Abstract
Pneumatosis cystoides intestinalis (PCI) is a rare disease that most frequently occurs in the large and small intestine and has no obvious clinical symptoms. The main pathological feature is the presence of air-filled cysts in the intestinal submucosa, intermuscular wall, and subserous membrane. Conservative treatment is the first choice when no serious complications are present, whereas timely surgical treatment is needed for serious and life-threatening complications. This report presents the clinical and pathological analysis of PCI in a man in his early 90s. The patient was hospitalized because of acute abdomen and diagnosed with perforation of the sigmoid colon due to PCI with schistosomiasis after emergency surgery. Emergency partial sigmoid colon resection and permanent colostomy were performed under general anesthesia. Preoperative diagnosis of PCI is difficult because of the nonspecific clinical manifestations and endoscopic findings, and missed diagnosis and misdiagnosis easily occur. Pure PCI has no specific symptoms and does not require special treatment, and there is a lack of special treatment methods in clinical practice. However, when PCI is combined with other intestinal diseases such as schistosomiasis enteropathy, intestinal perforation is likely to occur, leading to severe acute abdomen with the need for prompt surgical treatment.
Keywords
Background
Pneumatosis cystoides intestinalis (PCI), which was first described by Du Vernoi in 1730, 1 is a rare disease without obvious clinical symptoms that most frequently occurs in the large and small intestine. 2 Preoperative diagnosis of PCI is difficult, and it may be easily missed or misdiagnosed. 3 Conservative treatment is the first choice when no serious complications are present; however, timely surgical treatment is required for life-threatening complications. The main pathological feature of PCI is the presence of air-filled cysts in the intestinal submucosa, intermuscular wall, and subserous membrane. 4
Schistosomiasis enteropathy can affect any part of the intestine. Schistosomes enter the skin after the human body comes into contact with infested water and mainly parasitize the portal vein and inferior mesenteric vein, especially the superior hemorrhoidal vein and inferior mesenteric vein; thus, most lesions are located in the sigmoid colon and rectum. 5 In the chronic stage of infection, eggs can be deposited on the intestinal wall or veins within the intestinal wall, leading to local fibrosis, insufficient blood supply to the mucosa, or even chronic enteritis. 6 Schistosomiasis enteropathy induces both acute and chronic inflammatory reactions. The most characteristic manifestation is the presence of gray-yellow or yellow-white schistosome nodules, similar to those in pseudomembranous enteritis and colitis. 7
The co-occurrence of PCI and schistosomiasis enteropathy is rare. Both of these diseases can cause accumulation of intestinal wall lesions and ultimately lead to perforation of the sigmoid colon. Therefore, patients who have PCI combined with other diseases that may lead to organic changes in the intestinal wall are at high risk of intestinal wall perforation. 8
Case presentation
A man in his early 90s presented with a >1-day history of sudden-onset abdominal pain, and he was admitted to the emergency department of our hospital. No obvious etiologies were found, and the pain became progressively exacerbated with no relief in any position or after eating. The patient also experienced nausea without vomiting. Abdominal computed tomography (CT) revealed free gas in the abdominal cavity and digestive tract perforation as indicated by the direction of the red arrows in Figure 1(a). He was diagnosed with peritonitis with digestive tract perforation and was admitted for further treatment. His medical history included coronary heart disease and long-term diabetes for which he was being medicated with aspirin and glucose-lowering drugs (metformin hydrochloride sustained-release tablets), but he reported no history of similar abdominal pain, gallstone disease, or kidney stones. Notably, however, the patient had a long-term history of constipation. Physical examination revealed a flat abdomen, no abdominal breathing, pain in the upper and right lower abdomen, muscle guarding, rebound pain, and weak intestinal tone.
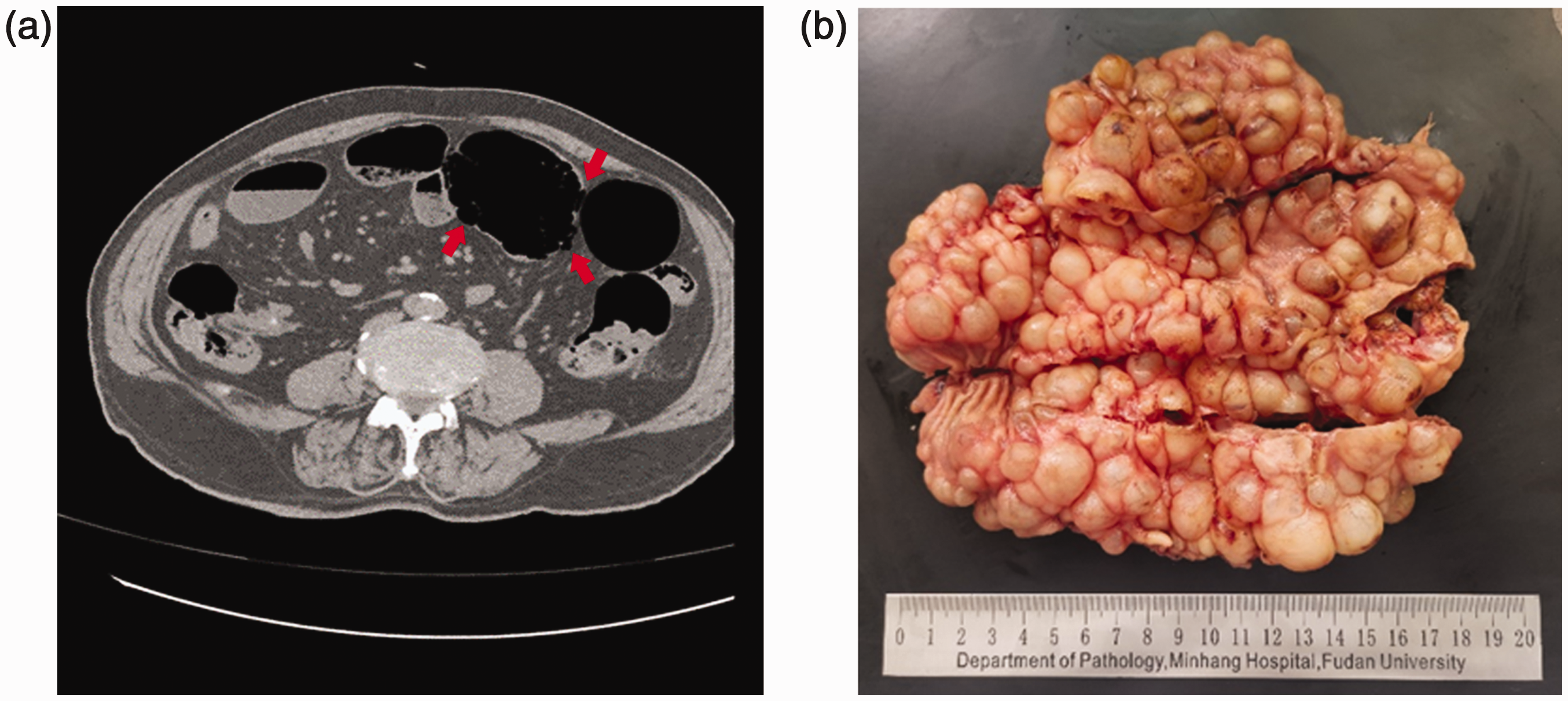
Computed tomography manifestations of pneumatosis cystoides intestinalis and photographs of postoperative specimens. (a) Imaging findings of digestive tract perforation with pneumatosis cystoides intestinalis and (b) Intestinal mucous membrane surface covered with small, soap bubble-like cysts.
There was no obvious mass in the abdomen. Auxiliary examination via abdominal CT indicated the presence of free gas in the abdominal cavity, suggesting digestive tract perforation. The small intestine had expanded secondary to gas and fluid accumulation; minimal inflammatory exudation of the lower right bowel was detected, and intestinal schistosomiasis was diagnosed. Because CT showed acute peritonitis caused by gastrointestinal perforation, a laparotomy was performed. During the operation, at the end of the reflux, the colon was irregular and slightly thickened with inflammatory changes. Air-filled cysts under the colonic mucous membrane and serosa were observed approximately 15 cm from the middle sigmoid colon. The sizes of the cysts varied, and the maximum diameter was approximately 2 cm. A 0.5-cm perforation surrounded by pus was also observed. No metastatic nodules were found in the pelvic cavity, liver, stomach, abdominal aorta, or abdominal wall.
Pathological examination
The surface of the intestinal mucous membrane was covered with small, soap bubble-like cysts. The cyst cavities had a diameter of 0.5 to 2.0 cm and were visible on the surface mucosa, muscle wall, and serosa of the colon (Figure 1(b)). Microscopic examination results indicated variously sized round or fissure-like cystic cavities with a clear boundary under the emphysematous mucous membrane as well as thinning of the mucous membrane. For the most part, the cyst wall was not lined with epithelium, and part of it was covered with flat or cuboidal epithelial cells (Figure 2(a)). The sac wall contained multinucleated giant cells. Deposition of schistosome eggs was observed in the intestinal wall (Figure 2(b)).

Morphological features of pneumatosis cystoides intestinalis under microscopic examination after hematoxylin–eosin staining. (a) Part of the sac wall was covered with flat or cuboidal epithelial cells and (b) Schistosome egg deposition in the intestinal wall.
Immunohistochemical staining
The endothelial cell markers CD34 (Figure 3(a)), CD31 (Figure 3(b)), and D2-40 (Figure 3(c)) were expressed in the epithelium lining the cyst wall as shown by immunolabeling. The multinucleated giant cells were positive for CD68 (Figure 3d). Pathological examination indicated PCI with schistosomiasis of the sigmoid colon.
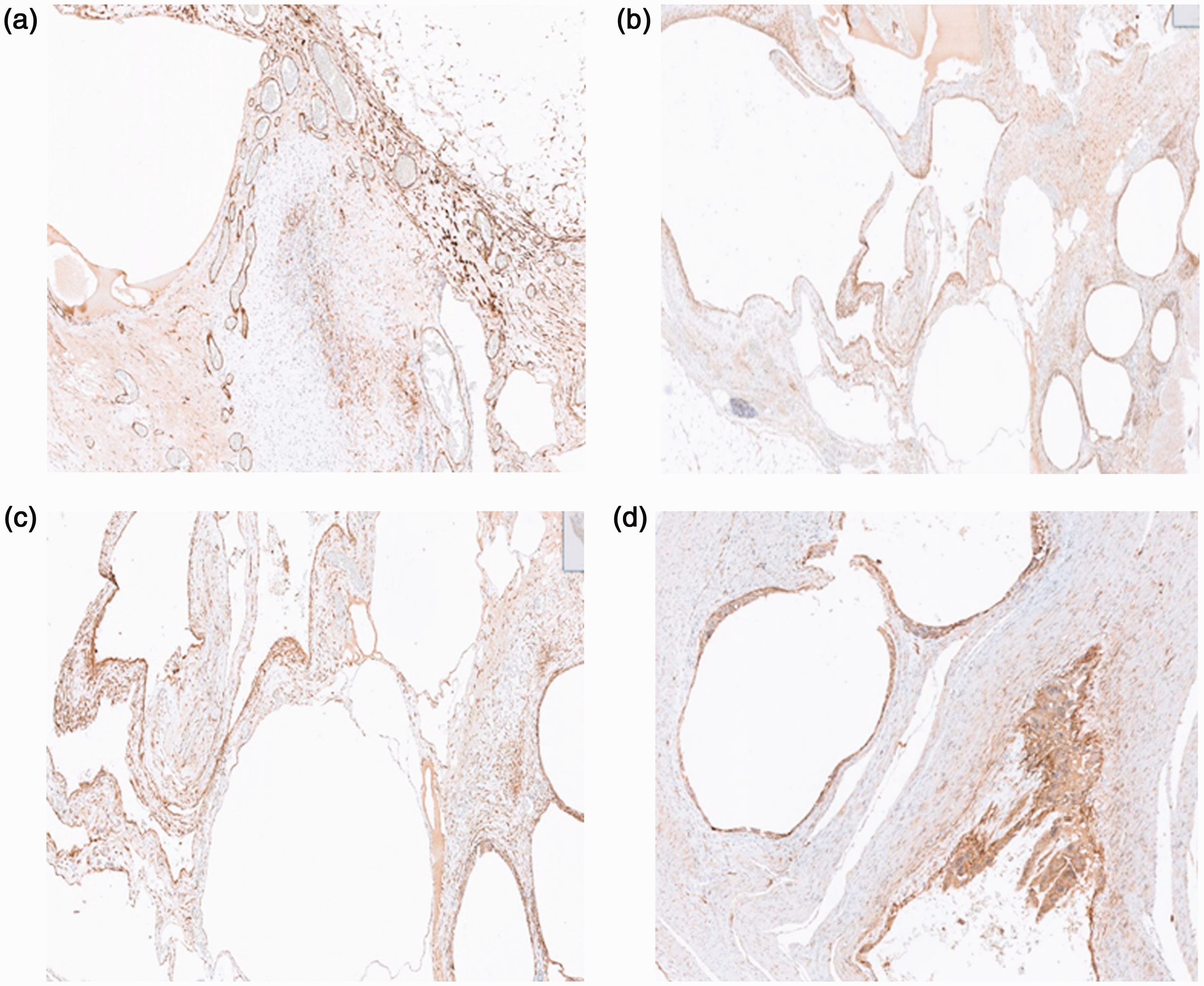
Expression of schistosomiasis-related immunohistochemical staining in pneumatosis cystoides intestinalis tissue. (a) Positive expression of endothelial cell marker CD34. (b) Positive expression of endothelial cell marker CD31. (c) Positive expression of D2-40 and (d) Positive expression of CD68 in multinucleated giant cells.
Postoperative follow-up
No recurrence was noted during follow-up, and the patient’s general condition was good 6 months postoperatively.
Discussion and conclusions
PCI is characterized by benign lesions. It can be classified as infant- or adult-type PCI depending on the age at onset, which is considerably varied. Infant-type PCI is observed at ≤2 years of age, whereas adult-type PCI is observed from 40 to 70 years of age. According to its pathogenesis, PCI is divided into primary and secondary types, accounting for 15% and 85% of all cases, respectively; the disease is attributed to various intestinal diseases and injuries.
The pathogenesis of PCI remains unclear, but three main hypotheses have been proposed. 9 First, the mechanical theory states that an increase in intraluminal pressure causes mechanical damage and mucosal rupture of the intestinal wall, leading to the migration of gas from the gastrointestinal cavity to the intestinal wall. Second, the pulmonary theory considers that chronic lung diseases such as chronic obstructive pulmonary disease, asthma, and interstitial pneumonia lead to alveolar rupture, causing mediastinal emphysema and the release of gas along the aorta and mesenteric blood vessels into the intestinal wall. Finally, the bacterial theory proposes that aerogenic bacteria penetrate the intestinal mucosal barrier, ferment in the intestinal wall, and produce gas.
PCI can occur in any part of the digestive tract,10–12 including the small intestine, colon, esophagus, intestinal membrane, liver and stomach ligaments, abdominal lymph nodes, and vaginal walls; however, it most commonly occurs in the sigmoid colon. Because the clinical symptoms PCI are nonspecific, preoperative diagnosis is difficult and may lead to undiagnosed or misdiagnosed PCI. With the improvements in imaging and endoscopic technology in recent years, imaging and endoscopic examinations have become effective means of aiding in preoperative diagnosis. However, histologic examination remains the best technique for a direct diagnosis. With respect to the imaging findings of PCI, 13 X-ray films show a wave-like cystic area of inflammatory fluid exudation on the intestinal edge; when the cyst ruptures, free gas can be seen under the diaphragm. Endoscopic examination is the most intuitive imaging technique, showing a submucosal polyp-shaped focus of emphysema protruding into the cavity. However, endoscopy can only be used to observe cysts at the colonic mucosal layer, not the manifestations of cysts in the colonic muscular layer or colonic serosa. Therefore, preoperative diagnosis should be combined with imaging and endoscopy to provide reasonable treatment.
PCI involves benign lesions and has a good prognosis. The principle of treatment is conservative endoscopic therapy. Surgical treatment is considered a supplement to comprehensive treatment. If the condition does not lead to intestinal obstruction, perforation, bleeding, or other life-threatening complications, surgical treatment is not required. Historically, PCI was often managed via conservative therapy; however, this standard has changed in recent years. Wang et al. 14 used waterless alcohol to dehydrate tissue cells, thus enabling protein solidification, blood vessel contraction, endothelial cell degeneration, and other phenomena. After aspirating the gas from the cyst with an injection needle and injecting 0.5 to 1.0 mL of anhydrous alcohol, the cyst collapsed. This method was used to successfully treat five cases of PCI with no bleeding or perforation. Surgery is performed under the following conditions: 15 patients fail to improve after conservative and endoscopic treatment; a wide range of lesions are present in the intestines; polyps and/or tumors are present; or infection, hemorrhage, perforation, obstruction, or torsion of the digestive tract is present. Our patient was treated by emergency intestinal perforation surgery with high efficacy and a satisfactory outcome. Follow-up results were good at 6 months postoperatively.
The patient in the present case had PCI with schistosomiasis enteropathy as well as a long-term history of constipation. The combination of multiple factors resulted in perforation of the sigmoid colon. CT was the most direct diagnostic technique, and timely surgical treatment was the most effective treatment.
It is important to note that surgical treatment should not always be the first choice if the disease can be managed conservatively. In our case, surgery was necessary because of acute abdomen caused by intestinal perforation. Surgical treatment should be performed strictly to provide a higher likelihood of improvement than other therapeutic options and should not be performed only to avoid conservative treatment. In short, treatment for PCI should be selected according to the patient’s age, history, symptoms, lesion scope, and possible complications. Physicians should increase their awareness of this disease because early detection, diagnosis, and treatment are crucial to avoid missed diagnosis and misdiagnosis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221105160 - Supplemental material for Pneumatosis cystoides intestinalis accompanied by schistosomiasis: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221105160 for Pneumatosis cystoides intestinalis accompanied by schistosomiasis: a case report by Chunyan Yuan, Fei Teng, Zhongyue Huang, Qiping Liu, Ling Du, Xiaoli Xie, Xuebing Jiang and Xia Sheng in Journal of International Medical Research
Footnotes
Acknowledgements
We sincerely thank the staff members of the departments of gastrointestinal surgery and radiology at our hospital for providing information.
Author contributions
CY, FT, and ZH contributed equally to this work.
CY and XS wrote the manuscript and acquired information of the patient.
ZH and QL performed the literature review and follow-up.
XS, LD, XX, and XJ revised the manuscript.
All the authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Consent for publication
The patient did not provide written consent for publication because he lived a great distance from the hospital, and returning to sign the required forms was difficult. Instead, verbal consent for publication was obtained by telephone. All details of the patient have been de-identified.
Ethics approval and consent to participate
This study was approved by the Human Research Ethics Review Board of Minhang Hospital, Fudan University. The patient provided written consent for treatment. The reporting of this study conforms to CARE guidelines. 16
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of Shanghai Minhang District Science and Technology Committee (Grant No. 2020MHZ076), the Fundamental Medical Project of Minhang Hospital of Fudan University Project Foundation (Grant No. 2022MHBJ03), and the Project of Shanghai Minhang District Characteristic Project (Grant No. 2020MHFC03).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
