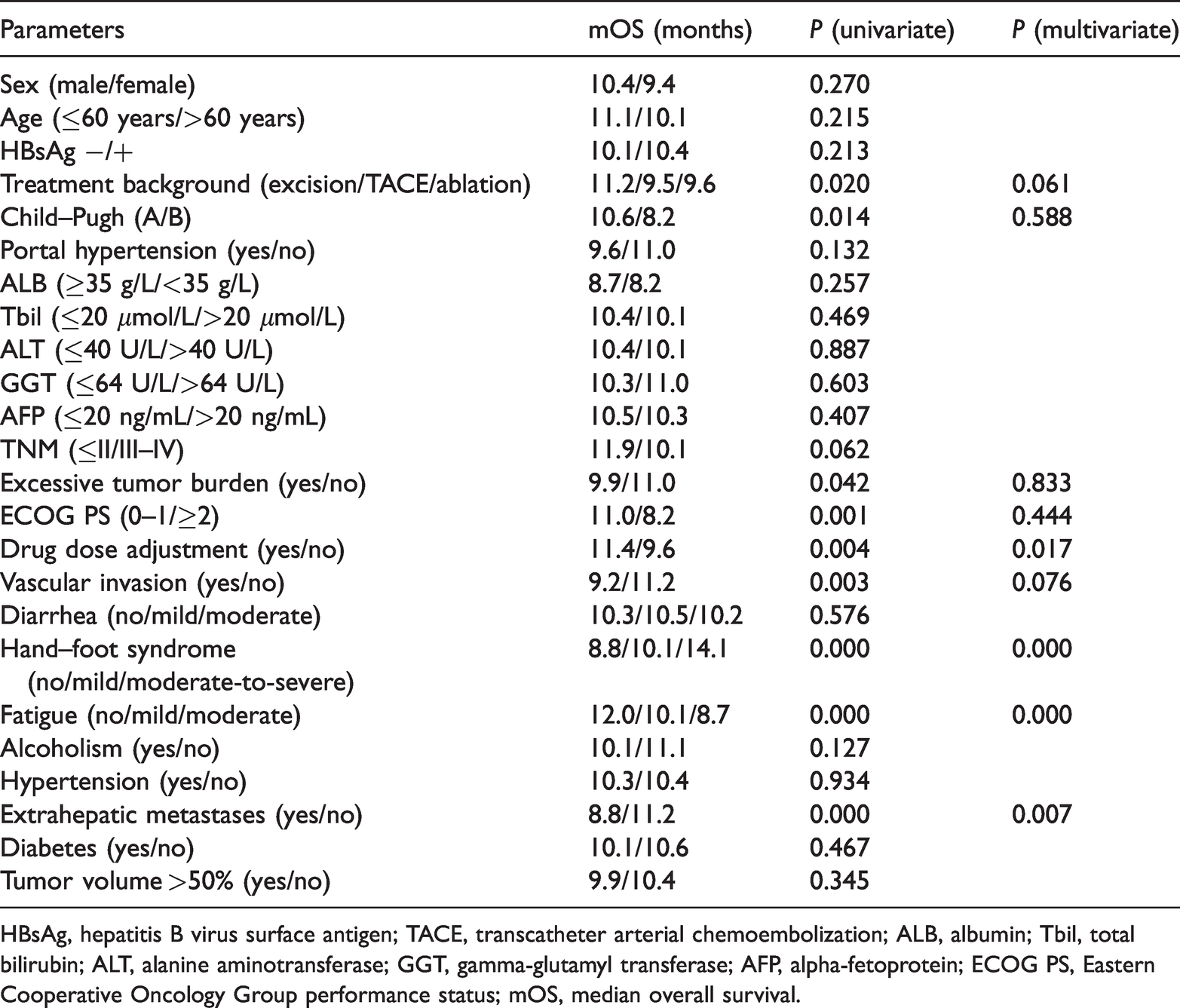
HBsAg, hepatitis B virus surface antigen; TACE, transcatheter arterial chemoembolization; ALB, albumin; Tbil, total bilirubin; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; AFP, alpha-fetoprotein; ECOG PS, Eastern Cooperative Oncology Group performance status; mOS, median overall survival.
Similarly, factors related to TTP and PFS were analyzed. As presented in Tables 3 and 4, fatigue and HFS were independent risk factors for TTP and PFS.
Factors influencing mTTP after regorafenib therapy.
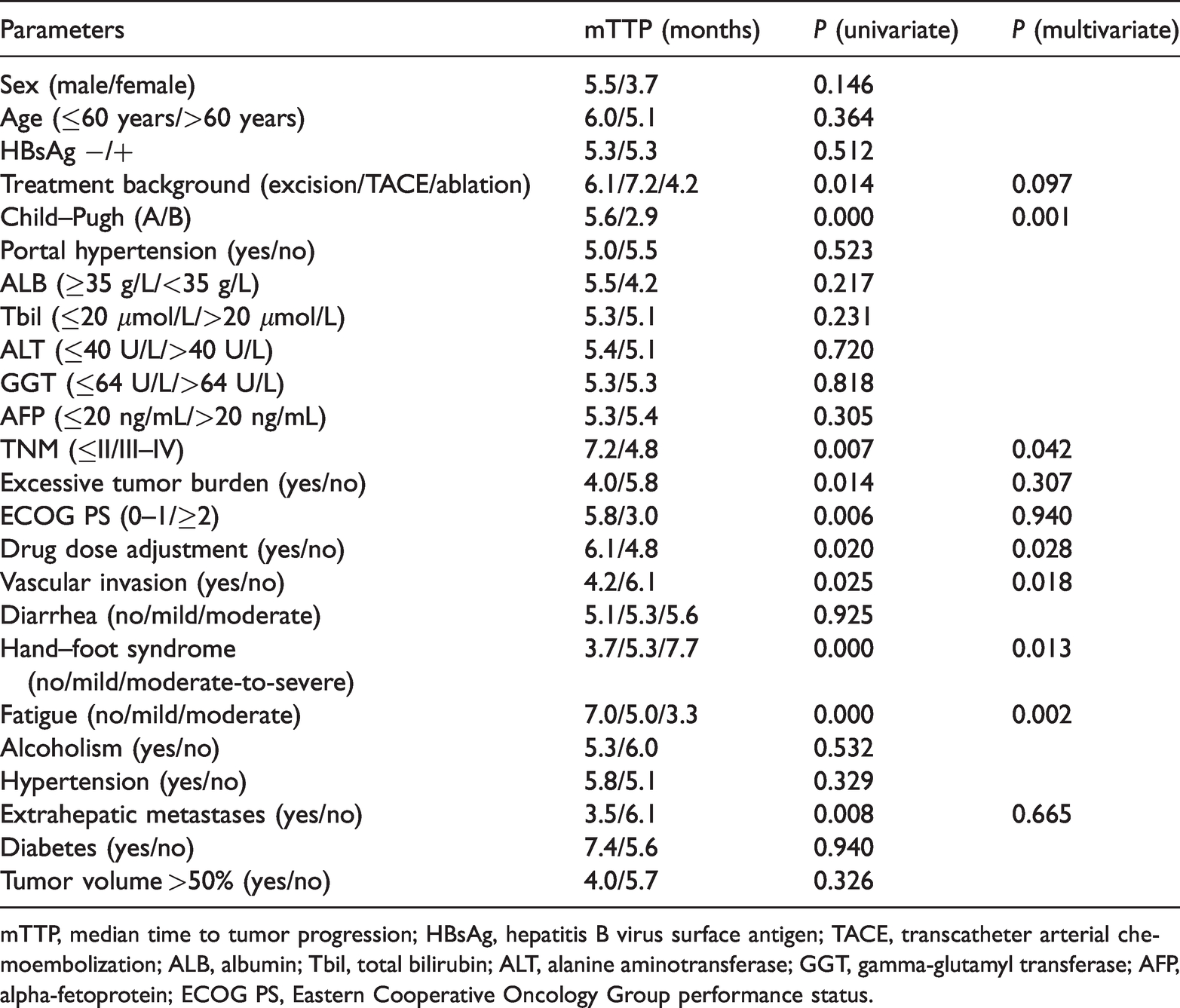
mTTP, median time to tumor progression; HBsAg, hepatitis B virus surface antigen; TACE, transcatheter arterial chemoembolization; ALB, albumin; Tbil, total bilirubin; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; AFP, alpha-fetoprotein; ECOG PS, Eastern Cooperative Oncology Group performance status.
Factors influencing mPFS after regorafenib therapy.
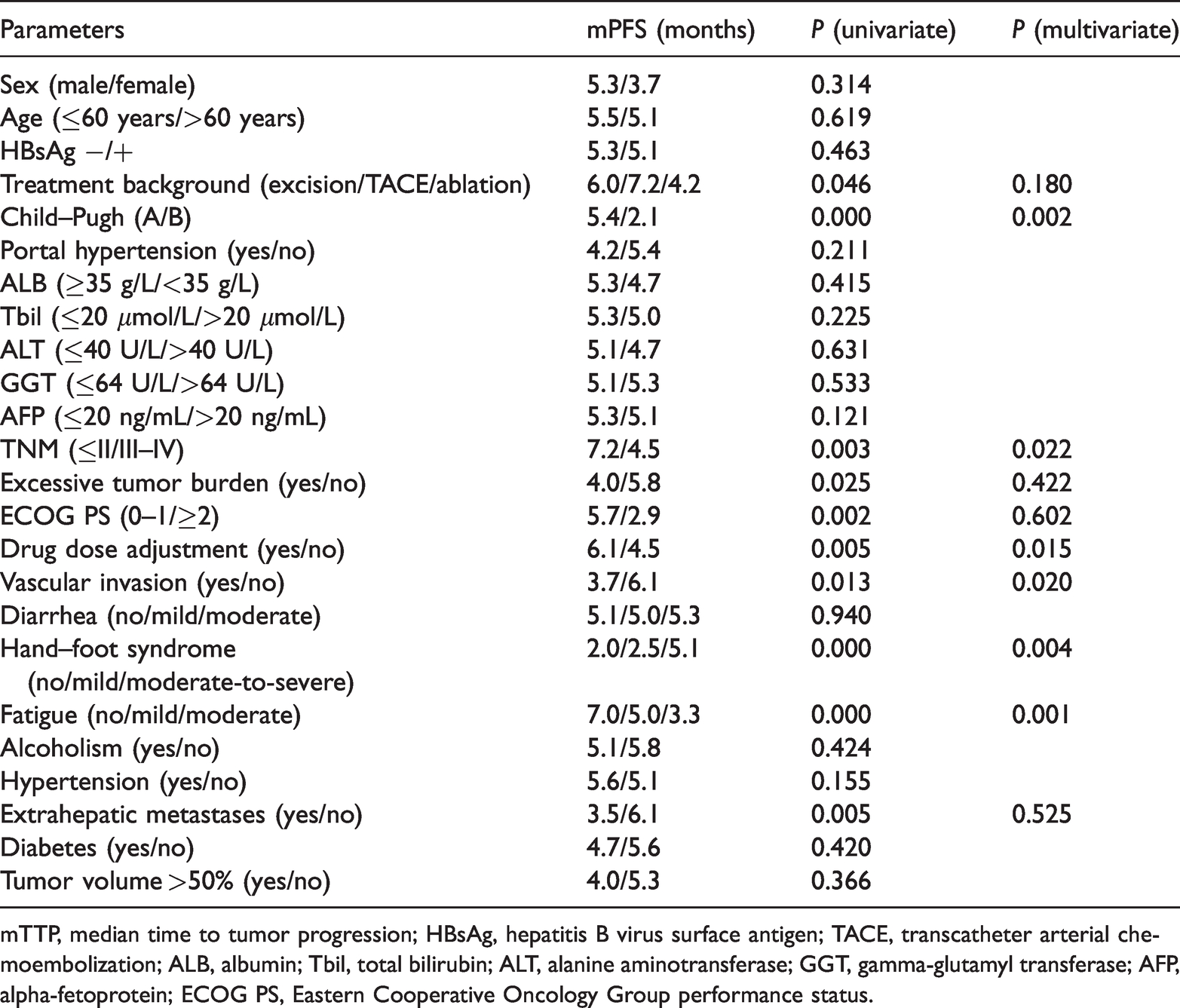
mTTP, median time to tumor progression; HBsAg, hepatitis B virus surface antigen; TACE, transcatheter arterial chemoembolization; ALB, albumin; Tbil, total bilirubin; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; AFP, alpha-fetoprotein; ECOG PS, Eastern Cooperative Oncology Group performance status.
Survival analysis
OS, TTP, and PFS analysis were analyzed by the Kaplan–Meier method. Median OS of this group of patients was 10.3 months (95% confidence interval [CI] = 9.67–10.94), median TTP was 5.3 months (95% CI = 4.66–5.94), and median PFS was 5.1 months (95% CI = 4.39–5.81; Tables 2–4). HFS and fatigue were independent risk factors for OS, TTP, and PFS among patients treated with sorafenib followed by regorafenib for advanced HCC.
HFS
We divided patients with HFS into grades I (n = 63), II (n = 60), and III (n = 50) according to NCI CTCAE v5.0 (Figure 1, Table 5). Among all patients, those without hand foot syndrome were classified into grade I.

Kaplan–Meier analysis of survival curves of the associations of (a) OS, (b) TTP, and (c) PFS with hand–foot syndrome in patients treated with sorafenib.
Hand–foot syndrome grading.

ADL, activities of daily living.
The median OS times for grades I, II, and III were 8.8, 10.1, and 14.1 months, respectively. OS significantly differed between grades I and III (χ2 = 43.284;
The median TTPs from the start of regorafenib treatment for patients in grades I, II, and III were 3.7, 5.3, and 7.7 months, respectively. TTP did not differ between grades I and II (χ2 = 2.898;
The median PFS times from the start of regorafenib treatment for patients in grades I, II, and III were 3.5, 5.0, and 7.7 months, respectively. PFS significantly differed between grades I and III (χ2 = 17.064;
Fatigue
To better study the effects of fatigue on this group of patients, we divided patients into grades I (n = 65), II (n = 63), and III (n = 45) according to NCI CTCAE v5.0 (Figure 2, Table 6).

Kaplan–Meier analysis of survival curves of the associations of (a) OS, (b) TTP, and (c) PFS with fatigue in patients treated with sorafenib.
Fatigue grading.

ADL, activities of daily living.
The median OS times from the start of regorafenib treatment for grades I, II, and III fatigue were 12.0, 10.1, and 8.7 months, respectively. OS significantly differed between grades I and III (χ2 = 33.195;
The median TTPs from the start of regorafenib treatment for grades I, II, and III were 7.0, 5.0, and 3.3 months, respectively. TTP significantly differed between grades I and II (χ2 = 6.271;
The median PFS times from the start of regorafenib treatment for grades I, II, and III were 7.0, 5.0, and 3.3 months, respectively. PFS significantly differed between grades I and III (χ2 = 28.176;
Discussion
Compared with sorafenib, regorafenib has greater inhibitory effects on angiogenesis-related receptors, mainly because although their structures are similar, the fluorine on the benzene ring is different. 9 Regorafenib targets a wide range of angiogenic factors, tumor microenvironment factors, and oncogenic kinases based on its ability to inhibit VEGFR1, VEGFR3, RAF, TIE2, KIT, RET, and BRAF.17,18
In the seminal phase III trials of regorafenib, most patients experienced AEs. The incidence of grade ≥ 3 AEs ranged from 45% to 75%. The most common AEs included HFS, fatigue, diarrhea, nausea, vomiting, decreased appetite, hypertension, and weight loss.
19
In each of these studies, a large proportion of patients experienced AE-related dose reduction or treatment interruption. AE-related dose reduction or treatment interruption was reported in 68% of patients treated with regorafenib (vs. 31% in the placebo arm).
12
Across all of these clinical trials, regorafenib displayed a consistent and predictable AE profile, with skin toxicities such as HFS being among the most clinically significant.20–22 Dermatological reactions are extremely common AEs of MKIs, especially regorafenib. In general, dermatologic AEs are not fatal, but they can cause both physical and emotional discomfort. Prompt recognition and treatment may alleviate symptoms and improve quality of life.
23
Belum
21
In this study, OS in patients who received regorafenib was 22.0 months (95% CI = 20.9–23.1). In this clinical trial, which was consistent with previous literature on sorafenib treatment, the prognosis of patients with vascular invasion was extremely poor. After grouping patients according to their HFS severity, the Kaplan–Meier method (log-rank test) was used, followed by Cox analysis. The median OS times for grades I, II, and III were 8.8, 10.1, and 14.1 months, respectively. This demonstrated that within a certain range, OS increased with increasing severity of HFS. Similar results were observed for TTP and PFS.
Fatigue is defined by CTCAE v5.0 as “a disorder characterized by a state of generalized weakness with a pronounced inability to summon sufficient energy to accomplish daily activities.” Fatigue can have a significant adverse impact on patients’ quality of life. It may be related to other symptoms such as sleep disorders, pain, and depression. Fatigue is also a common toxicity related to cancer treatment. 30 The early detection of fatigue symptoms is helpful for continued patient treatment. To ensure that patients understand when they are fatigued, they should be informed of the symptoms they might experience. These symptoms include eye or leg fatigue, general fatigue, shoulder stiffness, decreased or no energy, an inability to concentrate, weakness or discomfort, boredom or a lack of motivation, drowsiness, irritability, tension, and anxiety or impatience. Fatigue should be evaluated weekly during the first two cycles of regorafenib therapy, and follow-up evaluations can be performed every 2 weeks. 23 The methods of intervention and treatment for fatigue are divided into drug and non-drug treatments. Diseases such as anemia should be initially corrected in patients who complain of fatigue. At the same time, patients should be informed of the need for active non-drug treatment, including, nutritional support, improved sleep hygiene, and stress management. 31 When guiding patients to customize treatment plans, we must refer to patient fatigue-related indicators to reduce the risk of fatigue-related dose reduction or treatment interruption. Thus, once the fatigue risk factors are determined, the most vulnerable patients require early treatment. 32
In this study, after grouping patients according to their fatigue level, Kaplan–Meier analysis (log-rank test) was performed, followed by Cox analysis. The median OS times from the start of regorafenib therapy for grades I, II, and III were 12.0, 10.1, and 8.7 months, respectively, illustrating that the duration of survival decreased as the severity of fatigue increased. Similar results were observed for TTP and PFS. It is noteworthy that the prognostic role of adverse events in patients who underwent systemic treatment is of major clinical importance for improving the treatment strategy of HCC because prognostic scores based on laboratory parameters may be suboptimal, as recently demonstrated. 33
Our study had several limitations. First, its single-center nature might limit its representativeness; however, because our team had similar management experience, quality control was guaranteed. Second, it is undeniable that retrospective analysis might introduce some bias, but our team has improved the quality of the data as much as possible by prospectively collecting records. Finally, our cohort was relatively homogeneous in terms of race and etiology. Therefore, further research is needed to validate the current results.
Conclusion
Based on a study of 173 patients, HFS and fatigue are independent risk factors affecting TTP, PFS, and OS among patients treated with sorafenib followed by regorafenib for advanced HCC.
Footnotes
Acknowledgement
This work was supported by Qingdao Special Servicemen Recuperation Center of PLANAVY, Shandong Province, People’s Republic of China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Linglong Yingcheng Hospital Youth Foundation (2021QN001).
Declaration of conflicting interest
The authors report no conflicts of interest in this work.
Author contributions
Ning Li and Xuan Qiu collected patients’ data. Ning Li analyzed the data and wrote the manuscript. Ning Li and Xuan Qiu conducted this study and revised the manuscript. All authors participated in patient treatment. All authors made substantial contributions to study conception and design; data acquisition, analysis, and/or interpretation; and manuscript writing and/or critical revision for important intellectual content. All authors gave final approval of the version to be published, and they agreed to be accountable for all aspects of the work.
