Abstract
Objective
The role of ABO types and RhD antigen in coronavirus disease 2019 (COVID-19) severity has been investigated in several recent studies. Thus, the objective of this study was to identify the relationship of ABO and RhD types with symptomatic COVID-19 disease and determine the groups associated with an increased risk of hospitalization.
Methods
This observational case-control study was performed in 530 Iraqi-Kurdish patients with COVID-19. Among them, 184 were severe cases that required hospitalization, while 346 were mild to moderate cases that were treated at home. ABO and RhD antigen groups were compared between cases and 1698 control records from 1 year before the pandemic. The diagnosis of COVID-19 was based on real-time polymerase chain reaction tests and high-resolution chest computed tomography scans with the typical clinical presentation.
Results
There were no significant differences in ABO and RhD antigen distributions between the COVID-19 cases and non-COVID controls. No ABO group was associated with the risk of hospitalization as a marker of the severity of infection.
Conclusions
There was no significant association between symptomatic COVID-19 disease and any ABO group or RhD antigen type. No impact of ABO groups on hospitalization was documented.
Keywords
Introduction
The coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) is a rapidly spreading infection regarded by the World Health Organization as a pandemic.1,2 SARS-CoV2 is an enclosed RNA virus that belongs to the family Coronaviridae and is frequently spherical on electron microscopy imaging. 3
Certain ABO blood groups have been reported as risk factors for COVID-19 disease in several recent studies. The relationship of ABO types with the severity of COVID-19 symptoms, risk of hospitalization, and ventilator support is a subject for research.4,5 Several reports have suggested that ABO blood groups play an important role in cardiovascular, oncological, and other diseases, especially their susceptibility, development, and progression. 6 The antigens of the ABO blood group system (A, B, and H determinants) are complex carbohydrate molecules on the extracellular surface of red blood cell membranes. These antigens have been identified as key ligands and receptors for parasites, bacteria, immunologically important proteins, and viruses. 7
ABO blood groups are correlated with various diseases, such as peptic ulcers, malignancies, blood and circulatory diseases, cognitive disorders, microbial infections, and metabolic diseases. 7 Some researchers have recommended that the ABO blood group antigens be termed ABH histo-blood group antigens to highlight that they are primarily tissue antigens. 8
A previous study reported that susceptibility to SARS-CoV2 was related to antigenic elements of ABO blood groups. In particular, the proportion of infected patients with blood group A was significantly higher than that for blood group O. 9 Additionally, in a population-based cohort study, it was determined that the O blood group may be associated with a slightly lower risk for SARS-CoV-2 infection and severe COVID-19 illness. 10 However, Boudin et al. confirmed that ABO blood groups are not associated with a higher risk of SARS-CoV-2 infection in young adults. 11 In addition, Almadhi et al. indicated that variations in blood groups might not be an ideal biomarker to predict the risk of COVID-19 disease. 12
Given that ABO blood groups have been regarded as risk factors in several recent studies, 4 the current study aimed to identify the relationships of ABO and RhD antigen types with COVID-19 disease and determine the high-risk blood groups that are associated with the need for hospitalization and oxygen (O2) support in a Kurdish population from Sulaymaniyah city, Iraqi Kurdistan Region. Moreover, this study aimed to shed light on demographic data, case presentation, and COVID-19 complications.
Patients and methods
Study setting and data collection
This observational case-control study followed the Equator guidelines. 13 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14
The study was planned to assess the ABO blood groups and RhD antigen types in Iraqi-Kurdish patients infected with COVID-19. The test was based on the agglutination principle using the tile method. Severe or critical cases were those that required hospitalization, and mild to moderate cases included patients who were treated at home without O2 supplementation. The data were collected from Qala (at Kalar province), Shaheed Aso, and Shaheed Hemn Hospitals for Corona in Sulaimaniyah city, Iraqi Kurdistan Region, from August 2020 to November 2020 (the time of the original strain of the COVID-19 pandemic).
For the ABO control group, we used a large series of patients or healthy participants who attended the Central Laboratory, Sulaimaniyah city, Iraqi Kurdistan Region during 2019 before the COVID-19 pandemic.
Patient diagnostic strategy
Patients with COVID-19 who were enrolled in this study were diagnosed using the following techniques.
1. Real-time polymerase chain reaction (RT-PCR) test
This test established using the first sequences of SARS-CoV-2 and the GISAID database probes and primers (nCoV_IP2 and nCoV_IP4) was intended to target the RdRp gene surrounding nucleotides 12621 to 12727 and 14010 to 14116 (positions rendering SARS-CoV, NC_004718). 15
2. High-resolution chest computed tomography (HRCT)
HRCT is a scanning protocol in which thin sections (usually 0.625–1.25 mm) are acquired and reconstructed using a sharp algorithm (e.g., bone algorithm). Patients underwent non-contrast HRCT. 16 Therefore, the diagnosis of COVID-19 pneumonia was considered based on a report from a qualified radiologist together with the satisfaction of the treating physician with HRCT findings, particularly ground-glass opacities.17 –19
3. Typical clinical presentation plus history of close contact with patients with COVID-19 confirmed by RT-PCR
Patients were asked about their age, sex, first presenting symptom(s), duration of illness (the time from the onset of symptoms to complete remission or clinical stabilization), history of close contact (contact with patients(s) with confirmed COVID-19 within less than 6 feet for >15 minutes within 24 hours), and incubation period (the time from the contact to the onset of the first symptom(s) of the illness) using a well-designed questionnaire prepared by our team (Supplementary 1). Additionally, body mass index (BMI) was measured for each patient. Furthermore, patient histories of any risk factors, such as hypertension; diabetes; smoking; cardiovascular, renal, hepatobiliary, and gastrointestinal tract disease; neurological, hematological, and malignant conditions; or immunosuppressive medications, were also taken. 20 All patients were contacted by phone when there was any missed information in their case sheets.
Patient consent and ethical approval
The study was completed following the declaration of Helsinki. The aims of the study were explained to those who agreed to participate and provided their written informed consent. Control participants did not provide consent because their data were already available and used for reporting. The study was approved by the Ethical Committee of the College of Medicine, University of Sulaimani, Sulaimaniyah, Kurdistan Region of Iraq, with the approval number MED-UoS-05-2020.
Patient categories
Patients were subdivided into the following two severity groups according to the need for hospitalization or respiratory support.
For the severity grouping of mild to moderate or severe to critical cases and indication for hospitalization, we followed the USA National Institute of Health Guidelines.21 –23
Statistical analysis
The data were analyzed using IBM SPSS Statistics for Windows, Version 25 (IBM Corp., Armonk, NY, USA). Descriptive statistics were presented as the mean ± standard deviation, and frequencies were expressed as percentages. The chi-square test for categorical variables and student’s t-test were used to compare the significant difference between the two means. The adjusted odds ratio and confidence intervals were estimated using logistic regression. The level of significance (p-value) was set at ≤0.05.
Results
This study enrolled 530 COVID-19 cases and 1698 non-COVID-19 controls. Their ABO blood group and RhD antigen records were used for data analysis. Among cases, 184 patients were severe or critical cases that required hospitalization, and 346 were mild to moderate cases that were treated at home without O2 supplementation. The ABO control group included a large series of patients (n = 1698) or healthy participants who attended the Central Laboratory, Sulaimaniyah city, Iraqi Kurdistan Region, during 2019 before the COVID-19 pandemic.
ABO and RhD group comparisons between the controls and patients with COVID-19
The distribution of ABO blood groups in patients with COVID-19 compared with that in pre-pandemic control cases did not show any significant difference. In addition, the difference in the total numbers of RhD antigen-positive or RhD antigen-negative cases between patients with COVID-19 and controls was not significant (Table 1).
ABO type and RhD antigen distribution between non-COVID-19 controls and symptomatic patients with COVID-19.

aChi-square test.
COVID-19, coronavirus disease 2019.
ABO and RhD group comparisons between hospitalized and non-hospitalized patients with COVID-19
There were no significant differences among ABO and RhD groups regarding the severity of COVID-19 infection in terms of the need for hospitalization and O2 supplementation. In addition, odds ratios and confidence intervals estimated by logistic regression analysis did not show any significant difference in the rate of hospitalization between the ABO blood groups (Table 2).
The association of ABO and RhD types with the severity of COVID-19 in hospitalized and non-hospitalized symptomatic patients with COVID-19.

aChi-square test, bestimated by logistic regression.
COVID-19, coronavirus disease 2019; OR, odds ratio; CI, confidence interval.
Demographic, clinical, and prognostic factors
The mean age of patients was 43.58 ± 15.91 years, and men were infected more than women (296 vs 234 patients, respectively). The mean age of hospitalized patients was significantly higher (p < 0.0001) than that of non-hospitalized patients. Women were admitted more than men (p < 0.002). The duration of COVID-19 illness, need for plasma transfusion, and mortality rate were significantly higher in hospitalized patients with COVID-19 than in non-hospitalized patients (p < 0.0001) (Table 3).
Comparison of socio-demographic, clinical, and prognostic factors between hospitalized and non-hospitalized symptomatic patients with COVID-19.

aStudent t-tests, bChi-square test, p ≤ 0.05 regarded as significant. 1Close contact with confirmed COVID-19 case, 2any feature that persists after the acute phase and clinical stabilization.
COVID-19, coronavirus disease 2019.
Effect of sex on some parameters
The mean age of male patients with COVID-19 was significantly higher than that for female patients (p = 0.005), and the duration of illness in men was prolonged (p = 0.05) compared with that in women. However, there were no significant differences between the two sexes regarding incubation period, BMI, or mortality (Table 4).
Impacts of sex on parameters.

aStudent t-tests, bChi-square test, p ≤ 0.05 regarded as significant.
BMI, body mass index.
Percentages of 1st symptoms in patients with COVID-19
For 37.2% of patients with COVID-19, the initial presentation consisted of more than one symptom. For mono-symptomatic cases, myalgia was the most common documented symptom (28%) as the first (initial) presentation. In contrast, dry cough, malaise, shortness of breath, and fatigue were the least mono-symptomatic presentations (Figure 1).
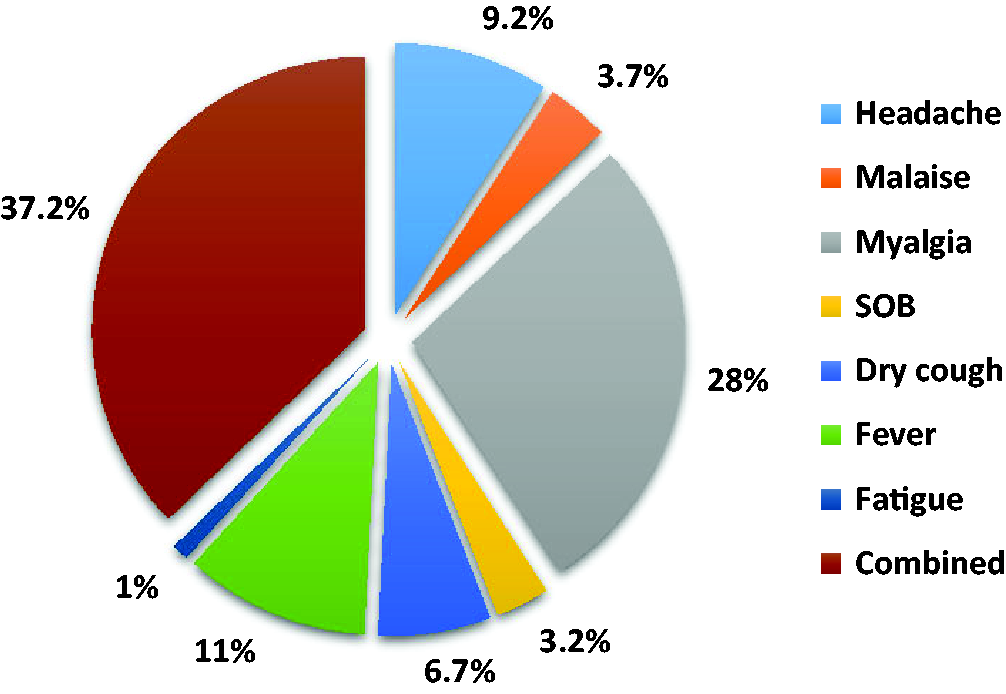
The distribution of the first symptom among patients with COVID-19.
Discussion
The frequencies of ABO types in our studied cases and controls followed the pattern of O > A > B > AB, with RhD antigen positivity in 92.40% of the controls and 92.07% of the patients. In agreement with our findings regarding the natural distribution of ABO, the same pattern was found in another study in a large Kurdish population (n = 53,234) by Jaff. 24 Furthermore, a similar pattern was documented in another two Iraqi studies conducted by Saleh and Abood 25 and Alubadi 26 in Baghdad. In all of these studies, RhD antigen positivity was found in approximately >90% of blood samples.24 –26
In the current study, we did not observe any positive association between symptomatic COVID-19 infection and ABO or RhD antigen groups, and no impact of ABO grouping on hospitalization (as a marker of the severity of infection) was documented. Consistent with our results, Ishaq et al. did not demonstrate any significant association of ABO groups with infection severity, associated death, or acute phase reactants in a retrospective cohort study of 1067 patients with COVID-19. 27 Moreover, in a multi-institutional study of 1289 patients with COVID-19, Latz et al. did not find any association between blood types and the risk of severe COVID-19 progression, intubation, or death or inflammatory marker elevation. Their observation of more positive COVID-19 tests in groups B and AB and less COVID-19 positive results in patients with RhD antigens and blood type O might be attributed to the lack of a comparative control. 28 Additionally, Wu et al. reported no significant correlation between ABO groups and COVID-19 infection severity and death. They validated group A as a high-risk group and group O as a low-risk group for COVID-19 infection. 29
Moreover, Almadhi et al. did not find any association between ABO blood groups and infection severity in terms of requiring intensive care unit admission. Although they found a higher risk of infection susceptibility in group B, exposure or severity was not associated with antibodies related to any blood group. 10 Additionally, Kabrah et al. performed a meta-analysis of 16 multinational studies and concluded that those with group A were more vulnerable to COVID-19 infection, and group AB individuals showed the lowest risk of COVID-19 disease. 30 In an observational clinico-hematological, radiological, and complication analysis of susceptibility to COVID-19 infection in Saudi Arabia, Kabrah et al. reported that symptoms were highly observed in blood group O, followed by A and B except for pharyngeal exudate in blood group A. 31 More recently, in a Spanish population-based prospective cohort study, Enguita-German et al. observed no association between ABO blood types and hospitalization, intensive care unit admission, or death in individuals with COVID-19. Regarding the risk of COVID-19 infection, they reported a protective effect in group O and a greater risk in A group. 32
Contradictory to our work, Ad’hiah et al. in Iraq studied the ABO blood group association with COVID-19 susceptibility in hospitalized patients. They found that group A was most common among hospitalized patients with COVID-19, whereas group O was the most common among blood donor controls. 33
The heterogeneity of the results among different studies might be due to the sampling size variations, lack of comparison with a suitable control group, presence of other confounders that might not be adequately adjusted by the authors, study of symptomatic together with asymptomatic infected individuals, genetic variations related to the geographical location, and the ethnicity of different populations.
In the present study, male patients with COVID-19 were older than female patients and showed a highly significant difference in illness duration, with a considerably higher mortality rate. This observation might be attributed to the fact that during the COVID-2019 pandemic, much emphasis was initially placed on the elderly, and now it is clear that being male is a risk factor. The epidemiological outcomes described across different parts of the world showed higher mortality and morbidity in men than in women. This result may be explained by several factors, such as higher expression of angiotensin-converting enzyme 2 receptors for coronavirus in men than in women and sex-based immunological differences driven by sex hormones and the X chromosome. 34 Furthermore, a large part of this difference in the number of deaths is caused by lifestyle, such as higher smoking levels among men than women. 35 The same outcomes regarding the association between ABO blood groups and sex were found by other researchers.34,36,37
In the current study, because of the increased severity of COVID-19 infection in hospitalized patients, the duration of illness, need for plasma transfusion, and mortality rate were significantly higher among hospitalized than non-hospitalized patients.
Regarding symptoms, our results revealed that 37.2% of patients with COVID-19 initially presented with more than one symptom. For mono-symptomatic cases, myalgia was the most common initial presentation. This result was consistent with that reported by Rothan and Byrareddy, who mentioned that early COVID-19 symptoms included a combination of headache, fever, dry cough, dyspnea, and myalgia, which were similar to the symptoms of another viral respiratory disease. 35
Conclusions
We found that there was no impact of ABO blood group/RhD antigen distribution on our study outcome because there was no significant association between symptomatic COVID-19 infection and any ABO group or RhD antigen type. As a marker of severity, ABO grouping did not impact hospitalization. The rate of mortality in male patients with COVID-19 was higher than that in female patients. The most frequent first presenting symptom was myalgia. The limitations of our study include its retrospective nature and the need for contacting patients by phone when there were missing data in their case sheets. We studied the original variant of SARS-CoV-2 that was first detected in Wuhan, China; therefore, our results may differ from subsequent studies on other variants that emerged later. Additionally, we experienced difficulties in terms of direct contact with patients or their relatives because of COVID-19-related restrictions and the global lockdown.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221110493 - Supplemental material for Correlation of SARS-CoV-2 infection severity with ABO blood groups and RhD antigen: a case-control study
Supplemental material, sj-pdf-1-imr-10.1177_03000605221110493 for Correlation of SARS-CoV-2 infection severity with ABO blood groups and RhD antigen: a case-control study by Nazaneen Akbar Omer, Sarwer Jamal Al-Bajalan, Heshu Sulaiman Rahman, Maryam Salih Mohammed in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221110493 - Supplemental material for Correlation of SARS-CoV-2 infection severity with ABO blood groups and RhD antigen: a case-control study
Supplemental material, sj-pdf-2-imr-10.1177_03000605221110493 for Correlation of SARS-CoV-2 infection severity with ABO blood groups and RhD antigen: a case-control study by Nazaneen Akbar Omer, Sarwer Jamal Al-Bajalan, Heshu Sulaiman Rahman, Maryam Salih Mohammed in Journal of International Medical Research
Footnotes
Acknowledgements
The authors greatly appreciate the staff of the Qala (at Kalar province), Shaheed Aso, and Shaheed Dr. Hemn Hospitals for Corona in Sulaimaniyah city, Iraqi Kurdistan Region for their kind help and support during this study.
Author contributions
N.A.O. and M.S.M. conducted the research, S.J.A. analyzed the data and performed the statistical analysis, and H.S.R. edited, revised, corrected, paraphrased, and submitted the manuscript. All authors participated in the writing of the original draft of the manuscript.
Declaration of conflicting interest
The authors declare that they have no competing interests.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
